14 The Aortic Valve
Introduction
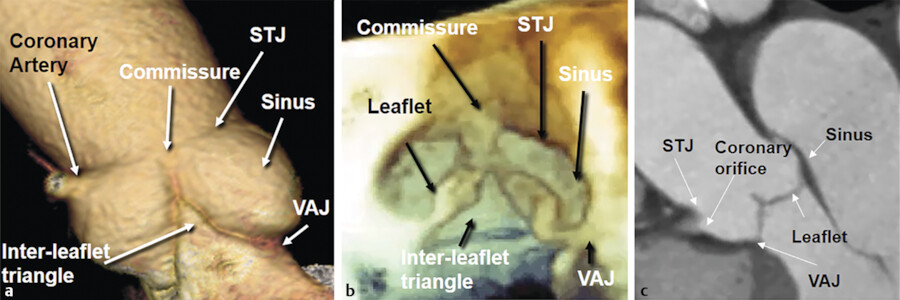
The aortic valve is a complex structure, which consists of different highly specialized components that interact with each other, modifying their shape and size during the cardiac cycle. Following the direction of the flow, we encounter the ventriculoarterial junction, leaflets, sinuses, the crown-shaped annulus, the interleaflet triangles, the commissures, the coronary ostia, and sinotubular junction. 1 , 2 Although in anatomical books this complex apparatus takes often the name of aortic valve, we prefer to use the more inclusive name of the “aortic root.” The job of the aortic root is definitely one of the most difficult. It (a) allows forward passage of 70 to 100 mL of blood with each systole in a low ventricle–aorta gradient, (b) prevents diastolic reflux, (c) assures wide flow variations (up to five times), and (d) maintains an optimal coronary perfusion.
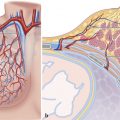
The most important component of the aortic root is the aortic leaflets. The aortic leaflets open and close about 3 billion of times in the course of an average lifetime. Despite their delicate and flexible tissue structure, the leaflets have a strong structural skeleton that withstands the diastolic aortic pressure. This constant workload would certainly cause premature degeneration of the leaflets, if the protective effect of the “aortic root” did not exist. The aortic root helps to preserve leaflets by distributing the stress between the various components. Because of its conical–cylindrical shape and its pivotal role in cardiac function role, the aortic root could be considered the “fifth chamber” of the heart (Fig. 14‑1). This chapter describes the anatomy of various component of the aortic root.
Anatomy
Ventriculoarterial Junction
The ventriculoarterial junction is a circumferential line which demarcates the site of connection of the left ventricle to the nadir of the aortic sinuses. The ventriculoarterial junction should not be confused with the aortic annulus. During aortic valve replacement, surgeons suture the basal ring of the prosthetic valve to the contour of the base of the aortic root describing this boundary as the “annulus.” No matter of what terminology used, it should be noted the ventriculoarterial junction as an anatomically rigid ring-like fibrous structure does not exist, otherwise it would create a gradient similar to that produced by prosthetic valves. On the contrary, the ventriculoarterial junction is able to expand and twist during isovolumic contraction and early phase of ejection to minimize pressure gradient across the valve. Therefore, the ventriculoarterial junction can be more precisely defined as the “boundary” between the fibroelastic wall of the aorta and the partly muscular and partly fibrous rim of the left ventricular outflow tract. The ventriculoarterial junction is not a rigid structure and similar to the other parts of the aortic root and the leaflets, it has a specific kinematics during the cardiac cycle. During the ejection phase, the ventriculoarterial junction contracts, while the sinotubular junction expands; together, they form a truncated cone with the smaller surface toward the left ventricle. This transient morphology facilitates the progression of the column of blood toward the ascending aorta.
Aortic Leaflets
The aortic leaflets represent the “core” of the aortic root. Describing them as a simple flap of thin fibrous tissue is incorrect. On the contrary, they are “live” structures with an architecture formed by three inner layers (fibrosa, spongiosa, and ventricularis) that are covered on both sides by the endothelial cells. The aortic leaflets contain cells that secrete proteins that form an extracellular matrix, nerve endings, blood vessels, and possibly some contractile activity (documented only by anecdotal observations). In other words, aortic leaflets are metabolically “alive” with the potential to self-regenerate. One of the secret of their longevity is probably the ability to repair.
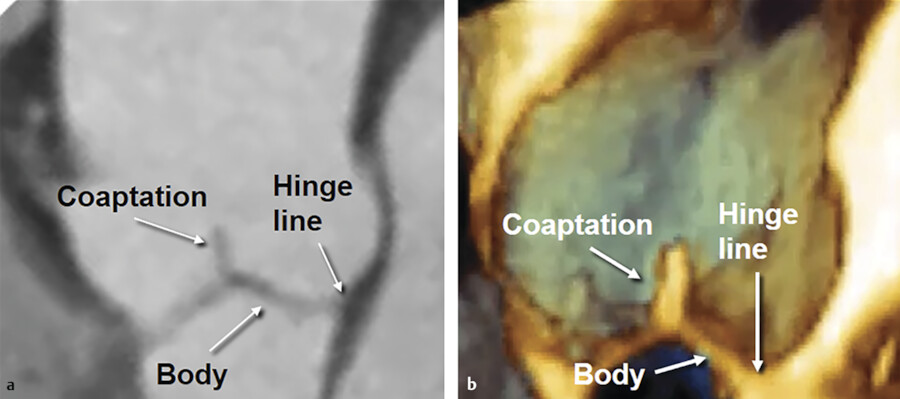
The shape of aortic leaflets resembles a “bird’s nest.” In each leaflet, one can distinguish a hinge line, which is the line of insertion of the leaflet on the wall, a body, and a free edge of coaptation. At the center of the leaflet free margin a fibrous nodule exist which is called the nodule of “Arantius.” When the valve closes, the leaflets approach one another along the surface of coaptation, which takes the name of “lunula” (Fig. 14‑2).
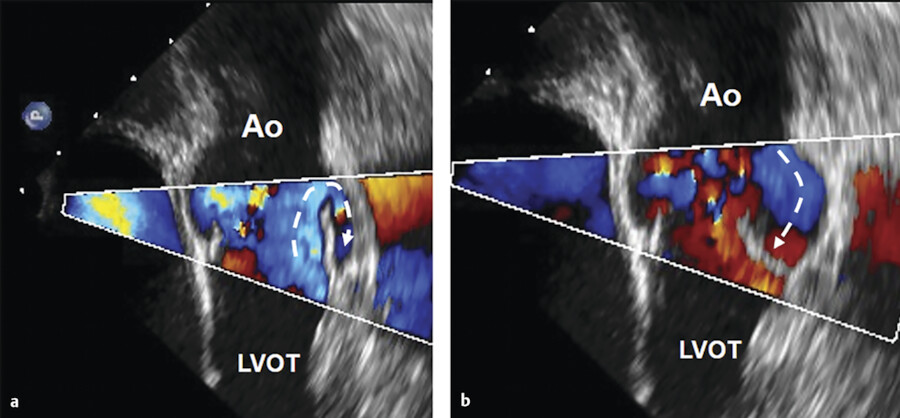
The impact that the leaflets have in the act of closure certainly would cause severe wear and tear. The reason that this does not occur prematurely is because when the pressure in the aorta exceeds that of the ventricle, leaflets are almost closed. Vortices formed within the sinuses of Valsalva promote the closure during systole when the flow starts to decelerate. In other words, the closure of the leaflets begins during ejection (Fig. 14‑3).
Aortic Annulus
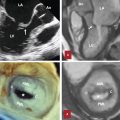
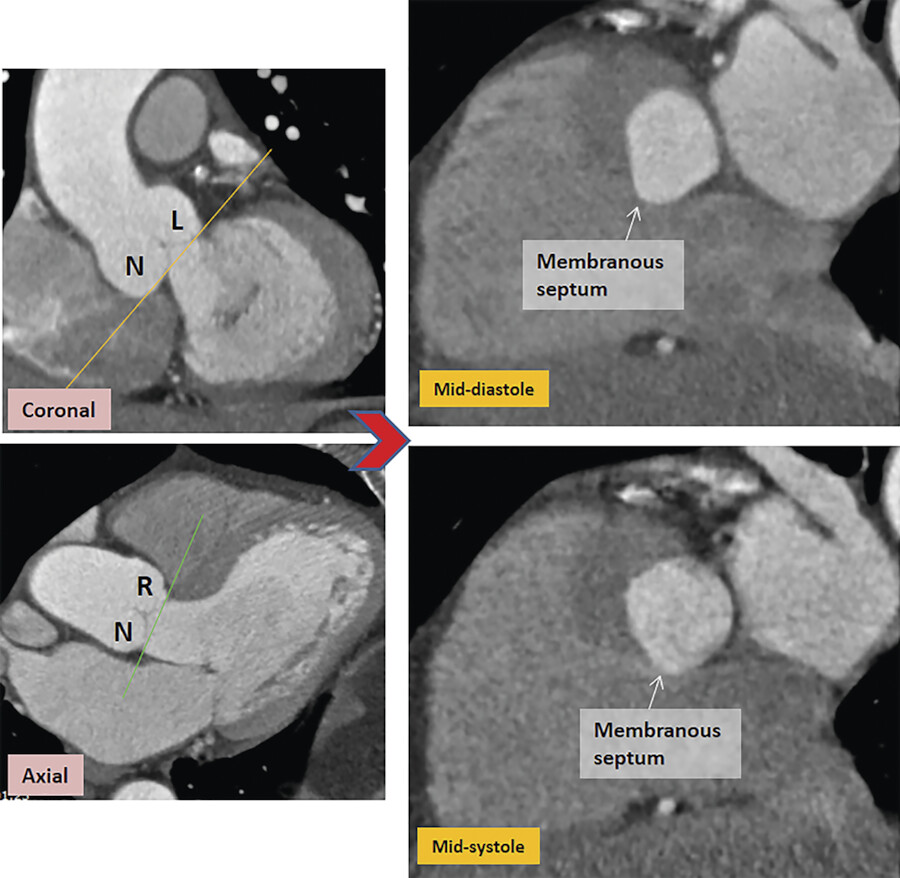
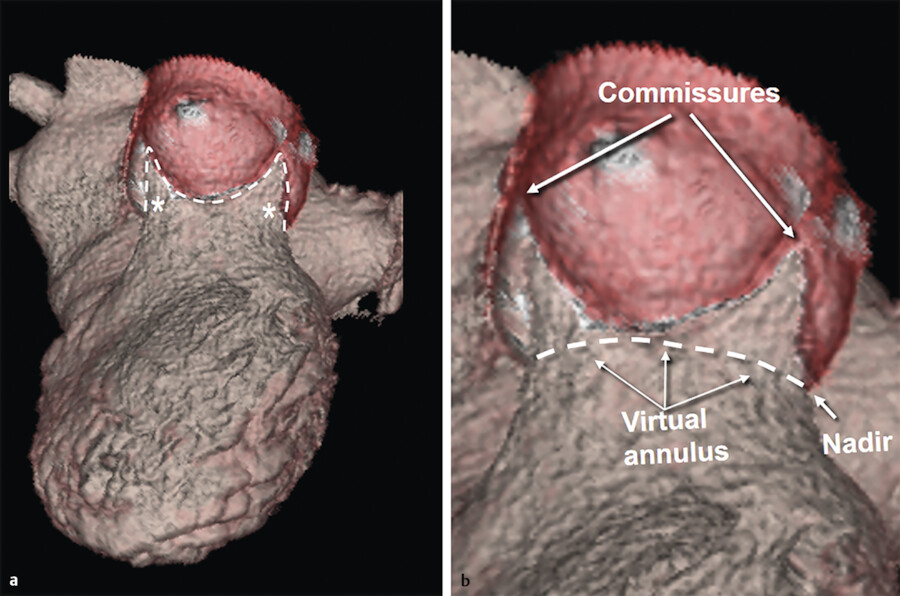
The term “annulus” of a valve would simply apply to a circular structure formed by a thick fibrous ring that sustains leaflets. 3 This is not the case for the aortic annulus. The “true” aortic annulus (i.e., fibrous structure that permits tight attachment of the leaflet to the aortic wall) has a “trident” or “crown-shaped” configuration. The lowest part of insertion (nadir) corresponds to ventriculoarterial junction and the highest part (commissure) to the sinotubular junction. At this level, the insertion of one cusp meets and continues with that of the adjacent cusps. As mentioned above, many use the term “annulus” to denote the location of the ventriculoarterial junction. For example, in the transcutaneous aortic valve implantation, the virtual annulus measurement is obtained by connecting the nadirs of the three leaflets at or slightly below the ventriculoarterial junction for selecting the right prosthesis size. In normal subjects, the virtual annulus is elliptical in diastole and circular in systole (Fig. 14‑4).
Interleaflet Triangles
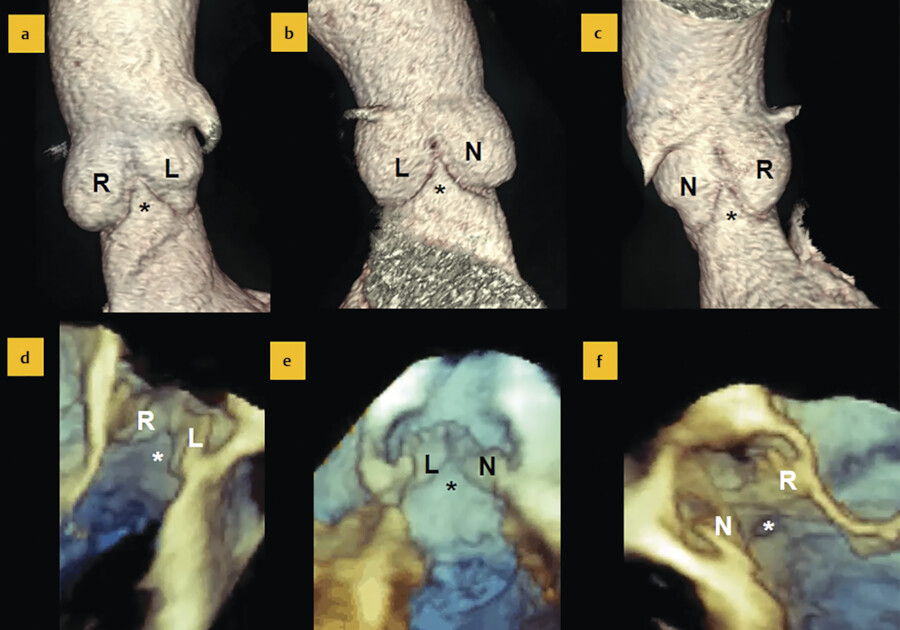
Given the particular crown-shaped configuration of the aortic annulus, three triangles of fibrous and muscular tissue, named interleaflet triangles (ILTs), arise from the left ventricular outflow tract between the sinuses. 4 The ILT between the right and left coronary sinuses may contain ventricular muscular tissue. The ILT between the left coronary and the noncoronary sinuses is a fibrous sheet that continues with the anterior leaflet of mitral valve (the mitral–aortic curtain). Finally, the ILTs between the noncoronary and right coronary sinuses comprise of fibrous tissue which is continuous with the membranous septum. Interestingly, the ILTs, which are unmistakably considered part of the aortic root, are, in fact, under the ventricular pressure (Fig. 14‑5 , Fig. 14‑6).
Sinuses of Valsalva
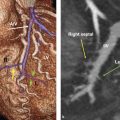
The sinuses of Valsalva are three protrusions at the aortic root. Similar to the valve leaflets, the aortic sinuses are named right, left, and noncoronary. The first two include the origin of the coronary arteries. The wall of the sinuses is mainly composed of an elastic lamina and smooth muscle cells and oftentimes it would not be easy to distinguish between the tunica media and tunica intima. Collagen and elastic fibers form the adventitia. In diastole, the aortic root assumes, when seen in cross-section at level of the midpoint of Valsalva’s sinuses, a trilobate conformation. In this way, each sinus becomes more spherical and with the correspondent leaflet forms a single, roughly hemispherical, functional unit. 5 Anatomists call this functional unit cusp. This trilobate conformation allows homogeneous distribution of the aortic pressure between the leaflets and the sinuses (Fig. 14‑7).
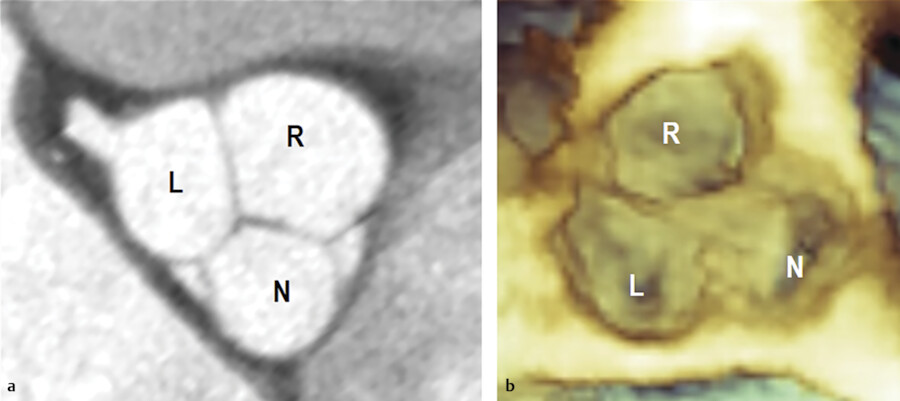
Sinotubular Junction
The sinotubular junction is the only fibrous ring of the aortic root and marks the border between the aortic root and the ascending aorta. This ring supports the cranial insertion of the leaflets: the commissures. The diameter of the sinotubular junction is 10 to 15% smaller than the diameter of the ventriculoarterial junction. However, during ventricular ejection the sinotubular junction dilates giving the aortic root the conformation of a truncated cone with the smaller circumference facing the ventricle.
Aortic Diseases
In this chapter, we will briefly discuss the most common pathologies affecting the aortic leaflets such as papillary fibroelastomas, bicuspid valve, aortic stenosis, aortic insufficiency and aortic endocarditis, and the most frequent disease affecting the aortic root and the aorta namely aortic aneurysm, and aortic dissection. We start with an aspect of the aortic leaflets that is between anatomical variant and disease: Lambl’s excrescences.
Lambl’s Excrescences
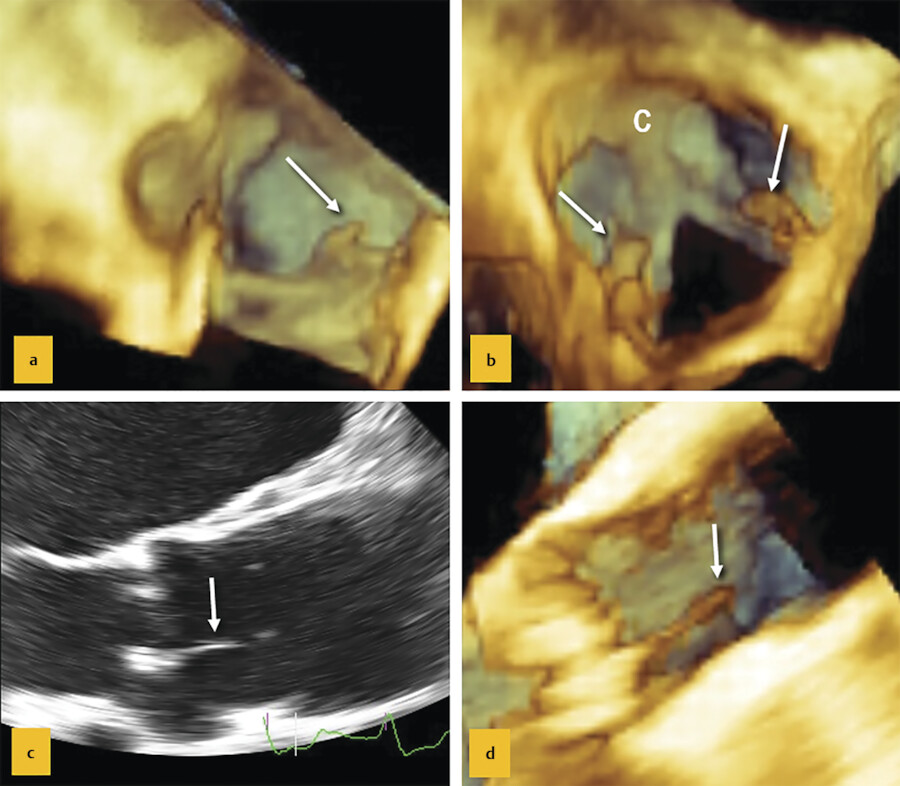
Lambl’s excrescences are filiform fibrous structures attached along the coaptation surface of the aortic leaflets, often at level of the nodules of Arantius, where the wear and tear may cause endothelial exfoliations and promote fibrin and small thrombi formation, which eventually heal in collagen filiform organization. 6 Usually asymptomatic and present in older individuals, Lambl’s excrescences are considered a benign age-related process. Lambl’s excrescences may occur in normal or calcified leaflets (Fig. 14‑8 c, d). However, occasionally, these excrescences may detach and reside in remote vessels (Fig. 14‑8 a, b).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree