Paul N. M. Lohle
BACKGROUND
In the pathophysiology of heavy menstrual bleeding (HMB), two groups may be distinguished: HMB based on anatomic or organic abnormalities (fibroids, adenomyosis, clotting disorders) and essential HMB.1 There is relatively little knowledge about the exact pathophysiology of HMB in relation to fibroids, polyps, and adenomyosis.
Adenomyosis, associated with HMB, is a benign invasion of endometrium into the myometrium that results in a diffusely enlarged uterus that microscopically exhibits ectopic nonneoplastic endometrial glands and stroma surrounded by the hypertrophic and hyperplastic myometrium.2 The prevalence of adenomyosis is estimated to be 5% to 8% and up to 40% to 70%.2–4 This range may be explained by the different study designs, the random testing described in studies, the lack of a gold standard for identifying adenomyosis, and the use of different transvaginal ultrasound (TVUS), magnetic resonance imaging (MRI), and pathologic anatomical criteria for adenomyosis. Adenomyosis has either a focal or diffuse distribution in the uterine wall. Another problem regarding the relation between HMB and adenomyosis is the fact that in 60% to 80% of the cases, the uterus with adenomyosis is accompanied with other pathology, mostly fibroids, sometimes even in large numbers, small and/or large.3 Risk factors for adenomyosis are caesarean section, induced abortion or curettage, multiparity, miscarriage, endometriosis, and smoking.5
The clinical diagnosis of adenomyosis is sometimes difficult because the presenting symptoms overlap with common uterine disorders such as uterine fibroids.4,6 Therefore, adenomyosis is frequently a so-called forgotten diagnosis because of its noncharacteristic clinical appearance.7 Adenomyosis may be the cause of disabling symptoms such as, in particular, HMB; abdominal, pelvic, low back, and heavy menstrual pain, with or without bulk-related symptoms; and fertility issues in premenopausal women.8
The information on the occurrence of uterine adenomyosis varies considerably. The prevalence of adenomyosis in the specimen obtained from hysterectomy is reported between 8.8% and 31%.9,10 With broad criteria for the diagnosis of adenomyosis, a prevalence as high as 70% in women between 40 and 50 years of age is suggested.4 The incidence of adenomyosis in the population at risk is estimated at 8.1% to 16.7%, with symptomatology in approximately two-thirds of women. Of women with clinical manifestations of adenomyosis, about one-fifth are younger than 40 years of age, but most are between 40 and 50 years of age.11–13
Imaging of Adenomyosis
The first-line imaging technique, in women with suspected symptomatic adenomyosis, is usually TVUS, which is inexpensive and readily available. Fundamental TVUS signs to diagnose adenomyosis are an increased myometrial echogenicity or linear hyperechoic bands extending deep into the myometrium, indicating the presence of islets of ectopic endometrial tissue; hypoechoic areas in the myometrium compatible with hyperplasia of the muscle tissue surrounding the ectopic tissue; anechoic areas due to glandular dilatation or myometrial cysts; poor definition of the junctional zone; and an enlargement of the uterus with asymmetrical thickening of one of the walls. The presence of at least three of these signs is highly suggestive of adenomyosis.14
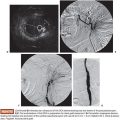
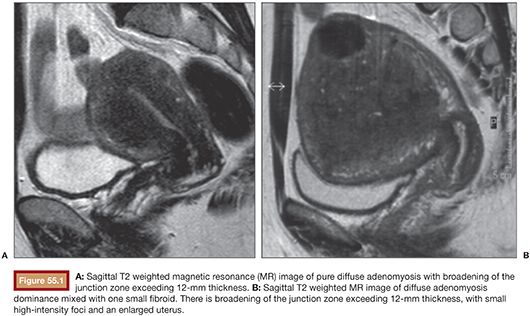
Particularly useful both in doubtful TVUS cases and in providing a complete evaluation of the disease with its panoramic views is MRI. The T2 weighted images with contrast-enhanced T1 weighted MRI provide the thickness of the junctional zone, which can be measured reliably; a thickness over 12 mm is considered diagnostic of adenomyosis (Fig. 55.1A). The occurrence of foci of high signal intensity within the myometrium of the uterus forms an additional but not an obligatory criterion (Fig. 55.1B). MRI can label adenomyosis as focal or diffuse and can be repeated in time to assess the effect of treatment. To assess the diagnostic power of MRI versus TVUS in women with adenomyosis, several studies have been conducted and published. A meta-analysis including six diagnostic accuracy studies of high quality compared TVUS and MRI with histologic proven adenomyosis.15 Champaneria et al.15 demonstrated that the correct diagnosis of the occurrence of adenomyosis was provided more often with MRI (sensitivity 0.77/specificity 0.89) than with TVUS (sensitivity 0.72/specificity 0.81), although without statistically significant difference. Another meta-analysis with the study of 1,898 women comparing the TVUS results with the histologic outcome of a symptomatic population of women showed similar sensitivity and specificity results as the meta-analysis of Champaneria et al.: TVUS sensitivity 0.83/specificity 0.85.16 In terms of accuracy, TVUS and MRI seem comparable for diagnosing adenomyosis. Regarding diagnostic imaging before uterine artery embolization, TVUS may very well identify fibroids and adenomyosis. Whether additional imaging such as MRI is needed in all cases is debatable. Nevertheless, in daily practice and from personal experience, MRI is less operator-dependent and therefore very useful and often better compared to TVUS in detecting adenomyosis and determining the vascularization and, thus, the infarction grade after embolization. The combination of both modalities in daily practice provides the best accuracy of ±90%.7,17
Treatment Option for Adenomyosis
Conservative Treatment for Adenomyosis
Women with symptomatic adenomyosis may receive medical treatment, which ranges from local treatment with the release of medications by an intrauterine device (IUD) to systemically administered treatment. IUD-released progestogens are used to reduce HMBs in women with adenomyosis. These medications, such as a Mirena coil, cause the decidualization of the endometrium of the uterus and, consequently, atrophic changes to decrease the amount of blood loss in these women with symptomatic adenomyosis.18
Existing medications for systemic administration include gonadotropin-releasing hormone (GnRH) agonists. GnRH agonists induce menopause by reducing the release of pituitary gonadotropins. The levels of estrogen are lowered, producing atrophy and reduced volume of the uterus with reduction of symptoms, because adenomyosis is an estrogen-dependent disease. If the therapy is stopped, the effect is reversible and symptoms will return.19
Surgical Treatment for Adenomyosis
The usual and preferred surgical approach for focal adenomyosis is excision or enucleation, but the kind of therapy is very much dependent on the type of lesion and the degree of myometrial involvement.19 The preferred surgical treatment for women with symptomatic adenomyosis and deep involvement of the myometrium is hysterectomy. The different surgical options and techniques for the treatment of symptomatic adenomyosis have their advantages and disadvantages. When hysterectomy is being considered for diffuse adenomyosis, transvaginal access is preferable to transabdominal access due to the lower morbidity and shorter hospital stay of the former. Concerning the complications of transvaginal hysterectomy, a study on two groups of patients with fibroids versus adenomyosis reported a higher risk of lesions to the urinary bladder in patients from the latter group.20 The reason is probably the greater complexity in identifying the vesicovaginal septum due to the presence of adhesions, which is explained by the frequent presence of pelvic adherences and endometriosis accompanying adenomyosis.8 Hysterectomy is regularly indicated as a definitive treatment. Rates of complication after hysterectomy range between 1.5% and 29.3%. Hysterectomy is associated with complications such as blood loss, bowel and general urogenital injury, pain, and infection. The recovery time ranges from 6 to 8 weeks,19,21,22 and health care–related expenses and lost time at work23 render hysterectomy an option associated with high costs.
Magnetic Resonance Imaging–Guided High-Intensity Focused Ultrasound for Adenomyosis
Magnetic resonance imaging–guided high-intensity focused ultrasound (MRI-HIFU) is a noninvasive image-guided technique that facilitates treatment of tumors by thermoablation with ultrasound waves. The applications are still under research. The therapy is entirely guided by MRI, providing therapy planning, monitoring, and visualization of the treatment result. MRI-HIFU has a broad spectrum of applications, including ablation of uterine fibroids.24 The advantage of the noninvasive character of the treatment is that it can be performed on an outpatient basis and that recovery is fast. MRI-HIFU has been used in adenomyosis with varying results. Therefore, although the technique seems to be a useful alternative, further studies are needed to clarify its effective role.25
TECHNIQUE
The gynecologist Ravina and interventional neuroradiologist Merland were the first to report the uterine artery embolization (UAE) treatment for symptomatic uterine fibroids in Paris.26 UAE has emerged as an effective therapy in the treatment of uterine pathology and, in particular, uterine fibroids. The clinical success rate of UAE for uterine fibroids with respect to symptomatic improvement of associated menorrhagia and pelvic pain ranges from 85% to 95% to 80% to 90%.27,28 This minimally invasive therapeutic alternative to surgery is reported to be associated with a high patient satisfaction.29–33 There is about 25% chance of failure of symptom control or recurrence after UAE for uterine fibroids at 5-year follow-up.34,35
The most common indication for selective uterine artery angiography is for embolizing symptomatic fibroids. Based on the resemblance of symptoms caused by fibroids and adenomyosis and the positive results after UAE for fibroids, this intervention has been investigated as an option to treat adenomyosis. Successful infarction of symptomatic fibroids with UAE has been obtained in women suffering from focal or diffuse adenomyosis with or without fibroids. The basic principle is selective infarction of the adenomyosis with particulate embolic materials delivered directly into the uterine arteries and/or uterine artery branches toward the adenomatous tissue in the uterus.
On the day of the UAE procedure, patients are prepared with or without intravenous antibiotic prophylaxis, sedation with 2.5 to 5 mg midazolam when required, and a urinary bladder catheter. Concerning the right moment to perform the UAE procedure in women with adenomyosis, Chen et al.36 evaluated UAE during the menstrual cycle in terms of differences in treatment efficacy. Patients were scheduled for UAE during the proliferative or luteal phase of menstruation. Their data showed that the difference in timing of UAE did not have a significant effect on efficacy in the treatment of adenomyosis.36
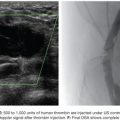
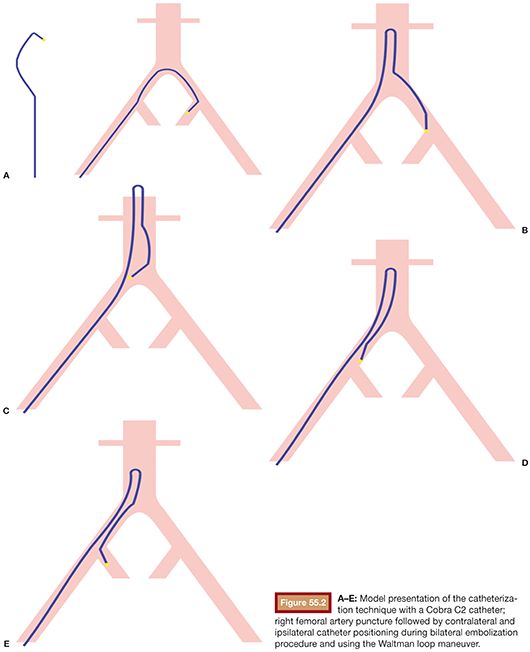
An applicable and accepted approach of UAE for adenomyosis is the bilateral UAE carried out after establishing arterial access to the common femoral artery. Selective catheterization of both uterine arteries is performed under fluoroscopic imaging. Bilateral embolization is mandatory. This is usually accomplished by a femoral artery puncture on one side followed by contralateral uterine artery catheterization and after the Waltman loop maneuver embolization of the ipsilateral uterine artery (Fig. 55.2). One may also favor a puncture of both femoral arteries, selecting the ipsilateral internal iliac artery with a recurved “hockey stick–like” catheter.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree