Outline
Normal Sonographic Anatomy of the Fetal Face, 243
Craniofacial Anomalies, 245
Typical Facial Clefts, 245
Atypical Facial Clefts, 250
Orbital and Ocular Defects, 252
Micrognathia and Retrognathia, 255
Macroglossia, 257
Tumors of the Face, 257
Ears, 262
Craniosynostosis, 262
Anomalies of the Neck, 265
Conclusions, 268
Summary of Key Points
- •
The diagnosis of a typical orofacial cleft involving the palate can be suspected as early as the first trimester by the presence of an anechoic space or discontinuity in the retronasal triangle (RNT) view.
- •
Using two-dimensional (2D) ultrasound, identifying typical clefts in the second trimester is best done by utilizing the coronal view of the nose and lips and the axial view of the fetal palate.
- •
Three-dimensional (3D) ultrasound, particularly the rendering function, can be helpful in the diagnosis of clefts of the hard and soft palate as well as in showing the surface rendered images to the family.
- •
Hypertelorism and hypotelorism, which can be defined by standard tables, are associated with a range of genetic syndromes.
- •
Micro- and retrognathia are identified in the sagittal profile view, and the diagnosis can be made subjectively or objectively with the use of facial angles.
- •
Craniosynostosis, the premature closure of cranial sutures, should be suspected in the presence of significant dolichocephaly or brachycephaly and may be associated with other anomalies or syndromes.
- •
Nuchal cystic hygromas are often associated with aneuploidy, with a higher frequency of trisomy 21 in the first trimester and Turner syndrome in the second trimester.
- •
Facial tumors and neck masses can compromise the fetal airway at birth and may require the use of an ex utero intrapartum treatment (EXIT) procedure at the time of delivery.
Normal Sonographic Anatomy of the Fetal Face
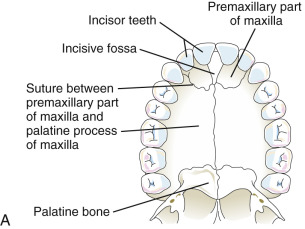
The fetal face can be evaluated in three different planes using 2D ultrasound—sagittal, axial or transverse, and coronal ( Fig. 10-1 ). Each plane has a unique contribution to the evaluation of fetal craniofacial anatomy. The sagittal plane allows for assessment of the fetal profile and can illustrate any dysmorphism of the forehead or nose and the presence of the nasal bone as well as the positioning of the fetal chin to evaluate for micrognathia or retrognathia ( Table 10-1 ). The axial or transverse plane is integral at two different levels ( Table 10-2 ). The first key image is that of the orbits and eyes, which can be obtained caudad to the image displaying the biparietal diameter. Romero and associates have published nomograms for the various ocular parameters including binocular distance, interocular distance, and ocular diameter ( Table 10-3 ). Moving the transducer further caudad on the fetal head, one arrives at the level of the superior lip and palate followed by the fetal mandible.
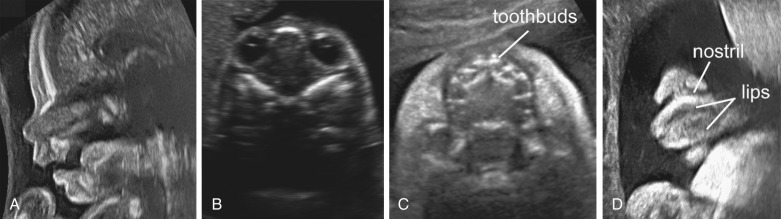
| Structure | Normal Appearance |
|---|---|
| Forehead | Almost linear immediately above the articulation between the nasal bones and the frontal bone, followed by a smooth backward bend. This view allows measurement of the thickness of the frontal skin (at the level of the middle of the frontal bend). |
| Nasal bones | Oblique along a frontocaudal direction. This view allows measurement of the length of the nasal bones and of the superior facial angle (angle between the vertical section of the frontal bone and the nasal bones). |
| Nasal soft tissues | The columella is oblique or horizontal but should not be vertical. |
| Upper lip | The philtrum is linear and should present no bulging. This view allows measurement of the length of the philtrum. |
| Secondary palate | Thick echoic line, beginning at the alveolar level, and extending horizontally backward. Its middle is marked by a notch, present on both the superior and inferior edges, and corresponds to the transverse palatal suture. The notch is mostly visible on the superior edge. |
| Oral cavity | The tongue is slightly oblique upward (10-15 degrees). Its tip lies immediately behind the alveolar ridge. |
| Inferior lip | Rests edge to edge with the upper lip. Both lips are arranged along the same axis; there should be no anteroposterior shift between them. |
| Chin | At the level of the vertical line traced on the prefrontal skin (esthetic vertical line of the face). |
| Structure | Normal Appearance |
|---|---|
| Orbits | The interorbital axis is perpendicular to the strict sagittal axis. This view allows measurement of the inner and outer interorbital lengths. |
| Nasal septum, molar | The nasal septum is perpendicular to the axial plane. The two malar arches are symmetric with regard to arches to the nasal septum. |
| Upper lip, maxilla | There is no lack of continuity of the upper lip. The maxilla appears as a regular, U-shaped echoic bend. The alveolus and tooth buds appear as hypoechoic spots regularly distributed along the alveolar ridge. There is no shift between adjacent alveoli. The hard palate appears as an echoic structure, with a complex shape. The anterior part is semicircular and lies immediately posterior to the alveolar ridge. The posterior part presents as a rectangular figure, with a notch on its distal side. This view allows measurement of the size of the maxilla. |
| Oral cavity | The tongue occupies the totality of the oral cavity and is glued to the alveolar ridge. Posteriorly, the tongue ends at the oropharynx level. This view allows measurement of the width and length of the tongue. |
| Mandible | Appears as a regular V-shaped echoic image. Both hemimandibles are almost rectilinear. The symphysis menti is clearly visible. The alveoli appear as regularly distributed hypoechoic spots. This view allows measurement of the size of the mandible (e.g., mandible width and computation of mandible width/maxilla width ratio). |
| Age (Weeks) | BINOCULAR DISTANCE (mm) | INTEROCULAR DISTANCE (mm) | OCULAR DIAMETER (mm) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 5th | 50th | 95th | 5th | 50th | 95th | 5th | 50th | 95th | |
| 11 | 5 | 13 | 20 | — | — | — | — | — | — |
| 12 | 8 | 15 | 23 | 4 | 9 | 13 | 1 | 3 | 6 |
| 13 | 10 | 18 | 25 | 5 | 9 | 14 | 2 | 4 | 7 |
| 14 | 13 | 20 | 28 | 5 | 10 | 14 | 3 | 5 | 8 |
| 15 | 15 | 22 | 30 | 6 | 10 | 14 | 4 | 6 | 9 |
| 16 | 17 | 25 | 32 | 6 | 10 | 15 | 5 | 7 | 9 |
| 17 | 19 | 27 | 34 | 6 | 11 | 15 | 5 | 8 | 10 |
| 18 | 22 | 29 | 37 | 7 | 11 | 16 | 6 | 9 | 11 |
| 19 | 24 | 31 | 39 | 7 | 12 | 16 | 7 | 9 | 12 |
| 20 | 26 | 33 | 41 | 8 | 12 | 17 | 8 | 10 | 13 |
| 21 | 28 | 35 | 43 | 8 | 13 | 17 | 8 | 11 | 13 |
| 22 | 30 | 37 | 44 | 9 | 13 | 18 | 9 | 12 | 14 |
| 23 | 31 | 39 | 46 | 9 | 14 | 18 | 10 | 12 | 15 |
| 24 | 33 | 41 | 48 | 10 | 14 | 19 | 10 | 13 | 15 |
| 25 | 35 | 42 | 50 | 10 | 15 | 19 | 11 | 13 | 16 |
| 26 | 36 | 44 | 51 | 11 | 15 | 20 | 12 | 14 | 16 |
| 27 | 38 | 45 | 53 | 11 | 16 | 20 | 12 | 14 | 17 |
| 28 | 39 | 47 | 54 | 12 | 16 | 21 | 13 | 15 | 17 |
| 29 | 41 | 48 | 56 | 12 | 17 | 21 | 13 | 15 | 18 |
| 30 | 42 | 50 | 57 | 13 | 17 | 22 | 14 | 16 | 18 |
| 31 | 43 | 51 | 58 | 13 | 18 | 22 | 14 | 16 | 19 |
| 32 | 45 | 52 | 60 | 14 | 18 | 23 | 14 | 16 | 19 |
| 33 | 46 | 53 | 61 | 14 | 19 | 23 | 15 | 17 | 19 |
| 34 | 47 | 54 | 62 | 15 | 19 | 24 | 15 | 17 | 20 |
| 35 | 48 | 55 | 63 | 15 | 20 | 24 | 15 | 18 | 20 |
| 36 | 49 | 56 | 64 | 16 | 20 | 25 | 16 | 18 | 20 |
| 37 | 50 | 57 | 65 | 16 | 21 | 25 | 16 | 18 | 21 |
| 38 | 50 | 58 | 65 | 17 | 21 | 26 | 16 | 18 | 21 |
| 39 | 51 | 59 | 66 | 17 | 22 | 26 | 16 | 19 | 21 |
| 40 | 52 | 59 | 67 | 18 | 22 | 26 | 16 | 19 | 21 |
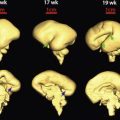
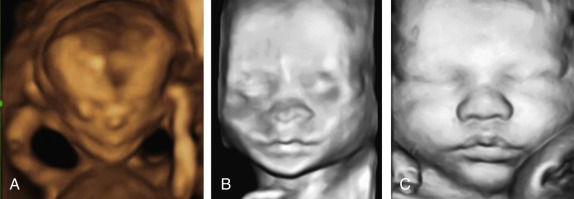
3D ultrasound has an integral role in the evaluation and diagnosis of craniofacial anomalies. Though not yet validated for routine use in low-risk pregnancies, its use in the context of a detected or suspected craniofacial anomaly is quite apparent. A single midsagittal volume obtained with 3D ultrasound can allow for the evaluation of the sagittal, axial, and coronal views using multiplanar reconstruction. The rendering mode can create a realistic image of the exterior facial features as well as a 3D image of the fetal palate. In order to obtain a volume, a pocket of amniotic fluid needs to be present in front of the fetal face. These techniques can be applied to the routine anatomic survey in the midtrimester as well as the late first trimester. In addition to being used as a diagnostic modality, 3D ultrasound can also help to illustrate the appearance of a particular anomaly and to display the location and severity of the anomaly to consulting surgical specialists as well as families.
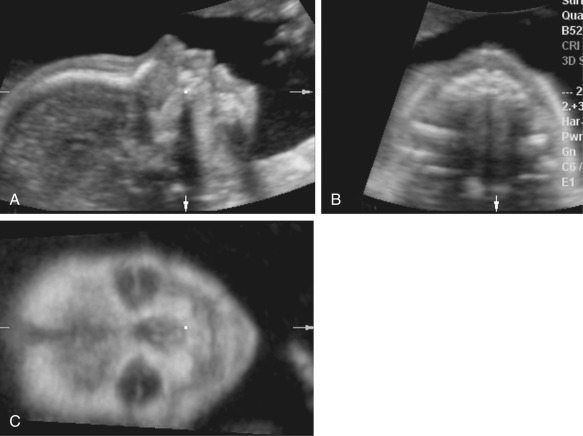
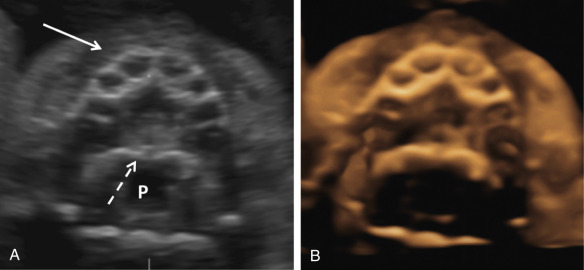
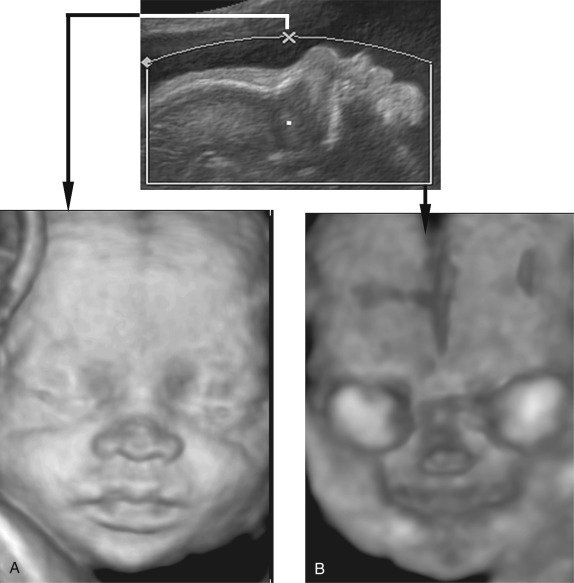
As discussed previously, there are multiple displays of 3D ultrasound that can be used to explore a craniofacial anomaly. The multiplanar display allows the simultaneous display of three perpendicular planes—namely, sagittal, axial, and coronal—of the fetal head and face ( Fig. 10-2 ). These planes can be manipulated by the sonographer to highlight the region of interest. The surface rendering mode can be used to create a model of a frontal view of the fetal face ( Fig. 10-3 ). The maximum and transparent modes can be mixed to evaluate the hyperechoic bony structures including the fetal sutures, which can be useful in the diagnosis of craniosynostosis ( Fig. 10-4 ). The same volume may be viewed with both surface rendering and skeletal displays.
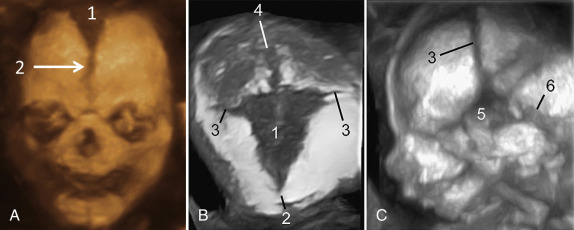
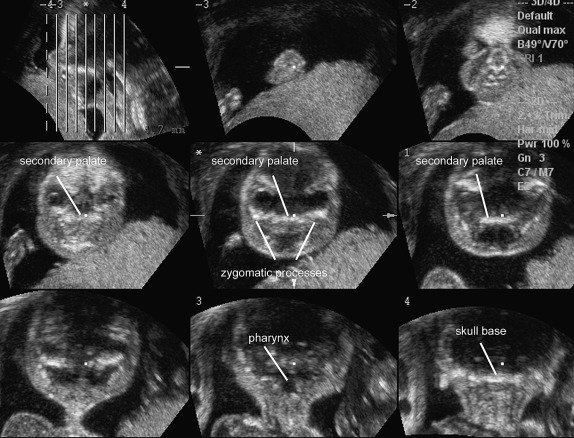
Abnormalities of the fetal hard palate, particularly the secondary palate, can be challenging to evaluate with 2D ultrasonography. Acquiring a volume in the axial plane and then rendering the hard palate from a viewing plane inferior to the palate in an axial plane can be helpful ( Fig. 10-5 ); this approach is a modification of the “flipped face” reported by Platt and colleagues and by Faure and coworkers. Other techniques that have been used include the “reverse face” view using a coronal plane through the hard palate and oblique face views ( Fig. 10-6 ). Parallel slice imaging can use a single 3D volume to create multiple parallel slices, similar to a computed tomography (CT) scan, which can be useful in diagnosing abnormalities of the fetal hard palate ( Fig. 10-7 ). Magnetic resonance imaging (MRI) can be helpful in selected cases to better elucidate fetal craniofacial clefts, particularly when there is a concern for associated neurologic abnormalities.
Craniofacial Anomalies
Craniofacial anomalies are relatively common findings, both in prenatal diagnosis as well as in liveborn infants. The basic ultrasonographic survey of the fetal face should include the upper lip, and a more detailed examination could potentially include the profile, nose, orbits and lenses, palate, maxilla, mandible, tongue, and ear position and size. The prenatal sonographic diagnosis of a craniofacial anomaly should indicate the need for a more detailed full anatomic examination to evaluate for concomitant anomalies. In addition, patients should be offered prenatal diagnostic testing to evaluate for associated genetic syndromes.
The accuracy of ultrasonographic diagnoses of craniofacial anomalies varies by the patient population as well as the expertise and experience of the institution and examiner. A systematic review of 21 studies cited a large range in the prenatal detection rates for orofacial clefts, from 0% to 73%, in low-risk women with 2D ultrasound. In a more contemporary study of a population of more than 30,000 women, both low and high risk, the sensitivity for cleft lip with or without cleft palate reached 88%. In high-risk women, in whom 3D ultrasound was used as an adjunct to 2D imaging, the detection rate in specialized centers has been quoted as high as 100%. 3D ultrasound has much to offer in the elucidation of the detailed anatomy and extent of craniofacial anomalies.
Typical Facial Clefts
Orofacial clefts are relatively common and occur in 1 in 700 live births. The rate varies by ethnicity, with higher rates seen in Asian and Native American populations and significantly lower rates in African populations. Typical facial clefts can occur as isolated cleft lip (CL), isolated cleft palate (CP), or as a combination of both cleft lip and cleft palate (CL+CP). They are often further subdivided into the categories of CL±CP and isolated CP. Clefts most often run from one or both of the nostrils to the central part of the posterior palate. An in-depth anatomic classification system was proposed by Tessier in 1976.
Of all orofacial clefts, CL with CP represents 50% of cases, isolated CL 25% of cases, and isolated CP 25% of cases. Clefts involving the lip are more common in males, with a ratio of 2 : 1, and isolated CP clefts are more common in females, also with a ratio of 2 : 1. A large study of almost 50,000 deliveries in Norway with 77 fetuses with CL±CP revealed that among cases of CL±CP, 64% were unilateral and 34% were bilateral, with only 3% being midline. The same study demonstrated that in unilateral clefts, 69% were left-sided and 31% were right-sided, with a ratio of 2.3 : 1. The left-sided predominance has also been shown in other studies; however, there is no definitive anatomic explanation for this finding. Median clefts have unique causes and associations and are detailed in the next section (“ Atypical Facial Clefts ”).
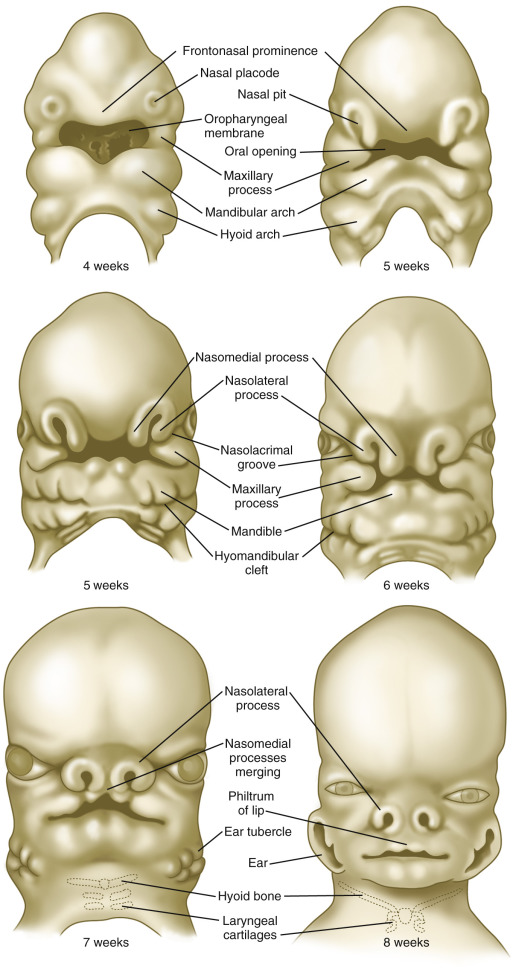
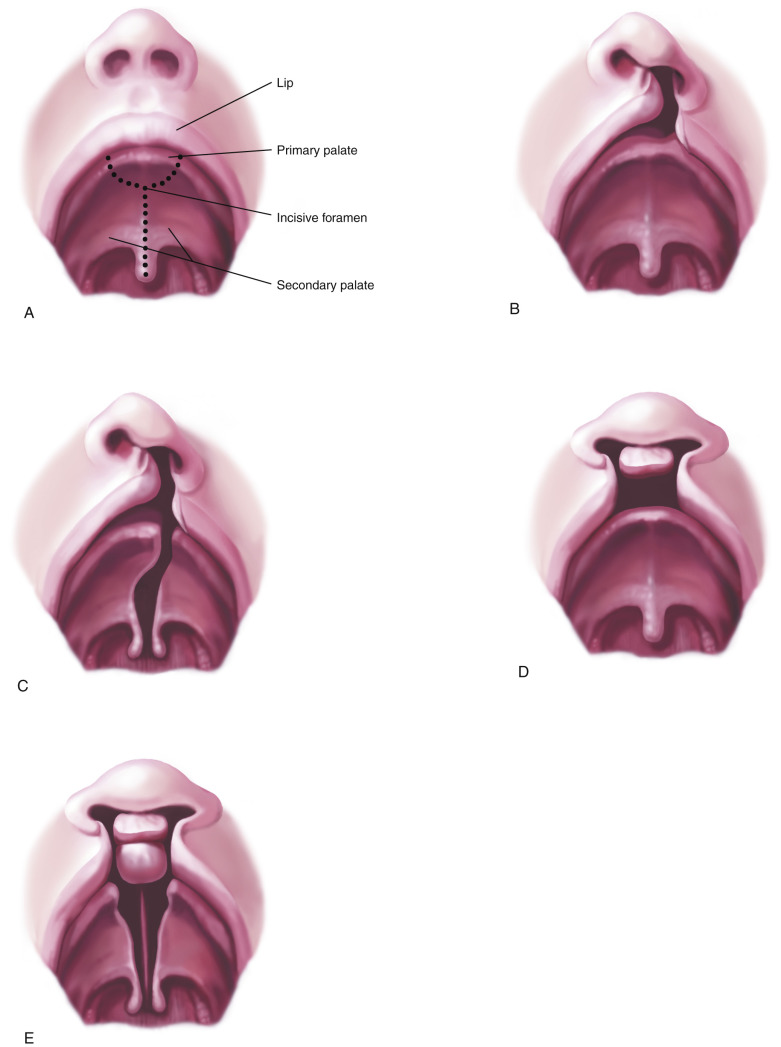
An appreciation for the embryologic development of the face can help in the understanding of the development of orofacial clefts. The face arises from five prominences that surround the mouth—the central frontonasal prominence and the paired maxillary and mandibular prominences. These prominences are composed of neural crest cells and are present by the fourth week of embryogenesis ( Fig. 10-8 ). By the fifth week, the nasal placodes have formed, separating each side of the inferior portion of the frontonasal prominence into lateral and medial nasal processes. The upper lip and primary palate are formed by the end of the sixth week, when the medial nasal processes fuse with each other and the paired maxillary processes. This sixth week is a sensitive time for development, and teratogenic exposures at this time can result in orofacial clefts. The purpose of the secondary palate is to separate the oral and nasal cavities. The development of the secondary palate begins in the sixth week and is completed by the tenth week. The secondary palate is formed from paired palatal shelves that are outgrowths of the maxillary processes. These shelves fuse during the seventh week and differentiate into the hard and soft palate. By the tenth week, the primary palate and nasal septum have fused with the secondary palate ( Fig. 10-9 ). Clefting can occur during multiple stages of the embryogenesis process and results in different anatomic variations depending on the timing and which prominences are affected ( Fig. 10-10 ).

Orofacial clefts are often divided into the categories of syndromic and nonsyndromic clefts. In CL±CP, 70% to 90% of clefts are nonsyndromic, and in isolated CP, 60% to 80% of clefts are nonsyndromic. Isolated CL should be counseled similarly to CL+CP because it has a similar developmental pattern. Table 10-4 reviews some of the more common syndromic associations with orofacial clefts. It is essential that parents with fetuses with the prenatal diagnosis of orofacial clefts be offered further genetic workup with chorionic villus sampling or amniocentesis and evaluation of chromosomal abnormalities with karyotyping and chromosomal microarray.
| Chromosomal Aberrations |
|
| Malformations and Sequences |
|
| Syndromes |
|
All types of orofacial clefts can potentially be associated with other structural anomalies. In one study, 43% of those with CL±CP and 58% of those with isolated CP had associated structural anomalies. Rates of associated anomalies vary, with another study reporting 35% of patients with CL±CP as having an associated anomaly or suspected genetic syndrome. A third study suggested a lower level of association with anomalies, reporting that those with unilateral CL±CP had a rate of associated structural anomalies of 9.8%, whereas bilateral CL+CP had associated anomalies in 25%. Most of these studies are biased by tertiary care subselection; in our practice, we counsel a risk of associated anomalies of approximately 10% when an isolated CL±CP is identified on prenatal ultrasound images.
In addition to genetic causes, orofacial clefts have been linked to several environmental factors and medications. Organic solvent and agricultural chemical exposure may contribute to the risk for cleft anomalies. Antiepileptic medications, including the older generation drugs such as diazepam, phenytoin, and phenobarbital as well as some of the newer generation drugs such as topiramate and lamotrigine have been associated with an increased risk of orofacial clefts in some studies. Early exposure to retinoid medications and corticosteroids has also been linked to fetal craniofacial anomalies. Maternal smoking has been consistently associated with CL±CP with a relative risk of 1.34 and CP alone with a relative risk of 1.22 in a large meta-analysis. There is inconsistent evidence suggesting that folic acid deficiency may yield an increased risk for orofacial clefts and thus that supplementation could potentially decrease the risk of recurrence in a subsequent pregnancy, but this relationship has not been definitively established. The early prenatal use of multivitamins has been associated with a 25% reduction in the risk for CL±CP and a nonsignificant 12% reduction in the risk for CP.
Prenatal diagnosis of clefts can allow parents to choose options for further genetic testing and can also prepare them for the child’s medical course if they continue the pregnancy. In 1981, Christ and Meininger reported the first prenatal ultrasonographic diagnosis of an orofacial cleft. Research has demonstrated that the detection of clefts has improved over time, with one study citing a detection rate of 34% in the period of 1987-1995 and 58% in the period of 1996-2004. A systematic review of 21 studies reported a broad range in prenatal detection rates for low-risk women, from 9% to 100% for CL±CP and 0% to 22% for CP only. Detection of isolated CP in particular continues to be poor, with some large contemporary studies showing a detection rate of 0%.
In the second trimester, CL±CP and CP can be best visualized by utilizing both coronal and axial planes. In the anterior coronal plane, with a view of both the nose and lips, a CL is identified as a defect extending from one nostril to the oral rim ( Table 10-5 ). These defects can cause distortion of the upper lip and nose, which can be seen well with a 3D rendered image ( Fig. 10-11 ). To determine whether the defect extends into the alveolus or the primary palate, the axial plane is the best approach ( Table 10-6 ). This view can demonstrate the extent of the cleft and determine if it also involves the secondary or hard palate ( Fig. 10-12 , Table 10-7 ). As discussed, identifying an isolated CP is quite challenging, and these defects are often missed, even with thorough 2D or 3D ultrasonography. In the case of bilateral CL+CP, the sagittal plane with the fetal profile can be helpful. This view can demonstrate the protrusion of the central palate and lip, often termed the premaxillary mass ( Fig. 10-13 ). The sagittal plane will likely be normal in cases of unilateral CL+CP or with bilateral CL. When any type of orofacial cleft is suspected, 3D ultrasound can often offer a more accurate portrayal of the facial anatomy and specifically the hard and soft palate ( Figs. 10-14 and 10-15 ). As previously reviewed, the “flipped face” and the “reversed face” view allow the examiner to evaluate the posterior hard and soft palates. MRI may better illustrate the extent of orofacial clefting, as it allows for improved visualization of the palate, particularly the hard or secondary palate.