Vascular Diseases
▪ ANATOMY
Arterial
The renal arteries branch from the aorta near the level of the L-1 to L-2 interspace. The right renal artery usually arises from the lateral or anterolateral aspect of the aorta and is slightly lower than the left renal artery, which arises from the lateral or posterolateral aspect of the aorta. Thus, aortography should be performed in the anteroposterior or slight right posterior oblique projections to demonstrate the origin of the renal arteries to best advantage.
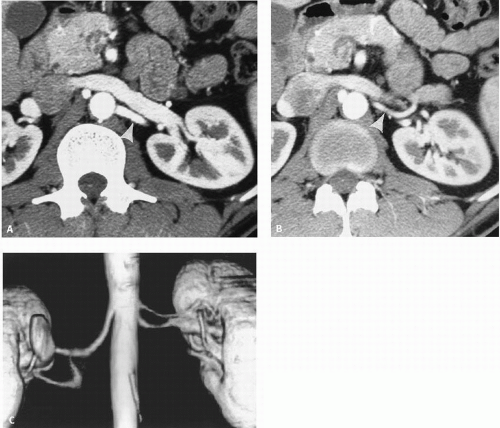
In as many as 40% of patients, one or both kidneys are supplied by more than one renal artery. Accessory renal arteries arise from the aorta and are usually inferior to the main renal artery. Occasionally, an accessory renal artery supplies the upper pole of the kidney; rarely, an accessory renal artery may arise from the celiac, hepatic, or mesenteric arteries. Anomalous kidneys, such as a horseshoe or pelvic kidney, almost always have multiple renal arteries that arise from the aorta or iliac arteries near the kidneys. Accessory renal arteries have traditionally been identified by aortography, but are now routinely seen on computed tomography angiography (CTA) (Fig. 10.1) or magnetic resonance angiography (MRA).
The inferior adrenal artery and arteries supplying the renal capsule, renal pelvis, and ureter arise from the main renal artery. Occasionally, the gonadal, middle adrenal, or inferior phrenic arteries may also arise from the renal artery.
The main renal artery divides into dorsal and ventral rami that run posterior and anterior to the renal pelvis. The larger ventral division supplies the anterior and superior aspects of the kidney, whereas the dorsal division supplies the posterior and inferior portions of the kidney. The junction of these ventral and dorsal divisions creates a relatively avascular plane (Brodel’s line), which is the preferred track for placing percutaneous nephrostomies.
Segmental branches arise from the dorsal and ventral rami and run along the infundibula before dividing into interlobar arteries. These interlobar arteries course between the pyramids and the cortical columns parallel to the outer surface of the kidneys before branching into arcuate arteries, which run along the bases of the medullary pyramids.
Collateral pathways that provide arterial supply to the kidney when the main renal artery is compromised include the inferior adrenal, capsular, ureteric, gonadal, intercostal, lumbar, and pelvic arteries. The upper three lumbar arteries allow blood from the aorta to communicate with pelvic, ureteral, or capsular arteries, which anastomose with renal branch arteries. When the ureteral artery serves as a major collateral, the dilation and tortuosity resulting from increased blood flow may cause the artery to impinge on the ureter, causing notching.
Venous
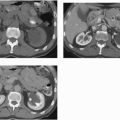
In general, the venous anatomy parallels the arterial circulation. Accessory renal veins are less frequent than accessory arteries and occur more commonly on the right. However, the left renal vein may bifurcate to encircle the aorta and become a circumaortic renal vein. This anomaly is caused by the persistence of the posteriorly located left supracardinal vein and by a midline supracardinal anastomosis between right and left vessels. It is a relatively common anomaly, reported in 2% to 16% of patients, according to anatomic and angiographic studies. Using computed tomography (CT), Reed et al. (1982) found a circumaortic left renal vein in 19 (4.4%) of 433 patients (Fig. 10.2). The posterior portion of this venous collar
typically runs inferiorly before crossing behind the aorta to reach the inferior vena cava (IVC). Knowledge of this anomaly is important when surgery is contemplated or during collection of renal or adrenal vein samples. The ventral vein usually drains the ventral and inferior portions of the kidney, whereas the dorsal component drains the dorsal and superior segments.
typically runs inferiorly before crossing behind the aorta to reach the inferior vena cava (IVC). Knowledge of this anomaly is important when surgery is contemplated or during collection of renal or adrenal vein samples. The ventral vein usually drains the ventral and inferior portions of the kidney, whereas the dorsal component drains the dorsal and superior segments.
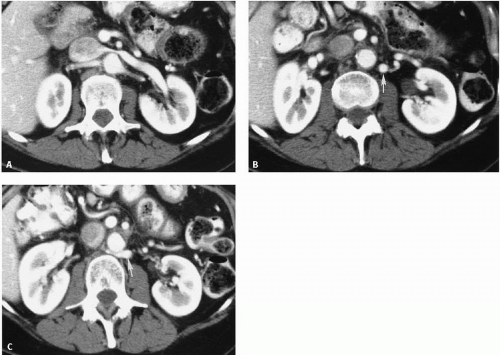
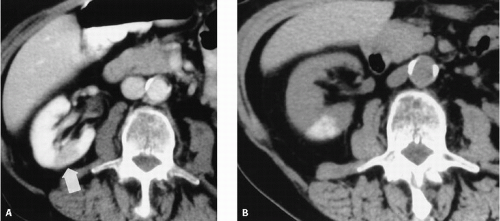
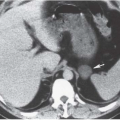
Another common anomaly, the retroaortic left renal vein, is seen less frequently. In the same series of CT examinations reported by Reed et al., 8 (1.8%) of 433 patients had a single retroaortic renal vein (Fig. 10.3). Both of these anomalies can be recognized by CT, and venography is seldom necessary for confirmation. The retroaortic component often extends caudally before passing behind the aorta. It is important not to confuse this with an enlarged lymph node on the axial CT image (Fig. 10.4). Patients are asymptomatic, but recognition is important if surgery involving this region is planned.
The left renal vein receives the inferior phrenic, capsular, ureteric, adrenal, and gonadal veins. In addition, rich collateral vessels usually anastomose with branches of the hemiazygos and ascending lumbar veins. These vessels are particularly important because they may preserve the kidney, should venous thrombosis occur. In patients with a left-sided IVC, the left common iliac vein continues cephalad as the left IVC and drains into the inferior aspect of the left renal vein.
The right renal vein is shorter than the left, and has a more oblique course to the IVC. It receives capsular and ureteric veins as well as some retroperitoneal collaterals, but the right inferior phrenic and gonadal veins enter directly into the IVC.
Valves may occur in the renal veins. They are infrequently seen on venography, and there is a marked variation in the reported incidence of renal vein valves in anatomic studies, ranging from 28% to 70% on the right and from 4% to 36% on the left. Their significance lies in surgical planning. Renal vein varices may be idiopathic or may result from renal vein thrombosis or portal hypertension. Like varicoceles, renal vein varices are more common on the left than on the right. Thus, an anatomic etiology is postulated. Compression of the left renal vein between the superior mesenteric artery and the aorta (“nutcracker” phenomenon) may result in left renal vein hypertension, hematuria, and varix formation. Abnormal branching of the superior mesenteric artery from the aorta has been demonstrated by magnetic resonance (MR) in some patients.
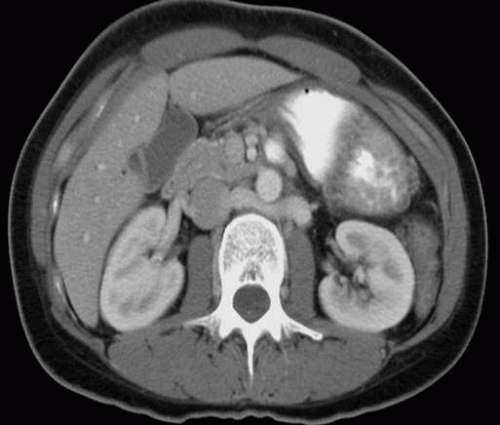
Obstruction of the left renal vein by the superior mesenteric artery is more likely to occur in thin patients in whom there is a paucity of retroperitoneal fat. Because a distended left renal vein may be seen by CT (Fig. 10.5) or ultrasound in 51% to 72% of the normal population, measurement of a pressure gradient between the IVC and the left renal vein is needed before renal vein compression should be diagnosed. The demonstration of collateral veins
using color-flow Doppler provides noninvasive evidence of renal vein compression.
using color-flow Doppler provides noninvasive evidence of renal vein compression.
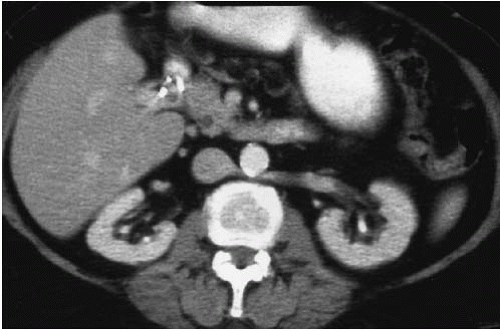 FIGURE 10.3. Retroaortic left renal vein. The left renal vein passes behind the aorta to enter the IVC. |
Collateral pathways exist for venous drainage of blood from the kidney in case of occlusion of the main renal vein. Inferior phrenic or adrenal, gonadal, and ureteric veins commonly enter the left renal vein, whereas only the ureteric vein enters the right renal vein. Any of these vessels, as well as a variety of small retroperitoneal veins that enter the renal veins, may function as collateral vessels. The right adrenal, inferior phrenic, and gonadal veins enter directly into the IVC.
In renal vein thrombosis, the clot usually propagates along the entire renal vein and these collateral vessels are also occluded. A local occlusion, such as surgical ligation, however, may allow these collaterals to take over drainage of the kidney. This occurs much more readily with occlusion of the left renal vein, as it receives more potential collateral vessels than the right renal vein.
Circumcaval (Retrocaval) Ureter
Persistence of the right subcardinal vein traps the ureter behind the IVC. The ureter crosses posterior to the IVC and then passes around the medial border anteriorly to partially encircle the cava. This anomaly, which occurs in approximately 1 in 1,100 patients, has been termed a retrocaval, or circumcaval, ureter. The term circumcaval ureter is preferred because it is possible for the ureter to lie behind the vena cava without encircling it. A circumcaval ureter is usually an incidental finding and is more common in men than in women. Most patients are asymptomatic, although right flank pain may be sufficient to bring this anomaly to attention.
The most common complication is obstruction, caused by constriction of the retrocaval segment of the ureter by the IVC. In a few cases, fibrous bands or adhesions of this segment of the ureter have been reported. The hydronephrosis and stasis predispose to stone formation and infection.
Two radiographic patterns of circumcaval ureter have been recognized. The more common form, type I, has an S-shaped deformity of the midureter as it courses around the IVC. The narrowing of the ureter occurs at the lateral border of the psoas muscle, suggesting that the obstruction is not caused by compression by the IVC. The second type (type II) has less severe hydronephrosis, but the point of obstruction is at the lateral wall of the IVC. However, ureteral obstruction is not necessarily present in a circumcaval ureter.
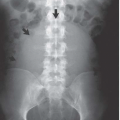
When the classic (type I) appearance is seen in urography, the diagnosis of circumcaval ureter can be suggested (Fig. 10.6). However, an inferior vena cavogram or CT (Fig. 10.7) is needed for confirmation. In the less severe form (type II), medial deviation must be distinguished from other etiologies, such as retroperitoneal fibrosis or a retroperitoneal mass.
Circumcaval ureter can be recognized on CT by following the opacified ureter around the IVC. CT also demonstrates the more lateral location of the cava, which usually lies lateral to the right pedicle of the third lumbar vertebral body.
Lymphatic
There is an extensive lymphatic system within the kidney that provides an accessory drainage route for excess fluid. In normal states, approximately one-fourth of lymphatic flow from the kidney occurs through small lymphatic vessels that permeate the capsule and communicate with lymph vessels in the perinephric space. The remainder of the renal lymph fluid is drained into large lymph vessels in the renal hilum. The renal lymphatics are not directly imaged, but enlarged vessels may be detected by CT.
▪ DISEASES OF INTRARENAL ARTERIES
A variety of entities affect intrarenal arteries. Although the etiology and clinical course may be different, the radiographic manifestations are similar. Because the disease process is usually generalized, both kidneys are affected and are reduced in size. Small infarcts result in a slightly irregular contour. Renal function may be markedly impaired, but if imaged by urography or retrograde pyelography, the renal collecting system will be normal. The most characteristic findings of diseases affecting intrarenal arteries are the multiple microaneurysms seen with renal arteriography.
Collagen Vascular Diseases
The vasculitides affect the glomeruli and other renal vessels. They may be classified as primarily involving medium and large renal arteries or small vessels and capillaries. Although any of the systemic vasculitides may involve the kidneys, this discussion will concentrate on those that do so commonly.
Polyarteritis Nodosa
Polyarteritis nodosa (PAN) is a panarteritis with foci of fibrinoid necrosis that begin in the media. The subsequent inflammation that spreads to involve the intima and adventitia results in small aneurysms and vascular thrombosis. Although the etiology of PAN is unknown, it is associated with rheumatoid arthritis, Sjögren syndrome, hepatitis B, cryoglobulinemia, and hairy cell leukemia. Renal involvement occurs in 90% of patients with PAN. Patients often present with hematuria, and renin-mediated hypertension is common. The small aneurysms seen in PAN may occasionally rupture and produce an intraparenchymal or perinephric hematoma.
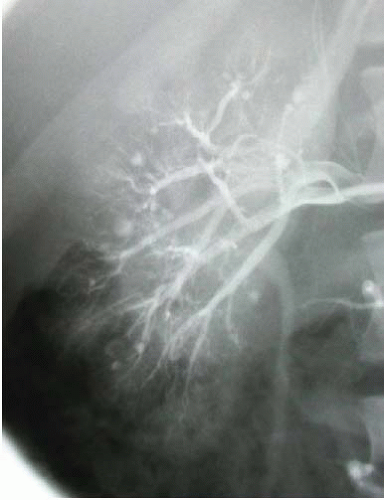
With the exception of arteriography, radiographic imaging studies demonstrate nonspecific findings. Parenchymal scarring may be seen with urography, ultrasound, or CT. Areas of hemorrhage due to aneurysm rupture are best defined by CT, but, if large, can also be detected by ultrasonography. The most definitive radiographic examination is arteriography, where small aneurysms occur at the bifurcation of interlobular or arcuate arteries (Fig. 10.8). These aneurysms are not limited to the kidneys, and arteries in the liver, spleen, pancreas, muscle, and gastrointestinal tract are often involved.
Although these small aneurysms are typical of PAN, they are not pathognomonic. Similar aneurysms may be seen in patients with systemic lupus erythematosus, Wegener granulomatosis, and intravenous drug abuse.
Wegener Granulomatosis
Patients with Wegener granulomatosis have necrotizing granulomas of the respiratory tract, a focal necrotizing angiitis involving small arteries and veins, and a focal necrotizing glomerulitis that leads to fibrin thrombi and local necrosis of individual glomerular tufts. It occurs most commonly in the fourth and fifth decades and has a slight male preponderance. Symptoms of upper airway involvement dominate the clinical presentation. Although nodular, infiltrative, or cavitary lung lesions are commonly seen, the pulmonary involvement is generally asymptomatic.
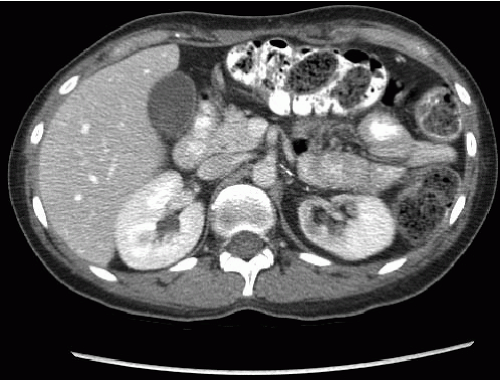
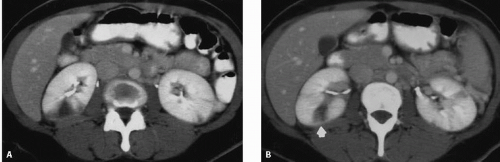
Renal disease may be absent in the limited form of Wegener granulomatosis, but a rapidly progressive glomerulonephritis is life threatening in the full syndrome. The most common manifestations of renal disease are hematuria and proteinuria. The radiographic manifestations in the kidneys are nonspecific, but include microaneurysms, renal infarction (Fig. 10.9), parenchymal scarring, and areas of hemorrhage. The findings are indistinguishable from those of PAN. Rarely, a renal mass may be seen as a manifestation of the disease.
Churg-Strauss vasculitis is similar to Wegener granulomatosis, but rarely involves the kidneys. Kawasaki disease is an acute febrile illness affecting infants and small children. This vasculitis involves small- and medium-sized arteries with a predilection for the coronary arteries.
Systemic Lupus Erythematosus
Most patients with systemic lupus erythematosus have renal involvement, and many die of renal failure. The larger renal vessels are usually unaffected, but interlobular arteries may be affected by inflammatory changes and may be narrowed. The predominant renal changes consist of a focal glomerulonephritis with thickening of the basement membrane resulting in the “wire loop” appearance on histologic preparations.
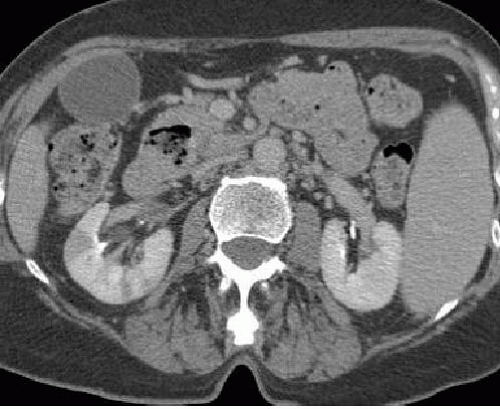 FIGURE 10.9. Wegener granulomatosis. A wedge-shaped area of infarction is seen in the lateral aspect of the right kidney. |
The radiographic appearance depends on the stage of involvement. Before the onset of renal failure, the kidneys appear normal. Microaneurysms, similar to those seen in PAN, may occasionally be seen on angiography. Although renal infarcts are common, they are usually too small to be seen without selective-magnification arteriography. Conventional ultrasound examination is often normal, but an elevated resistive index may be predictive of worsening renal function.
Intravenous Drug Abuse
The vasculitis associated with intravenous drug abuse has clinical and pathologic features similar to PAN. Although methamphetamine is a common drug used by patients in whom a vasculitis develops, they are usually exposed to multiple drugs. Circulating hepatitis antigen-antibody complexes or drug contaminants may also be responsible for vascular injury. The radiographic appearance of multiple aneurysms 1 to 5 mm in diameter occurring at bifurcations, vascular stenoses, and complete vascular occlusion with infarction is indistinguishable from other vasculitides.
Scleroderma
Progressive systemic sclerosis is a generalized disorder manifested by vascular and connective tissue fibrosis. Narrowing of the interlobular arteries due to intimal thickening may be present, as may fibrinoid necrosis of the afferent arterioles. It occurs most commonly in the fourth and fifth decades, and shows a significant female preponderance. The kidneys may be affected in up to 80% of patients, and renal failure is often the cause of death.
The radiographic manifestations are nonspecific. Before the onset of renal failure, the kidneys may appear normal. Hypertension is common, such that the vascular changes seen on angiography may be due to either nephrosclerosis or scleroderma. However, the microaneurysms seen with the vasculitides are not seen with scleroderma.
Radiation Nephritis
Radiation nephritis is a degenerative process that affects the tubules and glomeruli. It may be acute or chronic, cause hypertension, or result merely in proteinuria. The vascular changes of fibrinoid necrosis occur late in the course and involve primarily arcuate and interlobar arteries. Doses of at least 20 Gy (2,000 rads) within a 5-week period are needed to cause significant renal damage.
Acute radiation nephritis is manifested by proteinuria and hypertension after a latent period of 6 to 12 months. Uremia, malignant hypertension, and congestive heart failure follow. The prognosis of these patients is poor, with a mortality of approximately 50%.
Patients who develop chronic radiation nephritis may or may not have been affected by acute radiation nephritis. Chronic radiation nephritis has an insidious onset of mild proteinuria, anemia, and azotemia, beginning 18 months to several years after radiation exposure. Although the clinical course is more protracted, the mortality of 50% is similar to acute radiation nephritis.
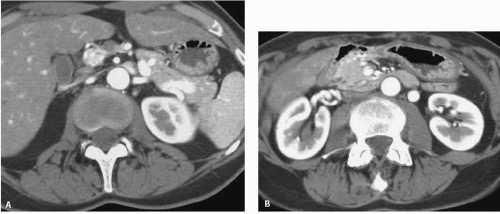
The radiographic appearance of radiation nephritis depends on the radiation dose and the time between the radiation exposure and the imaging study. In the acute phase, diminished renal function may be manifested only by decreased concentration of excreted contrast medium. In the chronic phase, an area of diminished enhancement is seen (Fig. 10.10).
Arteriolar Nephrosclerosis
Systemic hypertension affects the vascular tree of the kidney more consistently and more extensively than any other region of the body. The degree of change is largely a function of the severity and duration of the hypertension.
The clinical symptoms of patients with benign nephrosclerosis are usually limited to those of hypertension. In malignant nephrosclerosis, patients have a diastolic blood pressure >130 mm Hg and have papilledema. Neurologic symptoms and renal failure are common. Elevated plasma renin and aldosterone along with hypokalemia are manifestations of secondary aldosteronism, and proteinuria is common. Untreated malignant hypertension has a poor prognosis, and renal failure is the most common cause of death.
The radiographic findings reflect the degree of involvement of the kidneys. Unless there is a renal artery stenosis (RAS) to protect one of the kidneys, involvement is systemic. The kidneys are normal to small. The calyces remain normal, even in areas of marked cortical thinning. Increased tortuosity and more rapid tapering of intrarenal arteries are seen with angiography. More severe changes include filling defects and loss of cortical vessels.
▪ EMBOLISM AND INFARCTION
The most common source of renal artery emboli is a diseased heart. Patients with atrial enlargement secondary to valvular heart disease or a dyskinetic left ventricle after myocardial infarction provide the source of mural thrombi that may dislodge and become renal artery emboli. Smaller emboli may arise from an ectatic or aneurysmal aorta, or even from cholesterol plaques in a patient with severe atherosclerosis.
Unlike arterial stenoses that are slowly progressive and may cause atrophy or collateralization, emboli produce acute ischemia that may result in infarction. The typical clinical features include the sudden onset of flank pain, hematuria, proteinuria, fever, and leukocytosis. However, the presentation is variable, and the diagnosis often is missed. Despite documented unilateral involvement, a decrease in renal function is seen in many patients.
The radiographic appearance of renal embolism depends on the size of the embolus and location of the arterial occlusion. Excretory urography demonstrates an absence of enhancement of the affected segment of the kidney, reflecting lack of vascular perfusion. If the main renal artery is occluded, there is no renal function in the affected kidney. A swollen, edematous kidney is seen on ultrasound as an enlarged kidney with decreased echogenicity. Retrograde pyelography reveals a normal collecting system with sharply cupped calyces. If there is much swelling, there may be attenuation of the intrarenal collecting system.
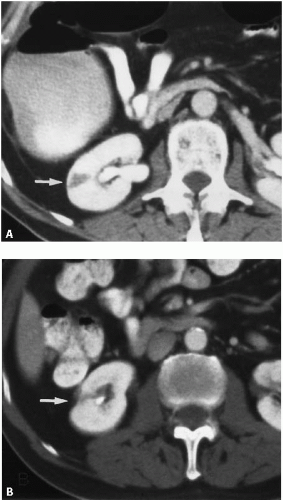
The absence of contrast enhancement in the affected renal tissue is best demonstrated by CT. Smaller infarcts are seen as wedge-shaped, low-density areas within an otherwise normal-appearing kidney (Fig. 10.9). If the entire kidney is affected, the increase in size due to edema can be identified by the large size and more rounded configuration. Even if the entire renal artery is occluded, capsular branches remain patent and enhance the outer rim of the kidney. The preservation of this outer 2 to 4 mm of the cortex is best seen on CT (Fig. 10.11).
Occasionally, high-grade stenosis of an intrarenal artery will diminish opacification, but will not cause frank infarction with scarring. Images obtained after contrast has washed out of the normal renal parenchyma may demonstrate persistent enhancement of this hypoperfused area (Fig. 10.12).
RENAL INFARCT
Low density
Wedge-shaped
Preserved cortical margin
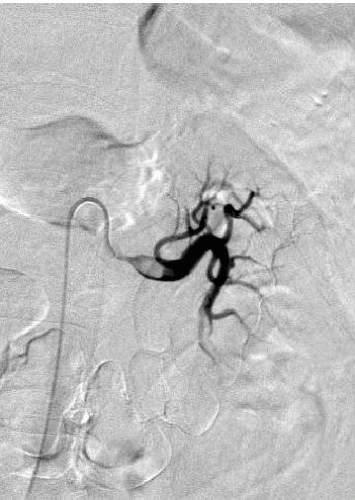
Arteriography is needed for a definitive diagnosis. Sharp vessel cutoff will be seen if the embolus completely occludes arterial flow. Incompletely occluding emboli appear as a filling defect within a contrast-filled artery (Fig. 10.13).
After the acute phase of renal infarction, atrophy begins. The infarcted tissue contracts, leaving a cortical scar (Fig. 10.14). The parenchymal loss reflects the distribution of the affected artery. If the main renal artery is occluded, the entire kidney will be affected. The kidney atrophies uniformly. There is no appreciable renal function, but renin may be elaborated and may cause hypertension.
Treatment of renal artery embolism depends on the patient’s underlying medical condition and the status of the contralateral kidney. Attempts at revascularization can be made with lytic therapy delivered directly into the renal artery through an arterial catheter. Although this is not rewarding as often as lysis of clot that forms behind an arterial stenosis, excellent results have been reported. Many patients are treated with anticoagulant therapy, although surgical revascularization may be attempted in selected cases.
Arterial Thrombosis
Thrombosis of the renal artery occurs most commonly as a complication of severe atherosclerosis. In such cases, atherosclerosis usually involves a variety of other arteries, including coronary and carotid arteries, that dominate the clinical picture. This gradual occlusion of the renal artery that finally results in thrombosis is usually clinically silent and results in ipsilateral renal atrophy.
Acute thrombosis of the renal artery may occur after trauma. It usually follows blunt abdominal trauma in which the forces of acceleration or deceleration produce intimal tears, with resulting dissection of the renal artery and thrombosis. Renal artery thrombosis may also result from subintimal dissection of the renal artery during arteriography. This is more likely to occur during an attempted transluminal angioplasty than during a diagnostic renal arteriogram.
With acute renal artery thrombosis, the kidney remains normal in size. Unless extensive renal artery collaterals have developed, there is no renal function, and intravascular contrast medium will not be excreted. Retrograde pyelography demonstrates a normal collecting system.
Color-flow Doppler ultrasound may also be used in the diagnosis of renal artery thrombosis. The most common finding is absence of an intrarenal arterial signal. If there is incomplete occlusion or collateral vessels are present, a severe tardus-parvus abnormality is detected. In some patients, ultrasound may demonstrate a proximal renal artery stump.
CT reveals lack of enhancement, although a thin peripheral rim often remains viable because of collateral circulation through capsular arteries. Because collateral blood flow may also come from ureteric, gonadal, lumbar, or adrenal arteries, additional portions of the kidney, such as the medulla, may also be preserved. Arteriography may be used to confirm the diagnosis of an occluded main renal artery.
With gradual occlusion, the kidney usually diminishes in size over time and a small kidney remains (Fig. 10.14). If collateral vessels are present, there may be a small amount of renal function preserved. The renal contour is smooth, unless small infarcts have already occurred, and the calyces remain normal.
▪ ANEURYSM
Atherosclerotic Aneurysm
Aneurysms of the renal arteries are uncommon. The most common etiology is atherosclerosis, but a dissecting aneurysm may also involve the renal artery. Mycotic aneurysms usually involve the aorta, but may occasionally affect the renal artery.
Most patients are asymptomatic, and the aneurysm is often discovered incidentally on imaging studies. Because hypertensive patients often undergo angiography to identify a renovascular etiology, it is not surprising that many patients found to have a renal artery aneurysm are hypertensive. In some patients, surgical resection of the aneurysm results in cure of the hypertension; however, these patients usually have an associated RAS and lateralizing renal vein renin levels.
Renal artery aneurysms often contain clot and may give rise to renal emboli with or without infarction. The risk of rupture is small, but is more likely in hypertensive or pregnant patients. Calcified aneurysms rarely rupture.
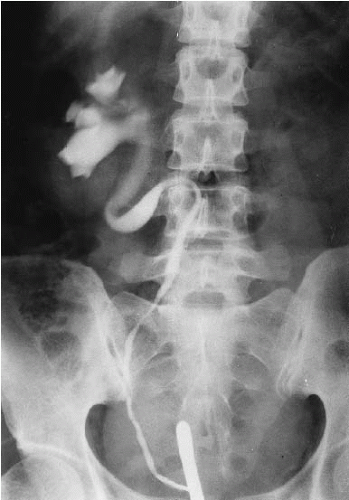
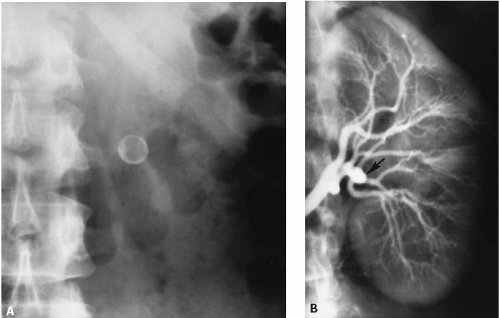
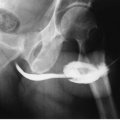
If calcified, a renal artery aneurysm can be recognized on the plain abdominal radiograph (Fig. 10.15). However, the appearance of a curvilinear calcification could be caused by a tortuous or wandering splenic artery, or even a nonvascular etiology.
On ultrasound, an aneurysm is seen as a hypoechoic mass along the course of the renal artery. The Doppler signal arising from the aneurysm depends on the amount of thrombus and the size of the neck of the aneurysm.
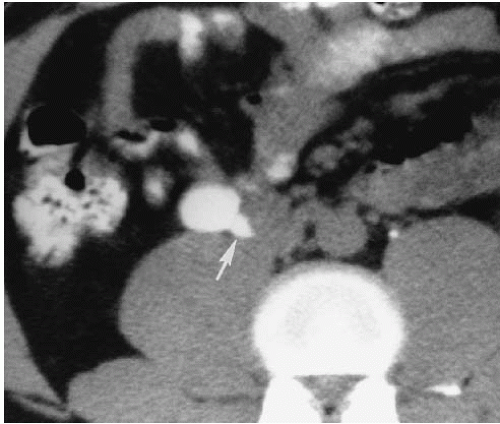
Renal artery aneurysms may also be demonstrated by CT. Calcification along the wall of the aneurysm is readily detected on unenhanced images (Fig. 10.16). After contrast administration, variable enhancement is found, depending on the amount of thrombus within the aneurysm.
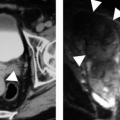
Renal artery aneurysms may be demonstrated on MR using a variety of techniques. Gadolinium-enhanced T1-weighted images using a phased array coil system are commonly employed (Fig. 10.17).
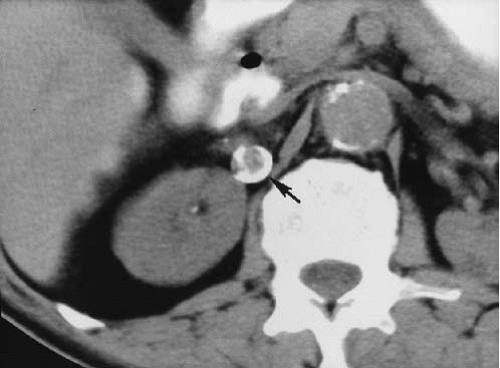 FIGURE 10.16. Renal artery aneurysm. A densely calcified right renal artery aneurysm (arrow) is clearly visible on this unenhanced CT examination.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|