Abstract
This chapter on advanced cardiac ultrasound focuses on describing three-dimensional echocardiography, contrast echocardiography, and deformation imaging by echocardiography. These advanced echocardiographic methods are already implemented in clinical cardiology. However, improvements in the image quality and availability of the methods are needed to fully exploit their clinical potential. Echocardiography as a method is evolving; at the end of the chapter, we describe some of the ongoing and future advancements of the method and potential new clinical applications.
Keywords
Three-dimensional echocardiography, Contrast echocardiography, Tissue Doppler imaging, Speckle tracking imaging, Molecular imaging
2.1
Introduction
The traditional echocardiographic techniques are widely used and form a routine complete echocardiographic examination:
- –
M-mode recordings, guided by two-dimensional (2D) echocardiographic images, are used for quantification of cardiac dimension and timing of rapid cardiac motions [ ]. It is recommended that left ventricular (LV) internal diameters and wall thickness are measured by M-mode since they show excellent temporal resolution and may complement 2D images in separating structures such as trabeculae adjacent to the posterior wall, false tendons, or the moderator band next to the septum from the endocardium [ ]. Linear measurements from M-mode have proven to be reproducible with low intra- and inter-observer variability. However, even with 2D guidance, it may not be possible to align the M-mode cursor perpendicular to the long axis of the ventricle, which is mandatory to obtain a true minor axis dimension measurement. One practical solution to this problem is to use the “anatomical” M-mode technique, which allows the user to steer the M-mode line to any angle that he or she chooses through the 2D image. The disadvantage of this technique is poorer temporal and spatial resolution compared to the real M-mode technique. Furthermore, linear measurements for size and function assessment are problematic when there are marked regional differences in function.
- –
2D echocardiographic images provide detailed anatomic data in a given tomographic image. With 2D images, pathology in the cardiac cavities, myocardial walls, and valvular apparatus are visualized, as are malformations, tumours, and masses. 2D images are important for quantifying chamber volumes and myocardial mass. LV function is traditionally assessed by ejection fraction (EF). End-diastolic and end-systolic volumes are measured by using the biplane method of discs’ summation (modified Simpson’s rule). LVEF is then calculated from these respective 2D LV volumes, making EF a strictly volumetric measure of LV function. In contrast, strain by deformation imaging directly measures the intrinsic contractile properties of the myocardium. Echocardiographic strain has been shown to be a more accurate tool for quantification of myocardial function compared to EF [ ].
- –
Doppler flow imaging, with continuous wave and pulsed wave, is analyzed by spectral analysis and assesses blood flow direction, velocity, and signal amplitude. From this, we can calculate pressure gradients over the cardiac valves and assess diastolic function by interpreting the blood flow patterns.
- –
Colour Doppler flow imaging (multiple sample volumes of pulsed wave Doppler along each ultrasound line) visualizes location, expansion, direction, and timing of blood flow in the cardiac chambers and through the valves or shunts.
These traditional parameters are first-line measures and provide important diagnostic and prognostic information that cannot be entirely replaced by more advanced echocardiographic techniques. In general, the traditional measures have high specificity. If they show evidence of true pathology, given acceptable image quality, there is often no further need for more advanced techniques. For example, EF has excellent diagnostic and prognostic value when myocardial function is severely depressed and EF is low (< 35%). The advanced imaging techniques have their strength in having higher sensitivity for minor pathologic changes. Echocardiographic strain may show subtle reduction in LV function before symptoms appear and EF falls and contrast echocardiography may reveal early regional dysfunction in a patient with poor image quality on normal 2D images.
In this chapter of advanced echocardiography, we will describe three-dimensional echocardiography, contrast echocardiography, and deformation imaging by ultrasound. We will end the chapter with a section describing promising echocardiographic methods with the potential of obtaining clinical importance in the near future.
2.2
Three-dimensional echocardiography [ , ]
Three-dimensional (3D) echocardiography represents a large and important advancement in cardiac ultrasound. Real-time 3D acquisition and imaging of the heart from any preferable angle have become possible with advancements in transducer and computer technologies. 3D echocardiography is especially useful and applies additional information over 2D echocardiography in the assessment of:
- –
Cardiac volumes and mass independently of geometric assumptions
- –
Heart valves
- –
Regurgitant lesions, jets, and shunts (3D echocardiography colour Doppler)
In addition, the 3D technique has theoretical benefits compared to 2D in:
- –
LV regional function and dyssynchrony
- –
3D stress echocardiography
2.2.1
Matrix-array transducer
Developments in hardware and software have resulted in fully sampled matrix-array transducers providing improved real-time imaging of the beating heart. 3D echocardiography matrix-array transducers are composed of about 3000 piezoelectric elements with operating frequency 2–4 MHz for transthoracic echocardiography (TTE) and 5–7 MHz for transoesophageal echocardiography (TEE). The piezoelectric elements are simultaneously active and arranged in a grid fashion. A large number of digital channels are required to connect the fully sampled elements. To avoid excessive power consumption and a wide transducer cable, several small circuits are incorporated into the transducer to allow partial beam forming in the probe. While the transducer is maintained in fixed orientation, the ultrasonic beam can steer automatically in multiple directions. This beam steering and signal processing take place automatically in the scanning probe itself. The newest generation of transducers is becoming smaller, and one single transducer can acquire both 2D and 3D studies of excellent quality. Transducers today provide better side-lobe suppression, less transducer footprint, and increased sensitivity and penetration.
2.2.2
Data acquisition—two modes
2.2.2.1
Real-time 3D echocardiographic imaging
Acquisition consists of multiple pyramidal datasets per second in one single heartbeat. The data are visualized in real-time scanning, beat after beat, while performing the examination as in conventional 2D scanning. Real-time 3D echocardiography imaging has no limitation imposed by rhythm disturbances or respiratory motion, but the temporal and spatial resolution is poorer than 2D imaging.
Real-time 3D narrow volume
Beat-by-beat view with a thicker image plane (slice) than for normal 2D imaging. The thick slice can be rotated.
Real-time 3D zoomed
A full-volume image of an enlarged area of interest. The perspective is like taking a photograph from inside the heart. Spatial and temporal resolutions are poor, and the image must be rotated to gain all anatomical information.
Real-time 3D colour Doppler
Colour Doppler acquisition at lower frame rates than for gray tone data.
2.2.2.2
ECG-triggered multi-beat 3D echocardiographic imaging
Multiple high-resolution narrow volumes of data over consecutive heartbeats are stitched together.
Full volume 3D (gated)
A single volumetric dataset is created from several smaller subvolumes not obtained simultaneously but from consecutive heart cycles. Small subvolumes provide high spatial and temporal resolution. Since the gated image is a sum of 2–7 heart cycles, it is susceptible to artifacts from body movement, respiratory motion, or irregular heart rhythms ( Figure 2.1 ).

2.2.3
Challenges and limitations
The technical aspects of acquiring high-quality 3D echocardiography suitable for diagnostic or therapeutic purposes are similar to those for 2D echocardiography. In addition, incorrect gain settings and respiratory or ECG gating may lead to reconstruction artefacts.
2.2.3.1
Temporal versus spatial resolution
The main trade-off in 3D echocardiography is between temporal resolution and spatial resolution. Increasing spatial resolution, i.e., the number of scan lines per volume, require more time for acquisition, resulting in a falling temporal resolution. By reducing volume size, one can increase temporal resolution by simultaneously maintaining spatial resolution.
2.2.3.2
ECG gating and breath hold
Gated imaging fails when the patient is unable to perform adequate breath holding or when the heart rhythm is irregular. These situations result in stitching artifacts, which are seen as artificial lines in the image, representing interjections between neighbor subvolumes attained from different cardiac cycles. Suboptimal ECG tracings without distinct R-waves deteriorate correct gating of the subvolumes.
The gating artifacts can be minimized in different ways. One should always wait until a regular rhythm appears and be aware that full-volume gated images are not possible in patients with atrial fibrillation. For reducing respiratory artifacts, one asks the patient for breath holding, preferably during the expiration phase.
2.2.3.3
Echo dropouts
Dropouts can result from low gain settings and appear as “holes” in anatomic structures. The dropouts can falsely be identified as defects in the tissue, and they may artificially eliminate anatomic structures, making them invisible. Dropouts cannot be recovered during post processing. To avoid dropouts, it is recommended that both time gain compensation and compression settings are adjusted in the midrange and optimized with slightly higher gain control than at normal 2D acquisition. This allows the greatest flexibility during post processing. Optimizing lateral and axial resolution is just as important during 3D as in 2D acquisition.
2.2.4
Cropping
Cropping can be done in 3D zoom mode and gated full-volume mode to view the interior of the heart. Unlike 2D echocardiography, cropping is essential for 3D echocardiography to enable the visualization of the region of interest and the bypassing of structures in front of this region. Different perspectives of a structure can be obtained by alternating between rotating and recropping. For example, the aortic valve can be seen from the aorta, from the LV or in a long-axis orientation. Cropping and rotation can be done at data acquisition, with the advantage of obtaining better spatial and temporal resolution, but with the disadvantage of the inability to uncrop later. If cropping is performed as part of post processing, one will retain more information, but at a cost of lower spatial and temporal resolution.
The most frequently used cropping planes are the transverse plane, dividing the heart into superior and inferior parts; saggital plane, dividing the heart into right and left parts; and coronal plane, dividing the heart into anterior and posterior parts.
2.2.5
Post acquisition display
The 3D dataset is displayed using 3D visualization software packages:
2.2.5.1
Volume rendering
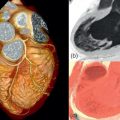
Volume rendering is a technique using different types of algorithms to preserve all 3D echocardiographic information and project it after processing onto a 2D plane for viewing. The volume-rendered 3D echocardiographic dataset can be electronically segmented and sectioned, cropped, and rotated to provide complex spatial relationships in a 3D display. It is especially useful for evaluating valves and nearby structures ( Figures 2.2 and 2.5 ).


2.2.5.2
Surface rendering
Structures or organs are shown to the observer as either a solid or a wireframe (cage) 3D object. It is obtained by manually tracing or using semiautomatic border detection algorithms to trace the endocardium in cross-sectional images generated from the 3D dataset. These contours are combined to generate a 3D shape. Surface rendering provides quantification of chamber volumes (can be displayed graphically throughout the cardiac cycle) and function by EF but often fails to provide detailed information of cardiac structures or textures ( Figures 2.3 and 2.4 ).


2.2.5.3
3D tomographic slices
By slicing the volumetric data, one can obtain multiple simultaneous 2D plane views. This provides the acquisition of different cutting planes from virtually any acoustic window, including 2D cutting planes which are difficult or virtually impossible to obtain with standard 2D transducer manipulation. 2D parallel tomographic slices with uniformly spaced 2D parallel slices can be obtained. The ability to “move through” the 3D dataset in any 2D plane allows accurate measurements of chamber dimensions, valve, or septal defect areas as well as improved evaluation of the morphology and function of structures. During stress echocardiography, the ability to display multiple LV planes simultaneously speeds the image acquisition.
2.2.6
3D colour Doppler
Colour Doppler adds flow information onto the 3D morphology. 3D colour Doppler acquisitions are performed using real-time 3D or multi-beat full volume acquisition.
TEE provides the best colour Doppler image quality and is recommended over TTE for detailed colour flow analysis.
3D colour Doppler analyses provide images for evaluation of distal jets, proximal flow field of valvular flow regurgitation, and flow through septal defects. 3D colour Doppler flow mapping is able to qualitatively visualize size and shape of valvular and paravalvular regurgitations as well as the underlying pathology, enabling exact definition of jet origin, size, propagation, and relationship with adjacent structures. Cropping can be performed according to the intended analysis. For example, to evaluate regurgitant jets, it is recommended that the 3D colour Doppler data be cropped to view two long-axis views (one at the narrowest and one at the broadest width of the jet) and the short-axis view at the level of the vena contracta.
Poor spatial and temporal resolutions are limitations to 3D colour Doppler acquisitions. Currently, live 3D colour Doppler is limited to small volumes with low temporal resolution. As an alternative, multi-beat full volumes deliver higher frame rates up to 40 voxels/second, but are limited by stitching artifacts with displacements between different subvolumes. New 3D colour Doppler technology is being developed to reduce these problems.
2.2.7
Stress echocardiography
The advantages of 3D over 2D stress echocardiography are better visualization of the LV apex, shorter scanning time, rapid acquisition of peak stress images before heart rate declines in recovery, and evaluation of multiple segments from different planes from a single dataset. Disadvantages are lower spatial and temporal resolution than with 2D acquisition. Most stress 3D echocardiography software allows displaying the same view at rest and different stress-stages simultaneously, side-by-side, for comparison. Both physical stress and pharmacologic stress (dobutamine, atropine, or dipyridamine) can be used. Contrast agents can be added for improved endocardial delineation or assessment of myocardial perfusion.
The main and maybe most advantageous difference between 2D and 3D stress echocardiography is the significantly shorter scanning time required at each stress level and for the complete study using 3D echocardiography. There is no need to change the transducer position during apical scanning once the optimal window is identified. Therefore, it is easier to catch the narrow time window of peak stress; this increases the sensitivity to detect ischemic mechanical changes, which is most likely to appear at the highest heart rates. Because of its ability to acquire the entire LV volume within one beat, 3D stress echocardiography is likely to be incorporated into clinical practice in the near future.
2.2.8
Implementation of 3D
Implementing 3D in clinical practice calls for a feasible workflow. So far, the 2D image quality obtained by the 3D probe has been insufficient compared to a modern 2D probe. It is too time-consuming to perform a full 3D echocardiographic examination on top of the traditional 2D examination. The newest 3D TTE and TEE probes deliver 2D echocardiographic images comparable to those delivered by dedicated 2D transducers. Ultrasound companies are developing more accurate automated chamber quantification and automated display of standard 3D and 2D cut planes from each window acquisition. What also needs to be optimized is the way 3D echocardiographic data is stored and recalled for analysis. A 2D exam requires about 300–500 MB of storage space for raw data, while a combined 2D and 3D examination requires 1.5 GB [ ]. The large datasets are a challenge to the local digital systems of laboratories in regard to transmission and overall storage capacity.
2.2.9
Conclusions
3D echocardiography’s full potential is far from being exploited clinically today. With advancements in 3D acquisitions techniques and software improving image quality and reducing the time it takes for image acquisition and post processing, the clinical role of 3D echocardiography will grow. Assessment of the right ventricle has failed with 2D echocardiography; hopefully, 3D imaging will improve the accuracy and reproducibility of RV volume and function assessment. Potential clinical applications where 3D echocardiography may be routinely performed are dyssynchrony assessment, strain imaging, and evaluation of prosthetic valves. Today, low spatial and temporal resolution of larger volumes remain as limitations. Obtaining a real-time single-heartbeat-full volume dataset with high spatial and temporal resolution and live 3D color Doppler of wide angle should be feasible in the future and will increase utility and efficiency of 3D echocardiography.
2.3
Contrast echocardiography [ ]
2.3.1
Ultrasound contrast agents
The ultrasound contrast agents are acoustically active microbubbles consisting of an outer phospholipid or albumin shell encapsulating an inner high-molecular-weight gas. They are strong ultrasound reflectors, i.e., the backscatter they create results in intense echocardiographic signals that are stronger than the ultrasound waves (echoes) from the surrounding tissue and blood cells. Because the agents remain intravascular, they act as red blood cell tracers. The first-generation agents (inner gas was air) were safe but dissipated too quickly to be of clinical value. Second-generation agents consist of a gas that has high density, high molecular weight, and low solubility, making the microbubbles more stable. The bubbles do not accumulate, are biologically inert, and are considered safe.
There are three second-generation contrast agents approved for echocardiographic studies: Definity, Optison and SonoVue (not approved in the US) ( Table 2.1 ).
| Gas | Bubble size | Shell composition | Side effects | |
|---|---|---|---|---|
| SonoVue | Sulfur hexafluoride | 2–8 μm | Lipid | Headache |
| Nausea | ||||
| Chest pain | ||||
| Injection site reaction | ||||
| Hyperglycemia | ||||
| Parasthesia | ||||
| Vasodilation | ||||
| Optison | Perfluoropropane | 3.0–4.5 μm | Albumin | Headache |
| Nausea/vomiting | ||||
| Warm sensation | ||||
| Flushing | ||||
| Dizziness | ||||
| Luminity (definity) | Perfluoropropane | 1.1–2.5 μm | Lipid | Headache |
| Flushing | ||||
| Back pain | ||||
| Rash/uticaria | ||||
| Wheezing | ||||
| Anaphylaxis |
2.3.2
Physics of microbubbles
The microbubbles must be able to resist destruction at normal ultrasound power outputs. Changes to both the inner gas and to a thicker outer shell confer stability and have improved the durability of the bubbles, ensuring the ability to cross the pulmonary capillary bed and reach the LV cavity intact. A microbubble’s ultrasound characteristics depend on its size and the composition of the outer shell and the gas. Backscatter is proportional to the sixth power of the radius, leading to best acoustic signal from the largest bubbles being able to cross through the lung microvasculature. Also, there is a positive linear relationship between concentration and signal intensity. Ultrasound wave pressure compresses and expands the microbubbles, which start to oscillate. The volume expansion of a microbubble is maximal at a specific frequency called the natural resonant frequency and is inversely related to its size. At this resonant frequency, the microbubble scatters and absorbs ultrasound and can present nonlinear vibrations when the insonifying acoustic pressure is high enough. Consequently, the bubble oscillation contains second and higher multiples of the transmitted frequency. The backscattered signal from the microbubble therefore contains not only the fundamental frequency but also harmonic frequencies, most notably at twice the fundamental frequency (second harmonics). This nonlinear reflection is not shown by tissue, allowing the separation of response from the microbubbles from that of surrounding tissue. Finally, as the peak ultrasound wave pressure becomes more intense, many of the microbubbles are disrupted, exhibiting an irreversible, transient, and intense scattering depending on the type of gas released and its dissolution in the liquid. This scattered signal is also highly nonlinear.
2.3.3
Administration
Contrast agents are made available by mixing saline with dry powder of contrast followed by intravenously administration as bolus injection or as a continuous infusion.
Bolus injections are acceptable for studies in which the aim is to improve endocardial border delineation, perform LV opacification, or enhance Doppler signals. It is not recommended in perfusion studies.
Continuous infusion provides a steady-state concentration of microbubbles and reduces the likelihood of artefacts. It also allows calculation of myocardial blood flow, as both microbubble velocity and myocardial blood volume can be calculated if there is a steady-state concentration of contrast. Continuous infusions are mandatory for perfusion imaging and are also often recommended for endocardial border delineation and LV opacification purposes.
2.3.4
Safety of contrast imaging
Several clinical trials have demonstrated safety of using contrast agents with no increase in adverse events. Side effects are usually mild and transient ( Table 2.1 ). Optison and Luminity may be used in patients with acute coronary syndrome. However, serious side effect have been seen at a low estimated incidence of 1:10 000. The Food and Drug Administration highlight the risk of serious cardiopulmonary reactions during or within 30 minutes following the administration of contrast agents and recommend that high-risk patients with pulmonary hypertension or unstable cardiopulmonary conditions should be closely monitored during and for at least 30 minutes after administration. Overall, it is concluded that contrast echocardiography is very safe in clinical practice [ ]. The only absolute contraindications for contrast agent administration are in patients with large intracardial shunts or with known hypersensitivity to the agent.
2.3.5
Mechanical index and imaging modes
Mechanical index (MI) is a measure of the power generated by an ultrasound transducer within an acoustic field.
2.3.5.1
High MI (0.4–0.6) in intermittent static imaging
When microbubbles are exposed to a high MI, they are disrupted almost immediately. With each destructive pulse, high amplitude backscatter rich in harmonics is returned to the transducer, enabling static images. The burst of harmonics provides high sensitivity for contrast detection (seen as bright colour), but the bubbles are irreversibly damaged and can no longer oscillate and thus generate further harmonic signals (i.e., they cannot continue to enhance the image). High MI imaging is therefore not suitable for continuous real-time imaging. Continuous high MI imaging would result in persistent bubble destruction, which prevents detection of microbubbles in the myocardium. Intermittent imaging mode is therefore used where one imaging frame with high MI is created every 1–10 cycles triggered by the ECG. The time between destructive pulses allows the microbubbles to replenish the myocardium. High MI imaging has high sensitivity, as the harmonic signals generated by bubble destruction at high MI are rich and stronger than those emitted at a lower MI. Disadvantages is that one cannot assess wall motion and function because only static images are created at the high MI ultrasound wave and the burst of of harmonics create confounding tissue signals and impairs endocardial delineation. Also, contrast-specific imaging modalities are necessary to suppress these signals (i.e., power Doppler, ultra-harmonics, and power pulse inversion), and it is technically more challenging than low MI imaging. Another disadvantage is that the technique uses large amounts of contrast agent, as microbubbles are repeatedly destroyed and readministered.
2.3.5.2
Low to intermediate MI (< 0.1–0.3) in real-time continuous imaging
Real-time imaging uses sufficiently low MI to ensure that microbubbles will oscillate and resonate but not explode from the continuous exposure to the ultrasound waves. Using low MI generates virtually no harmonic signal from non-contrast-enhanced tissue. Therefore, even low amplitude microbubble backscatter can be isolated from tissue signals for processing, and background subtraction techniques are usually not necessary. The low MI real-time imaging provides the best LV opacification with excellent LV endocardial border enhancement, as it demonstrates a sharp demarcation between the cavity with contrast and the myocardium. With an increased contrast concentration, the display of LV opacification of the myocardium shows myocardial perfusion. Continuous imaging, therefore, enables simultaneous assessment of myocardial wall motion and perfusion in real time. However, frame-rate is held low in order to reduce bubble destruction.
During real-time imaging, all the microbubbles can be intentionally totally destroyed by a “flash” of high MI ultrasound pulses and contrast replenishment from the continuous contrast infusion is then observed to allow qualitative and quantitative assessment of myocardial perfusion. There is less contrast needed than with intermittent high MI imaging. A disadvantage of low MI imaging is that the bubbles generate less acoustic signal, making the method less sensitive for contrast detection than high MI imaging.
2.3.5.3
Recommendations from the European Association of Cardiovascular Imaging (EACVI) [ ]
Given the advances in contrast-specific imaging techniques, the use of low MI imaging for myocardial contrast echocardiography is recommended, as it provides simultaneous assessment of myocardial function and perfusion.
2.3.6
Contrast imaging modes
2.3.6.1
LV opacification and endocardial delineation
The myocardial vessels comprise only 7% of the myocardium. The myocardial opacification is, therefore, always much less intense than chamber opacification, reassuring excellent endocardial delineation and differentiation between LV cavity and myocardium. This provides improved accuracy for calculation of LV volumes and EF, reduced inter- and intraobserver variability, and improved detection of regional wall motion abnormalities both at rest and in stress echocardiography.
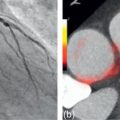
High MI imaging is not an option for LV opacification imaging, as the continuous bubble destruction would deteriorate image quality significantly. Thus, the two options are between low MI and intermediate MI. Intermediate MI (e.g., 0.1–0.3) is feasible as a real-time technique. There is more bubble destruction than with low MI imaging, but the advantage is that the stronger nonlinear oscillations generated by the microbubbles (since they resonate more with increasing MI) yield a stronger acoustic signal. Low MI imaging (< 0.1) generates virtually no tissue signal, which is good in most cases; however, sometimes the returning signals are very weak and thus subtle structural abnormalities may be missed ( Figure 2.6 ).


Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree