Chapter 76 James B. Spies and Ferenc Czeyda-Pommersheim Uterine fibroid embolization (UFE) was first reported in the United States in 1997 as a minimally invasive alternative to hysterectomy and myomectomy for treatment of fibroids.1 Since that time, its effectiveness has been confirmed by other investigators, including in two major randomized comparative trials,2,3 and it has rapidly been incorporated into practice. In 2008, the American College of Obstetricians and Gynecologists, based on good and consistent scientific evidence (level A), concluded that “based on long- and short-term outcomes, uterine artery embolization is a safe and effective option for appropriately selected women who wish to retain their uteri.”4 With this acknowledgment, uterine artery embolization is now widely accepted as an alternative to hysterectomy. Fibroid location is also a consideration in patient selection, since there is some evidence that location may impact outcome5 (Fig. e76-1 There are some specific subtypes of fibroids that may be best managed with surgery. Intracavitary fibroids, particularly when less than 3 cm, may be resectable using hysteroscopic methods. If a gynecologist skilled in hysteroscopic resection is unavailable or fails to remove the fibroid, embolization is certainly effective for this type of fibroid. Large serosal fibroids are often easily resectable by myomectomy, and the decision whether embolization or surgery is preferred depends on a number of factors. In most cases where the patient would like to become pregnant and when there has not been a previous myomectomy, surgery may be preferred. This is particularly true for very large fibroids that are mostly outside the uterine wall. The extreme example of a large narrow-based, completely pedunculated serosal fibroid is usually best treated surgically. This type of fibroid is likely to have relatively less shrinkage than other fibroids and is prone to adhesion formation. Similarly, for a woman with a massive uterus (>24 weeks size), the degree of shrinkage in the uterus may not be sufficient to provide long-term patient satisfaction, and these patients may have a better long-term outcome with surgery.6 Finally, there are some specific fibroid locations, notably broad ligament fibroids and cervical fibroids that anecdotally do not respond to UFE or respond poorly. These fibroids can be a therapeutic conundrum, since they are not easily treated surgically if the uterus is to be spared. Embolic material is the central requirement of the procedure. Although Gelfoam pledgets or slurry have been used in rare cases, there is relatively little study of the effectiveness of the material outside Japan. The vast majority of fibroid embolizations in Western countries have been performed using particulate or spherical embolic materials.7 These are discussed later in the section on technical controversies. The internal iliac artery provides the majority of the blood supply to the pelvis and inner thigh. The internal iliac artery bifurcates to an anterior and posterior division in 57% to 77% of the general population.8,9 Uterine fibroid tumors derive their main peripheral blood supply almost exclusively from the uterine artery.10 The uterine artery is the first or second branch of the anterior division of the internal iliac artery in 51% of the general population. The contralateral anterior oblique view is best used to image this variant. The uterine artery arises from a trifurcation of the internal iliac in approximately 40%, which is best imaged by the ipsilateral anterior oblique projection. In approximately 1% of the population, the uterine artery is absent; in these cases, it is often replaced by the ipsilateral ovarian artery.10 Once femoral access is obtained, a catheter is advanced over the aortic bifurcation to the opposite hypogastric artery. Most practitioners use a single catheter to treat both uterine arteries (first one side, then the other). The first vessel usually catheterized is the one opposite the puncture site. A Waltman loop is often used to enter the ipsilateral uterine artery,11 although there are interventionalists who use a Rosch inferior mesenteric (RIM) catheter for both sides, with the catheter pulled back from the left to the right to “hang” the catheter in the origin of the ipsilateral hypogastric artery, using it as a conduit to advance a microcatheter and wire into the right uterine artery. Another approach is use of the Roberts catheter (Cook Medical, Bloomington Ind.), which has a larger preformed loop similar to a Waltman loop. An alternative is to use a two-catheter approach, one from each common femoral artery. This has the advantage of allowing simultaneous embolization, reducing the fluoroscopy and procedure time substantially.12 It has the disadvantage of a second femoral puncture. Regardless of whether single or double puncture is used, once a catheter is in the hypogastric artery, a roadmap image is obtained to demonstrate the origin of the uterine artery so that it can easily be entered with the catheter or microcatheter. Once an adequate view of the origin has been obtained, an angiographic wire (or microwire) can be advanced into the uterine artery and a catheter advanced over it. A 4F or 5F catheter may be used, but if it causes spasm, a microcatheter should be used coaxially, and the outer catheter retracted into the hypogastric artery. If the vessel is not excessively tortuous, the catheter is advanced to the major medial turn of the vessel and placed in the proximal portion of the transverse segment of the uterine artery, preferably beyond the origins of any visible side branches. If the vessel is tortuous, the catheter may be advanced until it is just proximal to the first severely angulated segment to avoid spasm. Embolization should not proceed unless the catheter is well seated in the uterine artery because misembolization to other branches may occur, with potentially disastrous results. Prior to embolization, angiographic imaging is used to confirm catheter position and demonstrate the fibroid blood supply (Fig. e76-2, A Historically, the endpoint of embolization was complete occlusion of the uterine artery with coil or gelatin sponge plug adjuncts, but this has changed to subtotal occlusion in recent years. There are two endpoints of embolization in current use. The first is embolization until there is stasis or near stasis in the uterine artery, corresponding with complete occlusion of all its major branches. This endpoint is best achieved with polyvinyl alcohol (PVA) particles and other spherical embolics, with the exception of trisacryl gelatin microspheres (TAGM). For TAGM, less complete occlusion of the uterine arteries is effective and preferred, with the embolic injected slowly until the forward flow slows to sluggishness within the uterine artery. This corresponds to an angiographic image in which the fibroid feeding branches are distally occluded, but the uterine arteries and the proximal portions of its branches are patent, sometimes called the “pruned tree” appearance13 (Fig. e76-2, B The primary current controversy in UFE is the choice of embolic material. The efficacy of traditional PVA particles (Contour [Boston Scientific, Natick Mass.], Ivalon [Cook Medical], other manufacturers) has been well established in numerous clinical trials, both the early large case series14–17 and subsequent randomized trials.2,3 Similarly there are now considerable data to document the effectiveness of TAGM (Biosphere Medical, Rockland Mass.).18,19 A randomized comparative study compared TAGM to PVA particles and demonstrated comparable rates of symptom improvement and fibroid infarction.20 Despite an early presumption that the choice of embolic material was unlikely to impact outcome, this has not proven to be the case. In a randomized comparison of spherical PVA (sPVA, Contour SE, Boston Scientific, Watick, Mass.) and TAGM, there was a markedly poorer rate of fibroid infarction with the spherical PVA.13 These outcomes have subsequently been confirmed in two randomized comparisons of sPVA particles to other embolics, noting both poorer infarction rates21 and poorer long-term clinical outcomes.22 These findings taken together confirm a poorer outcome with spherical PVA and suggest they should not be used as an embolic for this procedure. At this time, there are no published data from randomized trials assessing two newer embolics, PVA hydrogel (BeadBlock Microspheres [Terumo Medical Corp., Somerset, N.J.]) or Polyzene F–coated hydrogel (Embozene Microspheres, CeloNova BioSciences, San Antonio, Tex.) microspheres for UFE, although at least one nonrandomized comparative study raises similar questions of effectiveness of PVA hydrogel particles.23
Uterine Fibroid Embolization
Indications
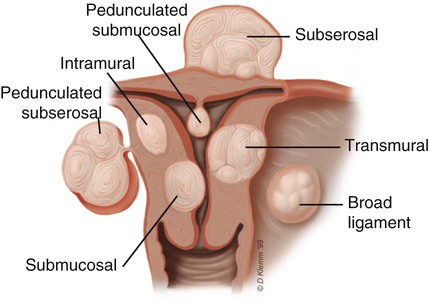
![]() ). Submucosal fibroids shrink more rapidly than intramural fibroids, and intramural fibroids shrink more rapidly than those on the serosa. There are some fibroid locations that are likely less favorable, and these are discussed further under Contraindications.
). Submucosal fibroids shrink more rapidly than intramural fibroids, and intramural fibroids shrink more rapidly than those on the serosa. There are some fibroid locations that are likely less favorable, and these are discussed further under Contraindications.
Contraindications
Equipment
Technique
Anatomy and Approach
Technical Aspects
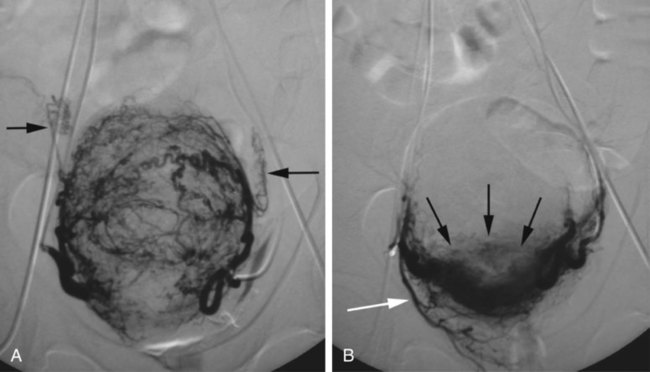
![]() ). As mentioned, there are several choices for embolic material, and these are discussed in greater detail under Controversies.
). As mentioned, there are several choices for embolic material, and these are discussed in greater detail under Controversies.
![]() ). Regardless of the embolic material used, it is important to wait a few minutes after the apparent conclusion to be sure there is no early recanalization, which may allow for reperfusion of the fibroids.
). Regardless of the embolic material used, it is important to wait a few minutes after the apparent conclusion to be sure there is no early recanalization, which may allow for reperfusion of the fibroids.
Controversies
Radiology Key
Fastest Radiology Insight Engine