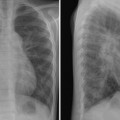
10 Aorta and Systemic Veins The fifth step of the chest radiographic interpretation is to assess the size and configuration of the ascending aorta, aortic arch, descending aorta, superior and inferior venae cavae, and azygos-hemiazygos veins. For example, this part of the report in a patient with known bicuspid aortic valve may read: “The right upper mediastinal border is mildly convex outward, suggesting a dilated ascending aorta in association with aortic stenosis.” The ascending aorta is not normally seen as a border-forming structure on a frontal chest radiograph in children and young adults. When it is dilated, the ascending aorta can bulge to the right beyond the superior vena caval border (Fig. 10.1). Dilatation of the ascending aorta can be caused by diseases affecting the aortic wall, increased blood flow volumes through the ascending aorta, or deformation of the aortic wall by the fast blood flow across a stenotic aortic valve (Table 10.1). Fig. 10.1 Dilated ascending aorta in an 11-year-old patient with aortic stenosis and insufficiency. The dilated ascending aorta results in a rounded bulging contour of the right upper mediastinum (arrows). This dilatation is caused by a poststenotic jet and increased blood flow volume from aortic regurgitation. The aortic knob (arrowheads) is also prominent when there is significant aortic insufficiency. Dilatation of the aortic root and ascending aorta is seen in various connective tissue diseases, particularly in Marfan syndrome (Figs. 10.2, 10.3). Aortic dilatation and mitral valve prolapse are the two most common cardiovascular manifestations of Marfan syndrome.1,2 Both manifestations usually develop later in childhood and progress gradually. Rarely, aortic dilatation is seen in early infancy, with very poor prognosis.3 The prevalence of both aortic dilatation and mitral valve prolapse in pediatric cohorts is approximately 80%.1 Aortic regurgitation develops as a complication of aortic dilatation during childhood and adolescence in 25% of Marfan patients, whereas mitral valve prolapse results in mitral regurgitation in 50%. Loeys-Dietz syndrome is a much more aggressive and lethal disease characterized by aortic and arterial tortuosity, aneurysm formation, and a propensity for early dissection.3 Aortic dilatation is also common in other connective tissue diseases, such as Ehlers-Danlos syndrome and pseudoxanthoma elasticum.4,5 Aortic dilatation, however, can occur without definable underlying disease and can cause aortic regurgitation.6 Familial dilatation of the aorta has also been reported. Aneurysm of the aortic sinus of Valsalva may cause a localized bulge of the lower part of the right upper heart border.
 Ascending Aorta and Aortic Sinus
Ascending Aorta and Aortic Sinus
| Diseases affecting the aortic wall | Marfan syndrome |
| Loeys-Dietz syndrome | |
| Ehlers-Danlos syndrome | |
| Pseudoxanthoma elasticum | |
| Familial | |
| Idiopathic | |
| Increased blood flow volumes | Aortic regurgitation |
| Aortopulmonary window | |
| Patent ductus arteriosus | |
| Congenital heart diseases with right-to-left shunt, for instance tetralogy of Fallot | |
| Truncus arteriosus | |
| Poststenotic dilatation | Aortic valve stenosis |
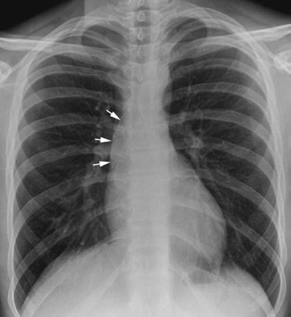
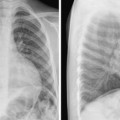
Fig. 10.2 Dilated ascending aorta in a 15-year-old patient with Marfan syndrome. The ascending aorta has a mildly convex contour (arrows) in the right upper mediastinum. The aortic knob and descending aorta appear normal
Aortic regurgitation results in aortic and left ventricular dilatation because of volume overload (Fig. 10.1). Aortic regurgitation can be due not only to aortic valvular abnormalities but also to aortic root dilatation, as discussed in the previous paragraph. Bicuspid aortic valve is often complicated by later development of aortic stenosis and regurgitation due to chronic mechanical injury and inflammation. Among patients with aortic valve stenosis or regurgitation, dilatation of the ascending aorta develops earlier and is more severe in patients with bicuspid aortic valve than in those with tricuspid aortic valve.7 Aortic regurgitation is typically related to shrunken valvular tissue in rheumatic or rheumatoid disease and ankylosing spondylitis and to perforated valvular cusps in infective endocarditis. Aortic valve prolapse is not an uncommon complication of a ventricular septal defect that is located below the aortic valve and may result in aortic regurgitation (Fig. 10.4).8 Congenital aortic regurgitation is rare. Congenital regurgitation presenting in the neonatal period occurs more commonly through a route in the paravalvular space, which is called an aorto–left ventricular tunnel.9
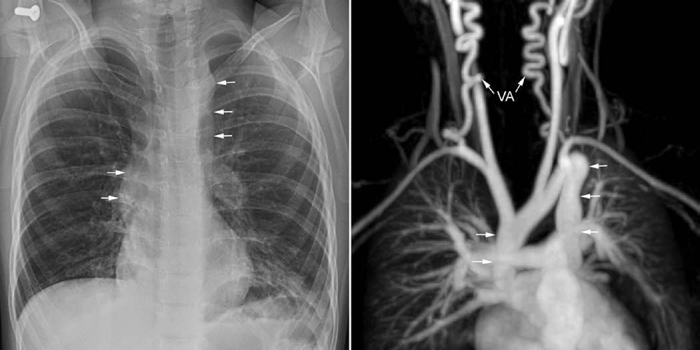
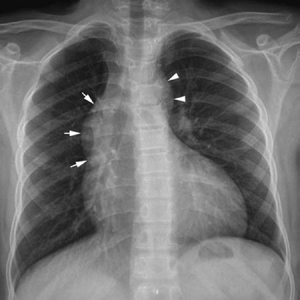
Fig. 10.3 Tortuous aorta and vertebral arteries (VA) in a 4-year-old patient with Loeys-Dietz syndrome. The contour of the ascending and descending aorta (arrows) seen on a frontal chest radiograph is well correlated with that seen on a contrast-enhanced magnetic resonance angiogram.
Left-to-right shunt lesions at the great arterial level also cause dilatation of the ascending aorta. They include patent ductus arteriosus and aortopulmonary window. However, we rarely see obvious radiographic findings of dilatation of the ascending aorta in these conditions in children. Congenital heart diseases with an intracardiac right-to-left shunt are also characterized by a dilated ascending aorta (Fig. 10.5). There is a reciprocal relationship in size between the two great arteries in most cases. The ascending aorta becomes smaller after surgery, but residual dilatation is common. Common arterial trunk in truncus arteriosus can also be seen as a round bulge of the right upper mediastinum. Dilated ascending aorta is more obvious when the thymus is shrunken after birth or is congenitally hypoplastic or aplastic in association with chromosome 22q11 deletion that is common in truncus arteriosus and tetralogy of Fallot.
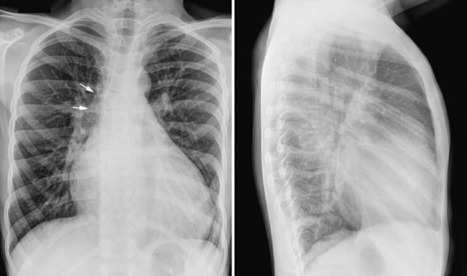
Fig. 10.4 Prominent ascending aorta (arrows) and aortic knob and cardiomegaly with a dilated left ventricle in an 11-year-old patient with a ventricular septal defect complicated by aortic valve prolapse and aortic regurgitation. The pulmonary vascularity is increased.
Significant aortic valve stenosis results in acceleration of the blood flow. The accelerated flow usually streams toward the right anterior wall of the ascending aorta, causing a rounded outward bulge of the right upper mediastinal margin (Fig. 10.1). The degree of poststenotic dilatation of the ascending aorta varies according to the severity of aortic stenosis. Newborns with severe aortic stenosis show a hypoplastic ascending aorta because of reduced blood flow volume through the aortic valve. Bicuspid aortic valve is the most common underlying substrate for later development of aortic valve stenosis. The bicuspid leaflets are prone to develop thickening and calcification later in life, causing stenosis, regurgitation, or both.7 Rheumatic fever, which was the leading cause of aortic stenosis up through the early 20th century, has become rare in developed countries. Recurrent inflammation from rheumatic fever causes fibrous thickening and contracture of the cardiac valve leaflets, resulting in stenosis and regurgitation. Rheumatic fever affects most commonly the mitral valve, less commonly the aortic valve, and rarely the tricuspid valve. Aortic stenosis is associated with left ventricular hypertrophy, although the left ventricle may dilate when it fails.
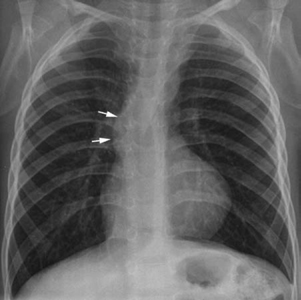
Fig. 10.5 Dilated ascending aorta in a 26-month-old patient with tetralogy of Fallot. The dilated ascending aorta (arrows) is clearly seen when there is involution of the thymus, which when present often hides the enlarged aorta.
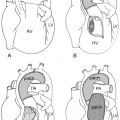
The ascending aorta may have an abnormal left-sided position in congenitally corrected transposition of the great arteries and double inlet left ventricle with the transposed aorta arising from the left-sided rudimentary right ventricle (Figs. 10.6, 10.7). The left-sided ascending aorta causes an unusual prominence of the left upper mediastinum, often with a rounded bulge. When this configuration is seen in the presence of dextrocardia, the diagnosis of congenitally corrected transposition or double-inlet left ventricle with discordant ventriculoarterial connection can be entertained. However, asymmetric prominence of the left lobe of the thymus may mimic this configuration (Fig. 10.8).
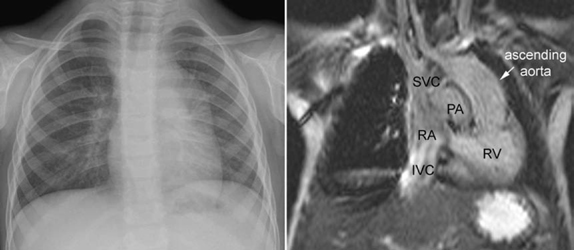
Fig. 10.6
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree