Chapter 2 ARTHROGRAPHY AND JOINT INJECTION AND ASPIRATION: Principles and Techniques
Joint injection/aspiration is a commonly performed procedure in many radiology practices, but in some practices it is uncommon and radiologists have variable experience. This chapter outlines simplified techniques for performing these procedures as well as different approaches to joints that have been described.
GENERAL TECHNIQUES
CT Arthrography
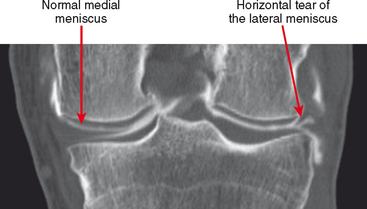
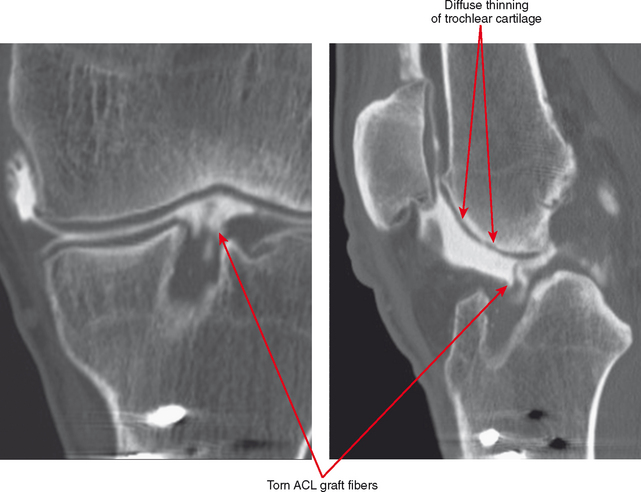
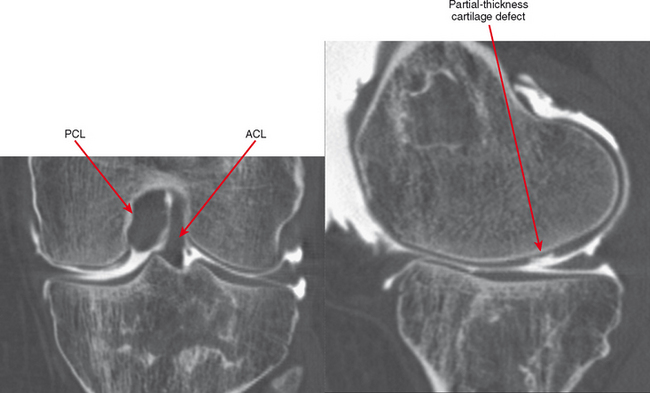
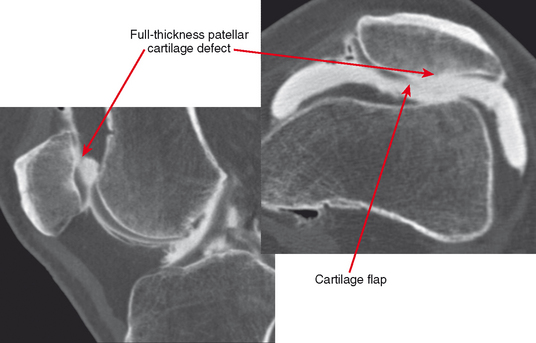
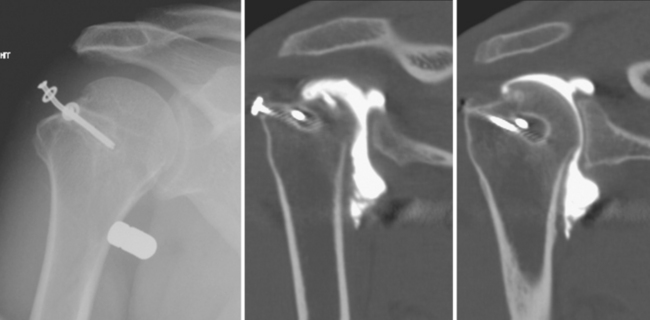
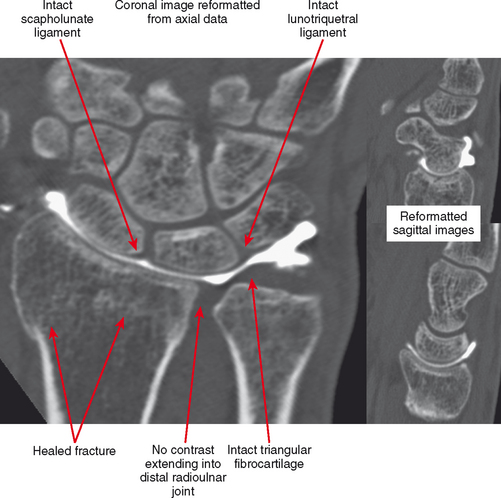
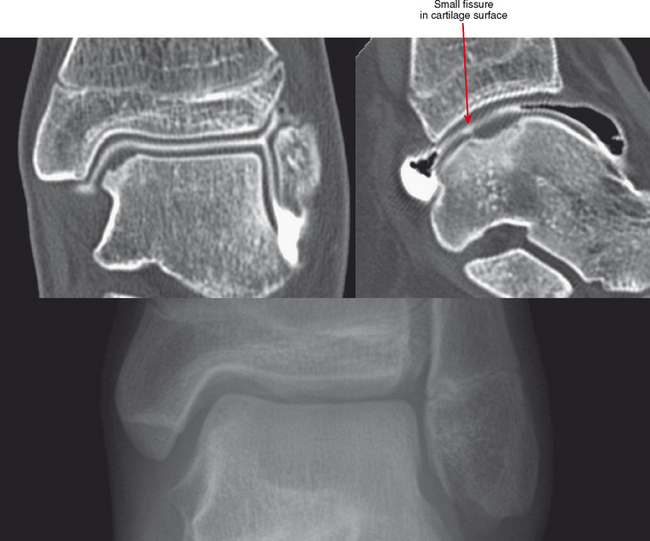
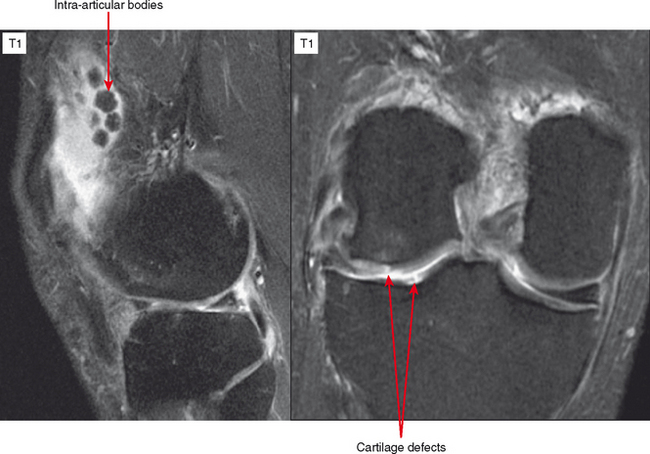
CT arthrography (CTA) is useful for demonstration of cartilaginous and osseous intra-articular bodies, cartilage defects, fracture fragments, synovial abnormalities, and ligamentous disruption (Figs. 2-1 through 2-7). CTA is usually reserved for patients with contraindications to MRI, but is being performed more commonly recently owing to improved scanner technology that provides isotropic volumetric data.
CTA involves the intra-articular injection of iodinated contrast material typically followed by axial CT scanning. With multidetector CT technology, very thin sections allow for acquisition of volumetric data, thereby allowing reconstructions to be performed in any plane without loss of resolution. The newergeneration multidetector CT scanners generate a higher photon flux, thus decreasing the streak artifact that often limited CTA on older scanners. Therefore, 300 mg/mL nonionic iodinated contrast can be used without the need for intra-articular air injection when a multidetector scanner is used and excellent quality images can be acquired. With older-generation CT scanners, it is prudent to dilute contrast (typically 50:50) with sterile saline or to use a less concentrated iodinated contrast preparation to avoid streak artifact. Many authors in the past have advocated injection of variable amounts of air as a negative contrast agent. This is useful to distend the joint without creating significant artifact, and the air can be moved about the joint by putting the patient in different positions. Some authors “coat” the synovium with only a few milliliters of contrast, filling the rest of the joint with air. These techniques have fallen by the wayside with the advent of multidetector CT technology.
MR Arthrography
Direct MR Arthrography
Conventional MRI has enjoyed great success in imaging the musculoskeletal system and has deservedly become the “gold-standard” imaging technique for suspected internal derangement of joints. There are, however, several limitations of conventional MRI examinations, including the inability to visualize small intra-articular structures and the fact that many pathologic processes have similar signal intensity to normal anatomic structures. Postoperative findings may also be similar in signal intensity to pathologic changes. Unless there is an effusion, conventional MRI may be somewhat limited owing to nondistention of the joint.
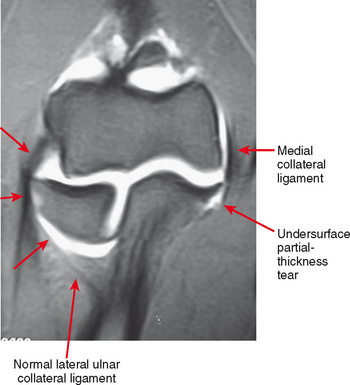
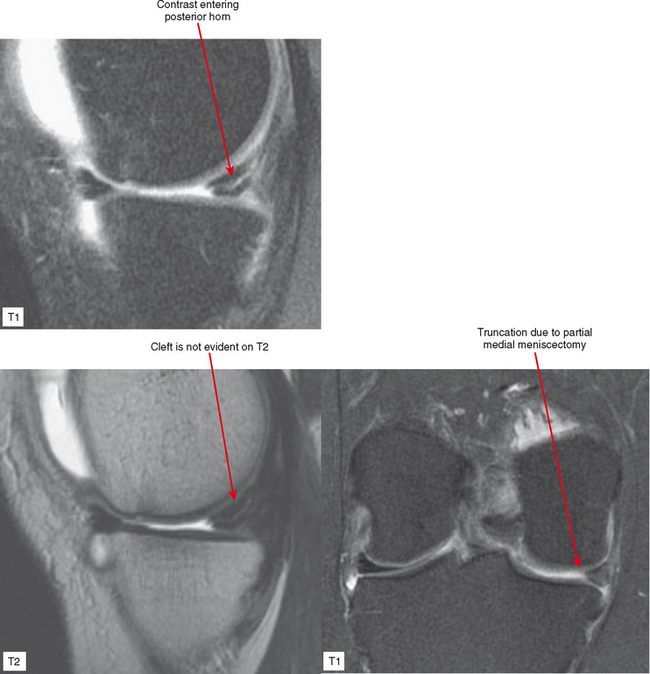
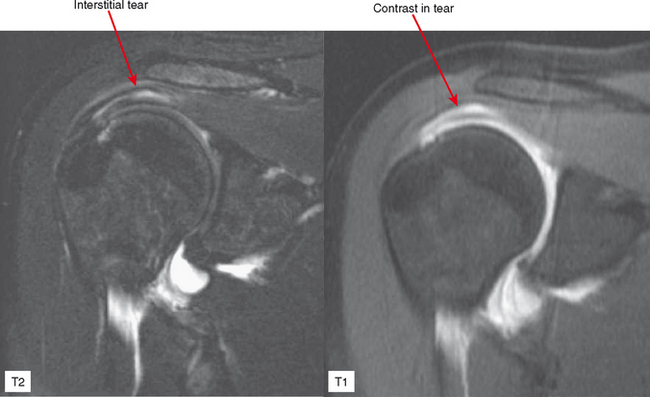
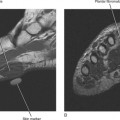
Direct MRA involves the direct injection of dilute gadolinium, followed by MR imaging. This type of MRA leads to improved intra-articular contrast owing to the T1 shortening effect of gadolinium and also provides distention of the joint, which allows smaller intra-articular structures to be visualized (Figs. 2-8 through 2-12). The distention effect alone of saline injection has proved to be better than conventional MRI in some studies. The distention effect also forces intra-articular contrast through and around pathologic entities. With adequate distention, intraarticular gadolinium enters any pathologic entity in communication with the joint space. MRA provides excellent soft tissue contrast and demonstrates many abnormalities beyond the resolution of conventional MRI. One of the main disadvantages of MRA is the presence of artifacts seen in patients with implanted prostheses and metallic hardware.
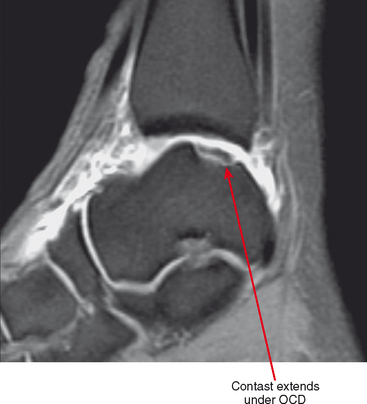
Figure 2-8 Direct MR arthrography of the ankle documenting instability of an osteochondral lesion of the talus.
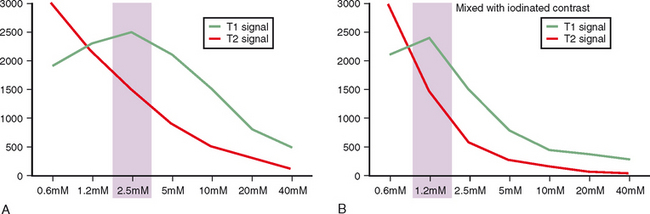
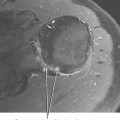
To perform direct MRA, the gadolinium injected should be diluted to 2.5 mM. To achieve this concentration we add 0.1 mL of gadopentetate dimeglumine to 20 mL of normal saline. The final gadolinium dilution ratio should be 1:200 to achieve maximal signal (Fig. 2-13). It has been demonstrated that iodinated contrast reduces the T1 shortening effect of gadolinium. This effect is seen on both high and low field-strength systems. It is therefore advisable to use as little iodinated contrast as needed when performing direct MRA examinations. The quality of the examination on direct MRA does not appear to be affected by patient exercise. We therefore encourage patients to ambulate normally to the MRI suite after injection, but we do not routinely prescribe nor avoid exercise after injection for direct MRA.
Indirect MR Arthrography
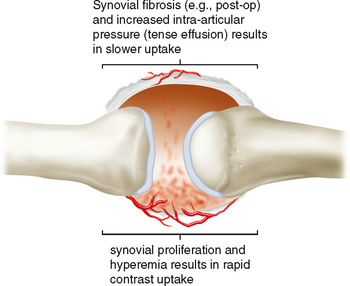
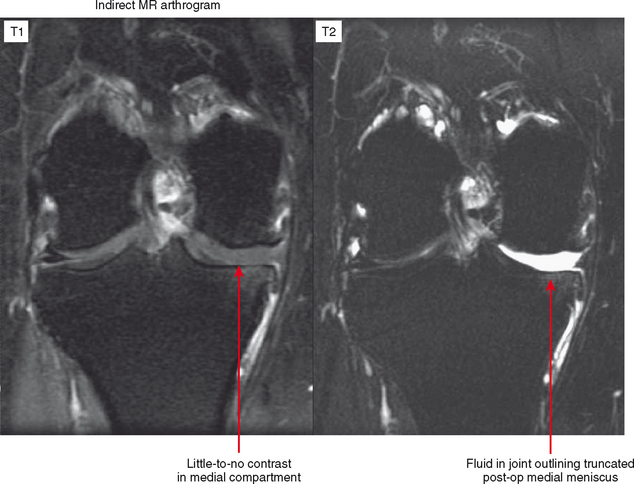
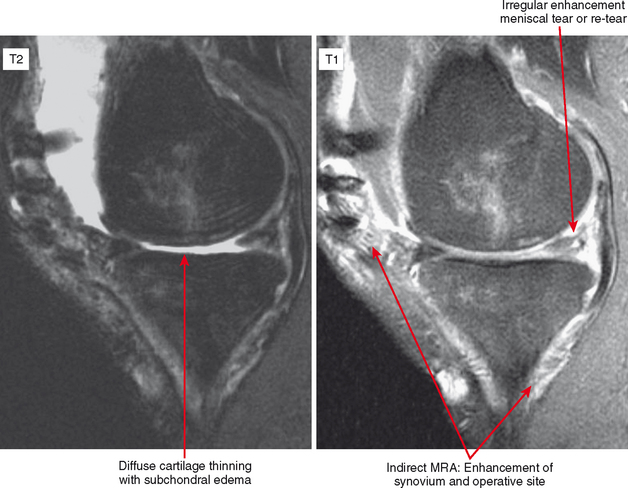
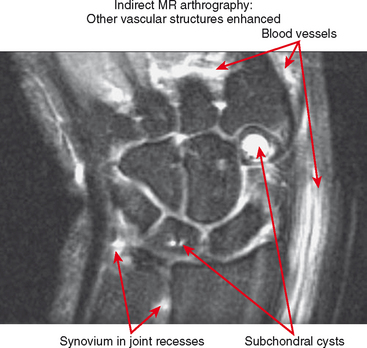
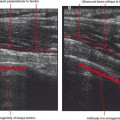
Indirect MRA involves the injection of a standard dose of 0.1 mmol/kg of intravenous gadolinium followed by delayed imaging to create an “arthrographic effect” as the contrast diffuses into the joint (Fig. 2-14). As the gadolinium diffuses from the bloodstream into the synovial compartment of the joint being imaged, the degree of arthrographic effect depends on the volume of synovial fluid within the joint and on the degree of synovial vascularity. Although this technique is attractive from a logistical standpoint, there is an interpretive learning curve, and certain limitations should be recognized. One of the main disadvantages of indirect arthrography is the lack of joint distention (unless a preexisting effusion was present) (Fig. 2-15). Typically, patients with small to moderate joint effusions and synovial reaction achieve a better arthrographic effect with indirect MRA. A tense effusion may prevent or delay contrast uptake because of increased intra-articular pressure. Also, a large effusion or insufficient delay (usually at least 30 minutes is required between injection and imaging) may cause heterogeneous or incomplete filling of the joint with gadolinium (Fig. 2-16). Smaller joints such as the wrist and ankle and the small joints of the hands and feet achieve a more consistent arthrographic effect owing to the high synovial area/joint volume ratio. Exercise after injection of intravenous gadolinium increases diffusional flow into the patient’s joint and improves the arthrographic effect. One of the main advantages of indirect arthrography is the fact that it can be performed offsite without the need for an invasive procedure. This technique may also be more acceptable to patients because it involves an intravenous injection only. When optimal, indirect MRA can be as effective as a direct MR arthrogram (Figs. 2-17 and 2-18).
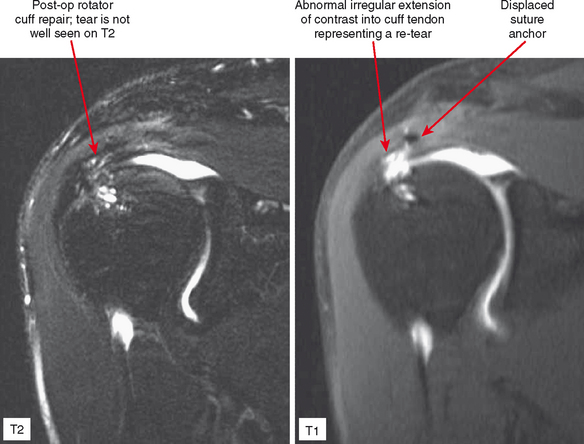
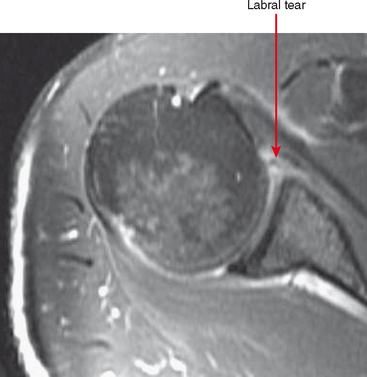
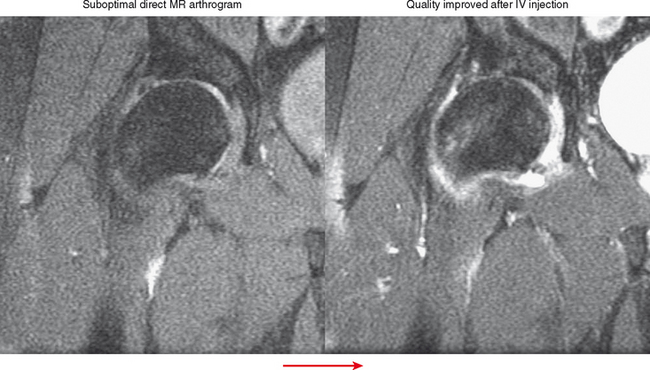
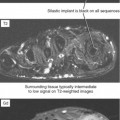
Indirect arthrography is not optimal in the setting of low signal-to-noise ratio or where fat suppression is not available (e.g., imaging with low-field MRI or deep structures such as the hip). Heterogeneous enhancement within large joints can lead to misdiagnosis, and optimally patients should be monitored and additional delay or exercise used if this is the case. Interpreter errors can occur with this technique owing to enhancement of normal intra-articular structures (such as the periphery of the triangular fibrocartilage complex) (Fig. 2-19). Indirect arthrography is also useful in the postoperative patient, but is considered less useful than direct MRA because postoperative granulation tissue can also enhance and may be confused with recurrent tear. On the other hand, this can be an advantage in the nonoperated joint. Granulation tissue is often intermediate-to-low signal on T2-weighted images; yet the tissue can exclude fluid or contrast. As a result, chronic injuries such as glenoid labral tears can potentially go undetected on routine MRI as well as with direct MRA. Although more research is needed, enhancement on indirect MRA could theoretically identify these lesions (Fig. 2-20). Intravenous injection can also be used to salvage a suboptimal direct MR arthrogram (Fig. 2-21). Advantages and disadvantages of indirect MRA compared with the direct approach are summarized in Box 2-1.
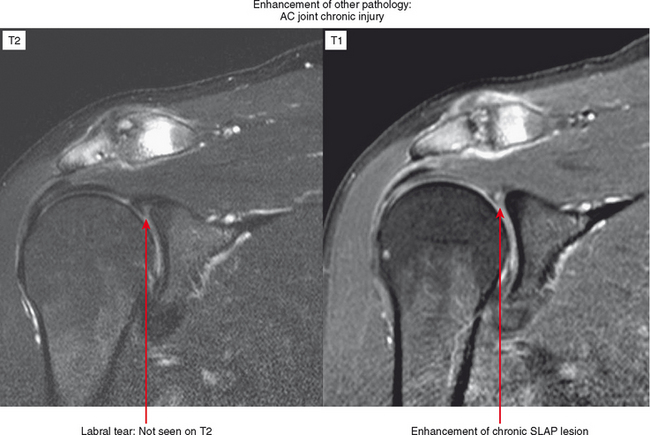
Figure 2-20 On indirect MR arthrography, scar enhances; therefore, this technique may prove to be very useful for detection of chronic injuries containing granulation tissue. Such injuries, when intra-articular such as the labral tear in Figure 2-15, may not allow contrast injected directly into the joint to enter if scar has formed. AC, acromioclavicular.