24 Liver Transplant Complications: Diagnosis and Management
Mikin V. Patel and Brian Funaki
24.1 Introduction
Orthotopic liver transplantation (LT) is the treatment of choice for end-stage hepatic disease. Over the years, improvements in surgical techniques, organ preservation therapies, and detection of postoperative complications have improved post-LT care such that 1-, 5-, and 10-year patient survival rates have increased to 88, 80, and 74%, respectively. 1 , 2 However, vascular and biliary complications continue to persist at an estimated rate of 25 to 27%. 3 Interventional radiology is an essential component of a successful LT program because it provides minimally invasive solutions to potentially lethal complications, typically precluding the need for major surgery or retransplantation. 3 , 4 , 5 , 6 , 7
24.2 Vascular Complications
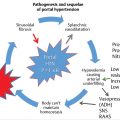
In LT patients with hepatic failure, bile leak, gastrointestinal bleeding, or sepsis, vascular complications are a primary diagnostic consideration, and prompt treatment is needed to salvage the graft in such cases. 8 , 9 , 10 Vascular complications occur in approximately 8 to 15% of LT patients; the incidence of arterial complications is approximately 5 to 10%; portal venous complications occur in 3 to 5% and outflow venous complications in < 2%. 11 Clinical and biochemical markers are generally nonspecific, so diagnosis requires imaging, usually with Doppler ultrasound (US), contrast-enhanced computed tomography (CT), or magnetic resonance (MR) imaging. 3 , 8
24.2.1 Arterial Complications
Arterial complications include hepatic artery thrombosis (HAT), hepatic artery stenosis (HAS), and hepatic artery pseudoaneurysm (HAP), with incidences of 0.8 to 9.3%, 1.9 to 16.6%, and 0 to 3%, respectively. 11 , 12
Hepatic Artery Thrombosis
Despite the use of modern surgical techniques for LT, patients undergoing this procedure are at risk of developing thrombosis at the arterial anastomosis. Multiple etiologies have been proposed for HAT, including differences in size of donor and recipient vessels, prolonged cold ischemia time, previous transcatheter arterial chemoembolization, variant arterial anatomy, and positive donor cytomegalovirus serology. 13 , 14 HAT can readily be diagnosed with Doppler US; on US, HAT will present as absent flow in the proper hepatic and intrahepatic arteries. CT and MR can also be used to perform contrast angiography, which will demonstrate abrupt cutoff of the hepatic artery, usually at the site of anastomosis.
Clinically, HAT is generally divided into early (< 30 days after LT) and late (> 30 days after LT) thrombosis, although this definition may vary. Early HAT invariably leads to devastating complications such as liver parenchymal necrosis, biliary necrosis, and sepsis if not treated. These patients face a mortality rate of 33.3% and are usually urgently listed for retransplantation. 14 , 15 Given the limited donor pool, however, revascularization is frequently attempted, with endovascular techniques emerging as the less invasive treatments of choice. 12 , 16 , 17 , 18 , 19 Prompt revascularization within 7 or 14 days of HAT yields graft survival rates of 81 and 62%, respectively, but graft survival drops to 0% if revascularization is delayed beyond 14 days. 18
Late HAT can remain clinically silent for months or years with an insidious course characterized by cholangitis, relapsing fevers, and bacteremia with nonspecific symptoms. 20 , 21 Arterial collaterals develop from the diaphragm, retroperitoneum, and the omentum as early as 2 weeks after LT, which explains why late HAT does not present with the devastating symptoms seen with early HAT. 12 , 22 , 23 As with early HAT, however, endovascular revascularization is a viable treatment option, yielding graft survival rates of 50%. 18
The first successful attempts at endovascular treatment of HAT began in 1989 with intra-arterial thrombolytic (IAT) infusion of urokinase (UK). 24 Since then, the use of IAT for HAT has been further studied and refined, yielding promising outcomes (▶ Table 24.1). 16 , 19 , 25 , 26 , 27 , 28 , 29 , 30 , 31 , 32 , 33 , 34 , 35 , 36 Larger reviews have found that IAT was successful in 47 of 69 (68%) patients, with complications occurring in 18 (26%) patients, 3 of which were fatal. 12 The risks of IAT include bleeding and rethrombosis of the hepatic artery; however, IAT has been used safely and effectively as early as 4 hours after transplant. 33 Nevertheless, before beginning IAT, the treatment team must be prepared to address hemorrhagic complications with termination of IAT, supportive blood products, and endovascular or surgical hemostasis as needed. Of note, recent research has raised concerns about the safety and efficacy of IAT, with a recanalization rate of 46% and a major complication rate of 42%. 36
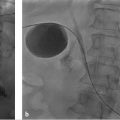
IAT for HAT (▶ Fig. 24.1) entails selective catheterization of the hepatic artery stump using a microcatheter, so knowledge of the patient′s anatomy and vascular anastomosis is critical. Infusion of thrombolytic drugs is initiated once the hepatic artery is selectively catheterized and the catheter is advanced to or within the thrombus. Although there is no consensus on dose and duration for IAT, alteplase (t-PA) infusion at 0.25 mg/h can be used for 12 to 14 hours with bolus infusions as deemed necessary. Use of the pulse-spray technique is encouraged because it may be able to reestablish flow in a single treatment and can prevent the need for overnight thrombolytic infusion. To limit bleeding risk, thrombolytic dosing should be monitored regularly through evaluation of fibrinogen levels. Adjunctive heparin is frequently used, with a target partial thromboplastin time of 1.25 to 1.5 times control. IAT success is determined by periodic evaluation with hepatic arteriography until the terminal branches of the hepatic artery are visualized. IAT is usually terminated if thrombus persists after 36 to 48 hours of therapy. Often, underlying stenosis is also present and must be corrected to avoid rethrombosis. For cases in which percutaneous techniques fail, surgical thrombectomy or retransplantation is the next step.

Hepatic Artery Stenosis
Post-LT HAS is insidious in onset; a high level of clinical suspicion and expert imaging are necessary for diagnosis. Early recognition and intervention can be important in HAS, as this condition often progresses to HAT and ischemic organ damage. 22 , 37 HAS typically occurs at or near the surgical anastomosis and is thought to be secondary to technical factors such as clamp injury, kinked vessels, faulty anastomotic suture placement, and differences in vessel caliber. 5 , 15 , 38 In the setting of graft rejection, however, HAS may be the result of severe hepatic dysfunction rather than its underlying cause. 39 Furthermore, the body may compensate for a slowly developing stenosis with collateral formation. 38 Ultimately, in patients with severe or late minimal hepatic dysfunction, intervention for isolated HAS may not affect graft survival and may not be warranted.
Doppler US is the method of choice for the diagnosis of HAS. Findings on US include focal increase in peak systolic velocity at the site of stenosis in the hepatic artery of two to three times the prestenosis velocity. Additional findings include poststenosis turbulent flow and the tardus-parvus intrahepatic waveform, with acceleration time greater than 8 ms and resistive index less than 0.5. When clinical suspicion for HAS is high, CT or MR angiography should be performed, as these modalities are more accurate in demonstrating narrowing of the hepatic artery. 2
The mainstay of endovascular treatment for HAS is percutaneous transluminal angioplasty (PTA) with or without stent placement (▶ Table 24.2). 37 , 40 , 41 , 42 , 43 , 44 , 45 , 46 , 47 , 48 , 49 , 50 , 51 , 52 , 53 , 54 , 55 Technical success rates of endovascular treatment for HAS (81–100%) are comparable to those of surgical revision (78–100%). 3 , 15 , 56 Overall, case series have demonstrated technical success rates of 90 and 98% and complication incidences of 16 and 19% with PTA and PTA plus stent placement, respectively. 57 Recurrent stenosis after PTA can be addressed by repeat PTA with or without stent placement, yielding 95 to 100% hepatic artery patency with 82 and 88% 5-year graft and patient survival rates, respectively. 41 , 58 Short focal lesions respond better to endovascular treatment than long segment or multifocal stenoses. 37 , 39 The major complications of endovascular HAS treatment include rupture, dissection, and technical failure. Although there is often concern about anastomotic rupture, PTA has been performed as soon as 10 or even 3 days after LT. 5 , 40
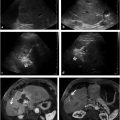
Endovascular treatment for HAS (▶ Fig. 24.2) begins by accessing and crossing the area of stenosis. The transstenotic pressure gradient can be measured to document a significant (> 10 mm Hg) gradient. A low-profile PTA balloon, usually 3 to 6 mm in diameter, is used to treat the stenosis. Heparin administration is standard before PTA, and a small amount of nitroglycerine can also be infused to prevent spasm. Patients should be observed and treated with anticoagulation overnight after PTA.

Metallic stents can be placed if patency is not adequately restored after PTA. Research has demonstrated generally comparable results for PTA with and without stent placement in HAS after LT. 57 In patients treated with stent placement, antiplatelet therapy should be considered, with recommendations varying from 30 days to > 1 year after the procedure. 52 , 55 , 57 Restenosis of previously treated HAS can be treated with repeat PTA or stent placement. Ultimately, in cases of HAS that cannot be treated with endovascular approaches, surgical therapies are indicated.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree