30 Osteonecrosis of the Hand Skeleton
Osteonecroses of the hand most often affect the lunate (Kienböck disease). Reparative processes play an important role in the natural course of the disease, which progresses in stages. CT imaging and MRI have influenced both early diagnosis and classification into stages of lunate osteonecrosis. Contrast-enhanced MRI provides evidence of reversible bone-marrow edema that cannot yet be detected radiographically (stage I), as well as zones of necrotic, reparative, and intact bone tissue in later stages. The precise identification of disease stages II–IV is best achieved with high-resolution CT, which identifies cancellous osteosclerosis, fractures of the proximal lunate circumference, and initial osteoarthritis. Recent findings in pathoanatomy and refined CT and MR imaging are reflected in Lichtman and Ross’s classification of Kienböck disease.
Synopsis
“Avascular” osteonecroses of the hand most often affect the lunate (Kienböck disease) and the proximal fragment in scaphoid nonunion. Manifestations on the rest of the hand skeleton are rare. Among these are nontraumatic osteonecrosis of the entire scaphoid (Preiser disease), as well as osteonecrosis of the capitate head, of all carpal bones (Caffey disease), of the metacarpal heads (Mauclaire disease), and of the bases of the phalanges (Thiemann disease).
Disease Entities
Lunate Osteonecrosis (Kienböck Disease)
Pathoanatomy and Clinical Symptoms
The etiogenesis of avascular osteonecrosis of the lunate is unclear. The following triggers, modulators, and predisposing factors have been discussed:
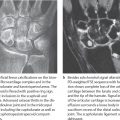
The most important etiology of lunate osteonecrosis is considered to be a traumatic injury of the nutrient vessels. Since the dominant hand of males in manual jobs is predominantly affected, chronic, repeated traumas are thought to be responsible for vascular occlusions. The special pattern of vascularization of the lunate is considered a predisposing factor. Vessels branching off from the radial artery enter the lunate via the dorsal horn and from the ulnar and the anterior intraosseous arteries via the palmar horn. Within the lunate these vessels form a Y-shaped (59%), an Ishaped (31%), or an X-shaped (10%) vascular network from which the proximal portion of the lunate is supplied only by some terminal arteries (Fig. 30.1a).
A single trauma to the wrist, especially when it involves the lunate with either an impaction or avulsion fracture, can probably trigger lunate osteonecrosis. This fracture theory is controversial, however, as lunate fractures often appear as secondary phenomena in the natural course of osteonecrosis.
Disturbance in venous drainage has recently been discussed as a possible cause of lunate osteonecrosis. Intraosseous measurements have revealed significantly elevated venous pressures in the necrotic lunate, especially when it is brought in an extended position.
Therapeutic consequences can be derived from the observation that lunate osteonecroses appear frequently together with a length discrepancy of the ulna. In about 70% of the cases, the ulna is more than 2 mm shorter than the radius when lengths are measured at the height of the distal radioulnar joint (negative ulnar variance). Two explanations are offered:
Changes in the transmission of force produce an increased axial load on the lunate in the radiolunate compartment.
Negative ulnar variances are often associated with a triangular form of the lunate and a changed cancellous structure. Lunate osteonecrosis has also been described in association with the pathologic radiolunate articulation in Madelung’s deformity.
Systemic causes are considered in neural, metabolic, and endocrinologic diseases, as well as in disturbances in ossification.
Pathoanatomically, lunate osteonecrosis is not merely a result of nonreactive, nonvital tissue, but a dynamic, viable remodeling process of the bone, analogous to osteonecrosis of the femoral head, as has been confirmed in animal experiments on lunate viability:
Following the influence of different triggers and modulators, repair and revascularization proceed from the remaining viable bony tissue. Angioneogenesis and the development of fibrovascular tissue occur.
A focal increase in circulation leads to focal osteopenia, which can cause a pathologic fracture, if this did not constitute the beginning of the chain of events.
For reasons not previously known, the fibrovascular reparative tissue around the fracture cannot be transformed into fibrous and lamellar bone (point of no return). This leads to separation and necrosis of areas in the bone.
Fracture and osteonecrotic tissue precede bony collapse. When fracture occurs in the osteonecrotic lunate, it leads to carpal collapse with progressive lost in height of the proximal carpal row and proximal migration of the capitate.
The final stage is osteoarthritis with destruction of the joint surfaces and progressive disability of the wrist.
Macroscopically, lunate osteonecrosis usually displays a three-layered structure, as illustrated in the radiograph of the excised lunate specimen in Figure 30.1b:
Maximum osteonecrosis is characteristically found on the proximal and radial circumference of the lunate, where the terminal zone of vascularization lies. The nonviable bone here has become severely sclerotic after unsuccessful reparative processes. However, the articular cartilage on the lunate remains intact for a long time.
In the middle layer, there is a reparative zone with fibrovascular connective tissue (granulation tissue), areas of decalcification, fractures, and removal processes. The region resembles a space of osseous nonunion filled with connective tissue.
The distal sections of the lunate including the palmar and dorsal horns contain viable bone marrow and bony tissue longer than the rest of the bone and only become necrotic in advanced stages of the disease.






Lunate osteonecrosis appears most often between the ages of 20 and 40 years; males are affected twice as often as females. The disease often begins asymptomatically. The first symptoms are load-dependent pain and restricted range of motion.
Swelling of the wrist appears later with loss of strength. In the advanced disease stages, there is also atrophy of the forearm musculature and signs of osteoarthritis of the wrist. During clinical examination, pain can be provoked by pressure on the dorsal wrist and later also on the palm and in the anatomical snuff box, as well as by axial compression on the third metacarpal.
Therapeutic decisions in lunate osteonecrosis are made according to the following parameters:
Stage of disease
Relative length of the ulna
Deforming osteoarthritis of the wrist
Age of the patient
Diagnostic imaging constitutes a decisive component in the therapeutic concept of lunate osteonecrosis by defining the first three parameters.
• Ståhl’s index of lunate height |
• Nattrass’s index of carpal height |
• Youm’s index of carpal height |
Diagnostic Imaging
Radiography
Conventional radiography constitutes the basic diagnostic procedure in Kienböck disease and is the basis of Decoulx’s classification. Both CT and MRI offer further definition of the pathomorphology of lunate osteonecrosis and subtle signs for classification into stages. Decoulx’s classification into four stages of disease should no longer be applied because
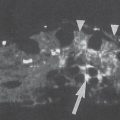
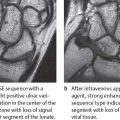
The initial stage of osteonecrosis can be detected only by means of MRI.
Subclassification of stage III depending on the presence of carpal alignment is useful.
The more recent Lichtman and Ross’s classification permits differentiation of the following stages of lunate osteonecrosis based on imaging parameters:
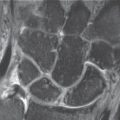
Stage I cannot be identified in radiographs because the cancellous structure and the shape of the lunate appear completely normal. As will be explained later, the initial stage of lunate osteonecrosis can be detected only in MR images, which show focal or diffuse bone marrow edema ( Fig. 30.2 ).
In stage II, there is the simultaneous presence of spotty osteosclerotic islands and cystic radiolucencies ( Fig. 30.3 ). These changes are either proximal or located diffusely in the bone, lending the lunate an inhomogeneous appearance. In the area of increased osteosclerosis, the trabeculae are thickened with indistinct contours. The sclerotic changes taking place during reparative processes are a sign of increased osteoblastic activity with formation of fibrous and lamellar bone tissue on the pre-existing trabeculae. The height and form of the lunate are maintained at this disease stage.
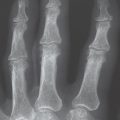
Stage III documents the initial fragmentation and progressive collapse of the lunate. Characteristically, the fracture begins subchondrally on the proximalradial articular surface of the lunate. In nondisplaced fracture without impaction of fragments, the lunate height is largely maintained, as is the stable alignment of the carpal bones – stage IIIa ( Fig. 30.4 ). However, if the lunate fragments are impacted, the carpal height progressively decreases – stage IIIb ( Fig. 30.5 ). Instability develops in which the scaphoid is in increased flexion toward the palmar side and forms a ring sign, and the triquetrum deviates toward the ulnar side. Differentiation between stages IIIa and IIIb is possible with the measuring procedure described in Table 30.1 .
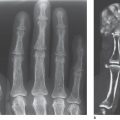
In stage IV, there is complete fragmentation, and the lunate is either wedge-shaped or impacted in its entirety ( Fig. 30.6 ). Often it has disintegrated into multiple fragments. The height index is decreased. There are signs of deforming osteoarthritis in the radiocarpal joint. Besides osteophytes, subchondral osteosclerosis, and narrowing of the joint spaces, small, intra-articular loose bodies (arthroliths) can appear.
Table 30.2 summarizes the criteria for classification of lunate osteonecrosis.
An important prerequisite for exact carpal assessment is radiographs in the neutral position, i.e., middle position between the rotational extremes of pronation and supination. The relative length of the ulna in relation to the radius depends to a large degree on the rotational position. In pronation, the ulna head is relatively distal to the radius; in supination, it has a more proximal position (radioulnar translation movement, relative ulnar variance). That iswhy only standard views in neutral position can be used to determine the relative length of the ulna.
Conventional tomograms with spiral or hypocycloid blurred motion figures are no longer recommended when high-resolution CT imaging is available.
Computed Tomography
High-resolution CT best visualizes the osseous anatomy of the lunate. If lunate osteonecrosis has already been diagnosed with another imaging procedure, CT should be performed to determine the exact stage of disease according to morphologic criteria.
Sagittal and coronal images reformated from axial source images are best suited for assessment of the proximal circumference of the lunate.
The introduction of multislice spiral CT fundamentally changed the technique of examining the lunate. Whereas sagittal and coronal images in the primary planes have to be acquired in sequential- and single-slice scanners, multislice spiral CT makes it possible to secondarily compute such images without artifacts as high-resolution, multiplanar reconstructions (MPR) in sagittal and coronal planes from a CT dataset initially acquired with thin axial slices. A prerequisite is slice thickness of less than 1 mm (0.5–1.0mm) and image reconstruction with a significantly overlapping increment. Advantages of this approach are the lack of “out-of-field” artifacts and better radiation protection, since only one dataset is acquired.
Computed tomographic staging of lunate osteonecrosis is indicated to provide
Evidence of stage II: Because of the high density resolution, osteoscleroses and pseudocystic inclusions are more evident in CT than in radiographs ( Fig. 30.7 ).
Evidence of stage IIIa: Linear fracture lines and impaction zones on the proximal lunate circumference are recognized earlier and with greater sensitivity in CT ( Fig. 30.8 ). Evidence of these fractures, which are often occult in conventional radiographs, confirms the presence of stage IIIa.
Evidence of stage IV: Initial stages of osteoarthritis in the form of small osteophytes, subchondral osteosclerotic zones, and asymmetrical joint spaces in the radiocarpal and midcarpal compartments can be confirmed with greater certainty in CT ( Fig. 30.9 ). A CT examination should be performed in suspected stage IIIb when osteoarthritis must be ruled out in planning surgical treatment.
Documentation of instability stage IIIb of lunate osteonecrosis does not constitute an indication for CT, as the criteria and height indices in Table 30.1 can be confirmed sufficiently with radiographs, as well as stage IV of lunate osteonecrosis, when it is clearly visible in conventional radiography.
CT often confirms a more advanced stage of lunate osteonecrosis than previously diagnosed. Table 30.3 summarizes the most important CT diagnostic criteria for lunate osteonecrosis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree