Acute Radiation Syndrome
▪ ACUTE RADIATION SYNDROME
The effect of ionizing radiation on the whole organism is discussed in this chapter. Data on the various forms of the acute radiation syndrome (ARS) have been drawn from many sources. Animal experiments provide the bulk of the data and result in a significant understanding of the mechanisms of death after exposure to total body irradiation. At the human level, data have been drawn from experiences in radiation therapy and studies of the Japanese survivors of Hiroshima and Nagasaki, the Marshallese accidentally exposed to fallout in 1954, and the victims of the limited number of accidents at nuclear installations, including Chernobyl. From these various sources, the pattern of events that follows a total body exposure to a dose of ionizing radiation has been well documented. To date, worldwide, about 400 humans have died of the ARS.
▪ EARLY LETHAL EFFECTS
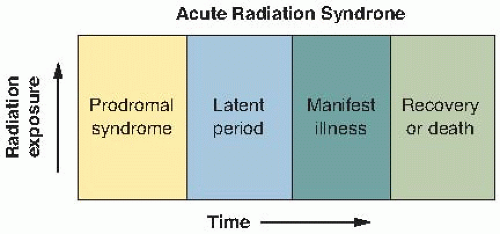
Early radiation lethality generally is considered to be death occurring within a few weeks that can be attributed to a specific high-intensity exposure to radiation. Soon after irradiation, early symptoms appear, which last for a limited period; this is referred to as the prodromal radiation syndrome. These symptoms may clear up after a few days, to be followed by a latent period before the development of the eventual life-threatening syndrome. This is illustrated in Figure 8.1. The eventual survival time and mode of death depend on the magnitude of the dose. In most mammals, three distinct modes of death can be identified, although in the circumstances of an actual accidental exposure, some overlap is frequently seen. At very high doses, in excess of about 100 Gy, death occurs 24 to 48 hours after exposure and appears to result from neurologic and cardiovascular breakdown; this mode of death is known as the cerebrovascular syndrome. At intermediate dose levels, approximately 5 to 12 Gy, death occurs in about 9 or 10 days and is associated with extensive bloody diarrhea and destruction of the gastrointestinal mucosa; this mode of death is known as the gastrointestinal syndrome. At lower dose levels, approximately 2.5 to 5 Gy, death occurs several weeks to 2 months after exposure and is caused by effects on the blood-forming organs; this mode of death has come to be known as bone marrow death or the hematopoietic syndrome.
The exact cause of death in the cerebrovascular syndrome is by no means clear. In the case of both of the other modes of death—the gastrointestinal and the hematopoietic syndromes—the principal mechanisms that lead to the death of the organism are understood. Death is caused by the depletion of the stem cells of a critical self-renewal tissue: the epithelium of the gut or the circulating blood cells, respectively. The difference in the dose level at which these two forms of death occur and the difference in the time scales involved reflect variations in the
population kinetics of the two cell renewal systems involved and differences in the amount of the damage that can be tolerated in these different systems before death ensues.
population kinetics of the two cell renewal systems involved and differences in the amount of the damage that can be tolerated in these different systems before death ensues.
▪ THE PRODROMAL RADIATION SYNDROME
The various symptoms making up the human prodromal syndrome vary with respect to time of onset, maximum severity, and duration, depending on the size of the dose. With doses of a few tens of gray, all exposed individuals can be expected to show all phases of the syndrome within 5 to 15 minutes of exposure. Reaction might reach a maximum by about 30 minutes and persist for a few days, gradually diminishing in intensity until the prodromal symptoms merge with the universally fatal cerebrovascular syndrome or, after a lower dose, with the fatal gastrointestinal syndrome.
At lower doses, dose-response predictions are difficult to make because of the interplay of many different factors. A severe prodromal response usually indicates a poor clinical prognosis and portends at the least a prolonged period of acute hematologic aplasia accompanied by potentially fatal infection: anemia and hemorrhage.
The signs and symptoms of the human prodromal syndrome can be divided into two main groups: gastrointestinal and neuromuscular. The gastrointestinal symptoms are anorexia, nausea, vomiting, diarrhea, intestinal cramps, salivation, fluid loss, dehydration, and weight loss. The neuromuscular symptoms include easy fatigability, apathy or listlessness, sweating, fever, headache, and hypotension. At doses that would be fatal to 50% of the population, the principal symptoms of the prodromal reaction are anorexia, nausea, vomiting, and easy fatigability. Immediate diarrhea, fever, and hypotension frequently are associated with supralethal exposure (Table 8.1). One of the Soviet firefighters at the Chernobyl reactor accident vividly described the onset of these symptoms as he accumulated a dose of several gray working in a high-dose-rate area. The prodromal phase is followed by a latent stage before the final radiation syndrome develops. In the symptom-free latent stage, the patient may seem and feel relatively well for a period of hours or even weeks. The duration of the latent stage is inversely proportional to the dose and may last a few hours for high exposures or as long as two or more weeks for lower exposures. Absence of a latent phase—that is, a progressive worsening from prodromal signs and symptoms directly into the manifest illness phase—is an indicator that the dose was probably very high.
The diagnosis of the ARS can also be based on laboratory data. During the prodromal phase, evidence of hematopoietic damage can already be observed by a drop in the lymphocyte count after an exposure as low as 0.5 Gy. The circulating lymphocytes are one of the most radiosensitive cell lines, and a fall in the absolute lymphocyte count is the best and most useful laboratory test to determine the level of radiation exposure in the early phase of observation. Among assays for biologic
dosimetry, chromosomal aberration analysis from cultured circulating lymphocytes is the most widely accepted and reliable. The dose-response relationships are well established in many laboratories around the world. The lower limit of detection of a dose by using this cytogenetic method is approximately 0.2 Gy of γ-rays or x-rays.
dosimetry, chromosomal aberration analysis from cultured circulating lymphocytes is the most widely accepted and reliable. The dose-response relationships are well established in many laboratories around the world. The lower limit of detection of a dose by using this cytogenetic method is approximately 0.2 Gy of γ-rays or x-rays.
Table 8.1 Symptoms of the Prodromal Syndrome | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||
▪ THE CEREBROVASCULAR SYNDROME
A total body dose of about 100 Gy of γ-rays or its equivalent of neutrons results in death in 24 to 48 hours. At these doses, all organ systems are also seriously damaged; both the gastrointestinal and hematopoietic systems are, of course, severely damaged and would fail if the person lived long enough, but cerebrovascular damage brings death very quickly, so that the consequences of the failure of the other systems do not have time to be expressed (i.e., death occurs before other symptoms have time to appear). The symptoms that are observed vary with the species of animal involved and also with level of radiation dose; they are summarized briefly as follows: There is the development of severe nausea and vomiting, usually within a matter of minutes. This is followed by manifestations of disorientation, loss of coordination of muscular movement, respiratory distress, diarrhea, convulsive seizures, coma, and finally death. Only a few instances of accidental human exposure have involved doses high enough to produce a cerebrovascular syndrome; two such cases are described briefly.
In 1964, a 38-year-old man working in a uranium-235 recovery plant was involved in an accidental nuclear excursion. He received a total body dose estimated to be about 88 Gy made up of 22 Gy of neutrons and 66 Gy of γ-rays. He recalled seeing a flash and was hurled backward and stunned; he did not lose consciousness, however, and was able to run from the scene of the accident to another building 200 yards away. Almost at once he complained of abdominal cramps and headache, vomited, and was incontinent of bloody diarrheal stools. The next day, the patient was comfortable but restless. On the second day, his condition deteriorated; he was restless, fatigued, apprehensive, and short of breath and had greatly impaired vision; his blood pressure could only be maintained with great difficulty. Six hours before his death, he became disoriented, and his blood pressure could not be maintained; he died 49 hours after the accident.
In a nuclear criticality accident at Los Alamos in 1958, one worker received a total body dose of mixed neutron and γ-radiation estimated to be between 39 and 49 Gy. Parts of his body may have received as much as 120 Gy. This person went into a state of shock immediately and was unconscious within a few minutes. After 8 hours, no lymphocytes were found in the circulating blood, and there was virtually a complete urinary shutdown despite the administration of large amounts of fluids. The patient died 35 hours after the accident.
The exact and immediate cause of death in what is known as the cerebrovascular syndrome is not fully understood. Although death is usually attributed to events taking place within the central nervous system, much higher doses are required to produce death if the head alone is irradiated rather than the entire body; this would suggest that effects on the rest of the body are by no means negligible. It has been suggested that the immediate cause of death is damage to the microvasculature, which results in an increase in the fluid content of the brain owing to leakage from small vessels, resulting in a buildup of pressure within the bony confines of the skull.
▪ THE GASTROINTESTINAL SYNDROME
A total body exposure of more than 10 Gy of γ-rays or its equivalent of neutrons commonly leads in most mammals to symptoms characteristic of the gastrointestinal syndrome, culminating in death some days later (usually between 3 and 10 days). The characteristic symptoms are nausea, vomiting, and prolonged diarrhea. People with the gastrointestinal syndrome lose their appetite and appear sluggish and lethargic. Prolonged diarrhea, extending for several days, usually is regarded as a bad sign because it indicates that the dose received was more than 10 Gy, which is inevitably fatal. The person with this syndrome shows signs of dehydration, loss of weight, emaciation, and complete exhaustion; death usually occurs in a few days. There is no instance on record of a human having survived a dose in excess of 10 Gy.
The symptoms that appear and the death that follows are attributable principally to the
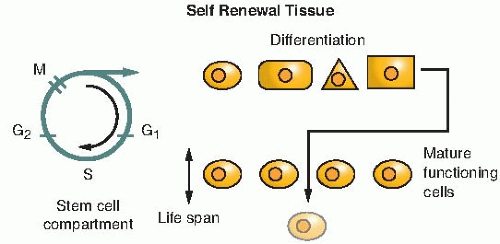
depopulation of the epithelial lining of the gastrointestinal tract by the radiation. The normal lining of the intestine is a classic example of a selfrenewing tissue; Figure 8.2 shows the general characteristics of such a tissue. It is composed of a stem cell compartment, a differentiating compartment, and mature functioning cells.
depopulation of the epithelial lining of the gastrointestinal tract by the radiation. The normal lining of the intestine is a classic example of a selfrenewing tissue; Figure 8.2 shows the general characteristics of such a tissue. It is composed of a stem cell compartment, a differentiating compartment, and mature functioning cells.
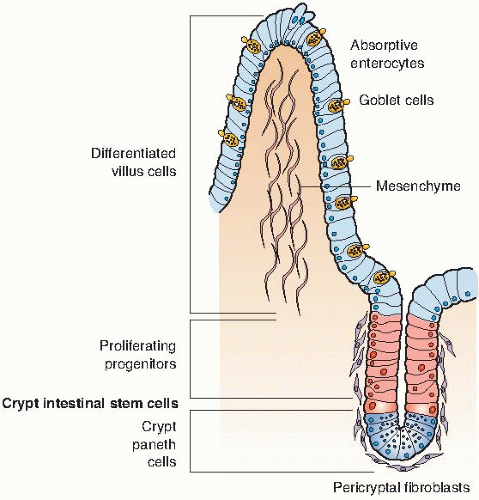
The structure of the intestinal epithelium is illustrated in Figure 8.3. Dividing cells are confined to the crypts, which provide a continuous supply of new cells; these cells move up the villi, differentiate, and become the functioning cells. The cells at the top of the folds of villi are sloughed off slowly but continuously in the normal course of events, and the villi are continuously replaced by cells that originate from mitoses in the crypts. A single-cell-thick barrier separates the blood vessels in the villi from the contents of the intestine.
A dose of radiation of about 10 Gy sterilizes a large proportion of the dividing cells in the crypts; a dose of this order of magnitude does not seriously affect the differentiated and functioning cells. As the surface of the villi is sloughed off and rubbed away by normal use, there are no replacement cells produced in the crypt. Consequently, after a few days, the villi begin to shorten and shrink, and eventually, the surface lining of the intestine is completely denuded of villi. The rate of cell loss and shrinkage depends on the dose. It occurs faster at higher doses than at lower doses. At death, the villi are very clearly flat and almost completely free from cells.
The precise time schedule of these events and the time required before the intestine is denuded of cells entirely vary with the species. In small rodents, this condition is reached between 3 and 4 days after the dose of radiation is delivered. In humans, it does not occur until about 9 to 10 days after irradiation. All of the individuals who received a dose large enough for the gastrointestinal syndrome to result in death have already received far more than enough radiation to result in hematopoietic death. Death from a denuding of the gut occurs, however, before the full effect of the radiation on the blood-forming organs has been expressed because of differences in the population kinetics of the stem cell systems involved.
Before Chernobyl, there was probably only one example in the literature of a human suffering a gastrointestinal death as a result of radiation exposure. In 1946, a 32-year-old man was admitted to the hospital within 1 hour of a radiation accident in which he received a total body dose of neutrons and γ-rays. The dosimetry is very uncertain in this early accident, and various estimates of total body exposure range
from 11 to 20 Gy. In addition, the man’s hands received an enormous dose, possibly as much as 300 Gy. The patient vomited several times within the first few hours of the exposure. On admission, his temperature and pulse rate were slightly elevated; other than that, the results of his physical examinations were within normal limits. His general condition remained relatively good until the 6th day, at which time signs of severe paralytic ileus developed that could be relieved only by continuous gastric suction. On the 7th day, liquid stools that were guaiac-positive for occult blood were noted. The patient developed signs of circulatory collapse and died on the 9th day after irradiation. At the time of death, jaundice and spontaneous hemorrhages were observed for the first time.
from 11 to 20 Gy. In addition, the man’s hands received an enormous dose, possibly as much as 300 Gy. The patient vomited several times within the first few hours of the exposure. On admission, his temperature and pulse rate were slightly elevated; other than that, the results of his physical examinations were within normal limits. His general condition remained relatively good until the 6th day, at which time signs of severe paralytic ileus developed that could be relieved only by continuous gastric suction. On the 7th day, liquid stools that were guaiac-positive for occult blood were noted. The patient developed signs of circulatory collapse and died on the 9th day after irradiation. At the time of death, jaundice and spontaneous hemorrhages were observed for the first time.
At autopsy, the small intestine showed the most striking change. The mucosal surface was edematous and erythematous, and the jejunum was covered by a membranous exudate. Microscopically, there was complete erosion of the epithelium of the jejunum and ileum, as well as loss of the superficial layers of the submucosa. The duodenal epithelium was lost, except in the crypts; the colon epithelium was somewhat better preserved. The denuded surfaces were covered everywhere by a layer of exudate in which masses of bacteria were seen, and in the jejunum, the bacteria had invaded the intestinal wall. Blood cultures postmortem yielded Escherichia coli.
Several of the firefighters at Chernobyl, including those who received bone marrow transplants, died between a week and 10 days after exposure, suffering from symptoms characteristic of the gastrointestinal syndrome.
▪ THE HEMATOPOIETIC SYNDROME
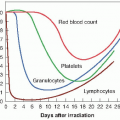
At doses of 2.5 to 5 Gy, death, if it occurs, is a result of radiation damage to the hematopoietic system. Mitotically active precursor cells are sterilized by the radiation, and the subsequent supply of mature red blood cells, white blood cells, and platelets is thereby diminished. The time of potential crisis at which circulating cells in the blood reaches a minimum value is delayed for some weeks. It is only when the mature circulating cells begin to die off and the supply of new cells from the depleted precursor population is inadequate to replace them that the full effect of the radiation becomes apparent.
The concept of the 50% lethal dose (LD50) as an end point for scoring radiation death from
this cause has been borrowed from the field of pharmacology. The LD50 is defined as the dose of any agent or material that causes a mortality rate of 50% in an experimental group within a specified period.
this cause has been borrowed from the field of pharmacology. The LD50 is defined as the dose of any agent or material that causes a mortality rate of 50% in an experimental group within a specified period.
Within a given population of humans or animals, there are many factors that influence the response of the individual to total body irradiation. For example, the very young and the old appear to be more radiosensitive than the young adult. The female, in general, appears to have a greater degree of tolerance to radiation than does the male. Figure 8.4 shows a typical relationship between the dose of radiation and the percentage of monkeys killed by total body irradiation. Up to a dose exceeding 2 Gy, no animals die, whereas a dose of about 8 Gy kills all the animals exposed. Between these two doses, there is a very rapid increase in the percentage of animals killed as the dose increases, and it is a simple matter by visual inspection of the graph or by a more sophisticated statistical analysis to arrive at a precise estimate of the LD50




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree