, Valery Kornienko2 and Igor Pronin2
(1)
N.N. Blockhin Russian Cancer Research Center, Moscow, Russia
(2)
N.N. Burdenko National Scientific and Practical Center for Neurosurgery, Moscow, Russia
The objective of treatment of brain metastases, in addition to a probable prolongation of life by monitoring the intracranial disease progression, is to reduce the severity of neurological symptoms or prevent their occurrence in order to preserve patient’s quality of life as long as possible. Due to the treatment success in general, the duration of life of cancer patients increases and, therefore, increased the frequency of detection of tumor metastases in the brain. Effective therapy and local control of brain metastases are of paramount importance for prognosis and ensuring a satisfactory quality of life (Law et al. 2002; Chiang et al. 2004; Opstad et al. 2004).
The choice of treatment, whether surgical removal of metastases, radiation exposure (hypofractionated radiation therapy, radiosurgery, whole-brain radiotherapy), or drug therapy, is based on an assessment of three main parameters: patient’s general status, morphological structure of malignant tumor, and the number and characteristics of sites of metastatic lesions. The key point for the clinician when selecting the treatment tactics is the expected extent of radical treatment; therefore, the possibility of surgical removal of the tumor(s) is estimated during the first phase.
4.1 Surgery
In 1926, Grant published his experience in resection of a metastatic tumor in the brain. In his opinion, a surgery for brain metastatic tumors is not only contraindicated but also “may harm patients.” Pessimism regarding the surgical treatment of cerebral metastases began to decrease after the publication by Oldberg (1933), who described three cases of removal of brain metastases with long-term positive outcomes: one patient with breast cancer metastases lived for 2 years after the surgery. In 1951, Stortebecker, based on the analysis of 125 cases of patients who underwent surgery for brain metastases, concluded that the removal of metastases increased the life expectancy of patients. Randomized trials in 1990s of the last century showed a significant increase in the life expectancy of patients with solitary metastases, who underwent a surgical removal of secondary tumors and subsequent brain radiotherapy, as compared with patients who received only brain radiotherapy (Patchell et al. 1990a, b; Vecht et al. 1993; Mintz et al. 1996). The most favorable outcomes with a median survival of patients from 10 to 15 months after the surgical removal of intracranial metastases were achieved for breast cancer (Defesche 1982; Wronski et al. 1996; Cappuzzo et al. 2000; Iwadate et al. 2000). Good outcomes of complex treatment in patients with solitary brain metastases with additional implantation carmustine wafers into the resected tumor bed and subsequent radiotherapy were published in the paper by Ewend (2001).
Despite the occurrence of alternative methods within the last decade and changes in the treatment paradigm for intracranial metastases, the role of surgery for certain groups of patients with metastatic brain lesions is fundamental for creating optimal conditions for the further complex antitumor treatment (Kalkanis et al. 2010; Caroli et al. 2011; Patel et al. 2012; Mut 2012; Owonikoko et al. 2014).
A surgical resection followed by whole-brain radiotherapy (WBRT) is currently the standard treatment for resectable solitary cerebral metastases. The presence of brain metastases with an untreated or undetected primary tumor is not a contraindication for surgery (Kryuchkov and Yartsev 1974; Lang and Sawaya 1996; Nakagawa et al. 1997; Iwadate et al. 2000).
The key factors affecting the survival of patients include the histological tumor structure and the extent of administered (adjuvant) treatment for postoperative cerebral metastases, the disease extent and the duration of a disease-free period, and the patient’s age and neurological status (Sundaresan and Galicich 1985; Pieper et al. 1997; Lagerwaard et al. 1999; Hatiboglu et al. 2011; Zaitsev et al. 2015). The best overall survival rates are achieved in patients with high performance status (≥80 on the Karnofsky scale) and control of extracranial tumor process. It should be remembered that low performance status in some patients may be due to the mass effect and can be improved as a result of a surgical removal of the tumor.
In case of multiple brain metastases, the role of surgery is limited to obtaining a biopsy or eliminating mass effect symptoms caused by large lesions (Loshakov 2005; Paek et al. 2005; Stark et al. 2005). There are some retrospective findings showing the benefits of a surgical tumor resection (best overall survival) for certain patients with a good prognosis and limited (2–3 lesions) brain metastases (Mintz et al. 1996; Schackert et al. 2000).
Surgical treatment is indicated in the presence of metastatic lesions causing:
Clinical manifestations of mass effect, accompanied by signs of intracranial hypertension
A dislocation of midline brain structures
An extensive perifocal swelling spreading to the nearby lobes and the opposite hemisphere
A threat of blocking the cerebrospinal fluid pathways
Accordingly, a surgical intervention solves the following issues: the maximum extent of tumor removal, while observing the principles of anatomical justification and functional safety, exact determination of the histological tumor structure, reduction of the tumor impact on the unaffected brain structures, and restoration of impaired liquor circulation. The main factor determining a favorable outcome of the surgical treatment is complete removal of metastases. To this end, the best methods of postoperative assessment are CT (2–7 days) or MRI studies with contrast enhancement (1–2 days).
Removal of intracerebral metastases is indicated with a limited (≤3) number of focal lesions and the presence of a resectable lesion greater than 3 cm in diameter. In case of solitary metastases (one metastasis in the brain and absence of extracranial manifestations), surgical treatment is indicated if the latter are >3 cm. When metastases are <3 cm, located in functionally important areas, and symptomatic (neurological symptoms) and there is no response to the steroid test (administration of 8–24 mg of dexamethasone per day for 5 days), a surgical removal can be considered. However, when choosing between surgical and radiosurgical methods, one should consider a higher probability of persistent residual neurological deficit after the surgery.
In cases of radioresistant metastases (kidney cancer , melanoma ) sized up to 3 cm, surgical treatment is preferable. In case of solitary metastases and extracranial manifestations of the disease, indications for surgical treatment are similar to the indications for treatment of solitary metastases. Surgical treatment is the most effective, provided control of extracranial metastases and presence of systemic treatment reserves.
Surgical treatment is also possible in multiple brain metastases and with at least one >3 cm lesion determining the severity of patient’s condition. Favorable outcomes depend on the control of extracranial manifestations and/or existence of systemic treatment reserves.
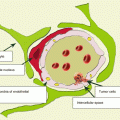
Surgery of brain metastases is characterized by removal of a metastasis as a single unit with the surrounding perifocal and perivascular area. This removal method reduces the risk of local recurrence up to 5%, in contrast to piecemeal removal, where the risk of local recurrence is 40%.
Surgery is also indicated in case of local recurrence of a brain metastasis or active progression of post-radiation necrosis. Surgical removal of an abnormal lesion is also possible in active pseudotumor course of post-radiation necrosis with mass effect and development of persistent disabling neurological symptoms. In this case, the point of surgery is to remove (not radically) the major part of the necrotic tissue in order to eliminate the mass effect.
Thus, despite a broad discussion on the role of surgical treatment of brain metastases and selection of alternative methods, we believe that to date surgery has no alternatives in identification of the histological tumor structure, rapid elimination of the tumor mass effect, perifocal edema, management of clinical symptoms caused by the tumor, and improvement of patient’s neurological status in general. As noted above, surgery helps create conditions for continued complex treatment of patients.
4.2 Radiosurgery
Radiosurgery (RS) in many cases is a direct alternative to surgical treatment. The choice in favor of radiosurgery is based mainly on the size of the locally irradiated area, which depends on the volume of the largest metastatic lesions or a total volume of the adjacent tumor foci forming a conglomerate.
According to RTOG 90-05 (Shaw et al. 2000), the maximum permissible doses of ionizing radiation during radiosurgical treatment of brain metastases (MTSs) are as follows:
15 Gy—for MTSs with the maximum diameter of 3–3.5 cm (recommended level 1)
18 Gy—for MTSs with the maximum diameter of 2–3 cm (recommended level 1)
24 Gy—for MTSs with the maximum diameter of 2 cm (recommended level 1)
Radiosurgical treatment is limited by the brain tissue volume irradiated with the dose 12 Gy, which should not exceed 15 cm3. This rule approximately corresponds to the limitation of an irradiated lesion size (not more than 14 cm3), which is a more useful parameter when considering the possibility of radiosurgery. When the volume is difficult to calculate, the maximum lesion diameter of ≤3 cm (14 cm3) is an alternative to the volume calculation.
A direct indication for radiosurgery is the presence of a limited number of metastases (≤3 foci) (lesions with the maximum diameter ≤3 cm). Patients with multiple (3–10) MTSs can also be candidates for radiosurgery (Golan et al. 2015).
Radiosurgical treatment as a separate method of treatment requires careful monitoring of patients in order to early detect a possible relapse and subsequently repeat the radiosurgical treatment. The usual practice is to perform follow-up using thin-slice (1 mm) MRIs of the brain and intravenous administration of gadolinium-containing contrast agent at least once in 3 months.
Another treatment option is a combination of radiosurgery and whole-brain radiotherapy (WBRT) that is often used in case of multiple metastatic lesions (more than 10 MTSs). In this case, radiosurgery is performed on lesions with the diameter greater than 1 cm, followed by (in 1–2 weeks) WBRT. For this combination, the recommended radiation dose is reduced by 20–25% from the usual dose.
Hypofractionated stereotactic radiotherapy is recommended for the treatment of large, inoperable lesions or conglomerates (>3 cm in diameter).
An important feature of radiosurgical treatment is the possibility of repeated treatment, both for local relapses and for the treatment of “new” distant metastases.
4.3 Hypofractionated Stereotactic Radiotherapy
Stereotactic radiation therapy (SRT) of brain metastases can be considered an effective method to achieve a certain level of local control for large (>3 cm in diameter) MTSs. This level for large tumors can probably be improved using higher doses of radiation with a large number of factions in a hypofractionated mode. In phase II of a prospective study, Ammirati et al. (2010) confirmed the effectiveness of CPT regimen with total boost dose (TBD) = 30 Gy in 5 fractions. A biologically equivalent dose (BED) was calculated as 40 Gy (SBD = 2 Gy) for acute effects and as a 60 Gy (SBD = 2 Gy) for delayed effects, assuming the ratio of α/β = 10 Gy for acute effects and α/β = 2 Gy for delayed effects (Mehta et al. 2010). This study allowed to use equivalent (for BED) fractionation modes: 3 fractions of 8 Gy, TBD = 24 Gy; 5 fractions of 6 Gy, TBD = 30 Gy; and 7 fractions of 5 Gy, TBD = 35 Gy.
According to published studies and our own experience, adjuvant CPT on the resected tumor bed in certain clinical situations may replace preventive WBRT. Using high TBD on the minimum volume of surrounding normal brain tissue around the removed metastasis bed improves local control by 70% (for WBRT) to 85.5% (for stereotactic radiation therapy) and has low neurotoxicity (Golan et al. 2015).
Indications for hypofractionated stereotactic radiotherapy are as follows:
Metastases larger than 2.5–3 cm in diameter
Metastases located in critical brain structures
The bed of a removed single metastasis in the postoperative period
Metastatic lesions at the skull base, orbit, and posterior pole of the eye
A promising option is so-called staged radiosurgery when a 3 cm lesion is initially irradiated with a recommended dose of 10–12 Gy with a view to achieve a short-term reduction of the metastasis and to decrease the risk of local radiation necrosis. The effect of such treatment is unstable; therefore, within 1 month after the first treatment session, provided a reduction in the tumor volume, the second stage of treatment is performed with a higher recommended dose, to reinforce the desired effect. Preliminary results are encouraging but require a more detailed further study.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree