Chapter 27 Breast Cancer
Epidemiology
Breast cancer remains the most common cancer diagnosis and the number two cause of cancer-related death in women in the United States, with an estimated 40,610 deaths in 2009.1,2 Leukemia is the leading cause of death in females 20 years and younger, and lung cancer is the leading cause of death in women 60 years and older.1 The latest data on worldwide estimated number of deaths from breast cancer totaled 519,000 in 2004, with 69% occurring in developing countries.3 Breast cancer survival rates vary greatly around the world, ranging from less than 40% in impoverished or developing nations to 80% and higher in North America.4 This disparity in breast cancer–related mortality is most likely related to the lack of screening and early-detection programs, as well as inadequate treatment facilities, in countries with limited healthcare resources, which results in a higher proportion of women presenting with late-stage disease.3
Screening mammography has been demonstrated to reduce breast cancer–specific mortality by 20% to 30% in randomized clinical trials. Between 1991 and 2005, the overall death rate from breast cancer decreased by 37% in the United States1; this decrease was most likely related to progress in treatment options and screening technology and the implementation of the Mammography Quality Standard Act of 1992, which established a national screening program. A national and government-funded mammography screening program is too costly for many countries because it requires a substantial financial investment to build the healthcare infrastructure required to maintain this program long term. However, the financial investment in preventive healthcare would be worthwhile because mammography screening in the community setting has been shown to reduce breast cancer mortality not only in the United States but also in other developed countries.5,6
In the United States, the segment of the population 65 years and older will increase as the baby boom generation, individuals born between 1946 and 1964, ages.7 Because breast cancer incidence increases with age—from 1 in 208 for women 39 years and younger to 1 in 16 for women 70 years and older2—the incidence of breast cancer is expected to increase as well, even though the incidence of newly diagnosed breast cancer cases decreased by about 3.5% per year from 2001 to 2004.1 This decrease in new breast cancer cases has been most strongly linked to a decrease in the use of hormone replacement therapy during this period.8,9
In addition to age and country of residence, other factors affect the incidence of and mortality from breast cancer in women worldwide. Such factors include race and ethnicity, with a higher incidence of breast cancer in African American and white women living in the United States than in women living in Asia, South America, or Eastern Europe. Asian American women born in the West have a higher risk of breast cancer than those who were born in Asia; this finding suggests that environmental and behavioral factors, rather than genetic predisposition alone, contribute to the risk of breast cancer.10 Studies identifying potentially modifiable risk factors for breast cancer provide better opportunities for breast cancer prevention among women who have an average-to-high risk of developing breast cancer.
Screening
Mammography
Mammography is utilized for two major purposes: (1) to screen asymptomatic women for breast cancer and (2) to evaluate breast problems or abnormalities. Although the United States does not have a government-funded screening program, the screening guidelines recommended by the National Cancer Institute advise that women 40 years and older who are at average risk of breast cancer undergo screening mammography every 1 to 2 years.11 Since the introduction of screening mammography, the number of deaths from breast cancer has declined in every age and racial group of women in the United States.12 Some investigators attribute this decline to mammography screening, whereas others believe it is related to advances in therapy. The 2002 Swedish screening trial reported that screening mammography reduced breast cancer mortality by 30%, supporting the concept that the decline in breast cancer mortality rates is related to the utilization of screening mammography.13 Today, the benefits of screening mammography for women between the ages of 50 and 69 years is universally accepted whereas screening for women between the ages of 40 and 49 years remains controversial despite the statistically significant reduction in breast cancer mortality14 and significant increase in survival rates15 reported.
Screening mammography consists of mediolateral oblique and craniocaudal views of each breast. In the United States, a standardized terminology for interpreting mammograms, the Breast Imaging Reporting and Data System (BI-RADS), has been developed by the American College of Radiology to help facilitate uniformity in mammography reporting across facilities.16
Currently, mammography studies are performed using either film screen or full-field digital mammography (FFDM) technologies. FFDM technology has slowly replaced film screen mammography because FFDM provides a lower radiation dose in large breasts; increased image quality associated with a higher contrast resolution; shorter examination times, and higher patient throughput; elimination of costs associated with film such as film storage, film, and processing; and facilitation of teleradiology for mammography.17 Furthermore, cancer detection rates with current FFDM technology are equivalent to those with film mammography.17 In addition to advancing detector and processing technology, advanced techniques that operate in conjunction with FFDM, such as digital tomosynthesis, computer-assisted diagnosis algorithms, and contrast-enhanced mammography, are current areas of research that may improve the detection of small early-stage cancers and decrease the false-positive recall rate (Figure 27-1).
The potential role of physician-performed screening ultrasonography, as an adjunct to mammography screening, has been evaluated in a multi-institutional trial that demonstrated many barriers to widespread implementation, such as high false-positive rates and low reimbursement rates.18 In high-risk populations, such as women with a genetic predisposition for breast cancer, MRI has been reported to have a higher sensitivity than either ultrasonography or mammography for the detection of breast cancer; therefore, MRI is a more appropriate adjunct to screening mammography in these cases.
Magnetic Resonance Imaging
Use of screening mammography in the general population has been shown to reduce mortality associated with breast cancer by at least 24%.19 Women at high risk of breast cancer owing to a known genetic predisposition should be engaged in aggressive surveillance and screening, which should begin at a younger age than screening for women in the general population because of the potential for early onset associated with familial breast cancer. Current surveillance protocols for screening women with a BRCA1 or BRCA2 mutation include a clinical breast examination every 6 months and annual mammography and MRI.20
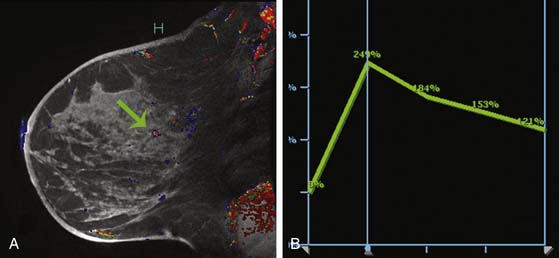
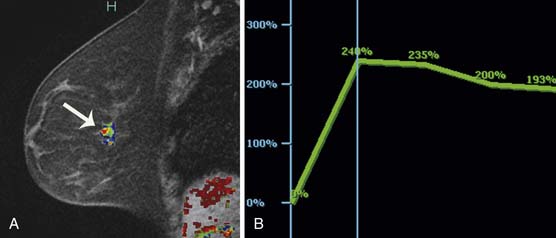
Multiple studies have demonstrated higher cancer detection rates with MRI than with mammography, although the differences were not statistically significant.21–23 Despite variations in breast cancer screening techniques between facilities, MRI is consistently reported to have a higher sensitivity (77-100%) than mammography (25-40%).24–26 MRI detects more breast cancers than either mammography or sonography.24–26 However, MRI also consistently demonstrates a higher false-positive rate and, thus, is associated with a higher biopsy rate—8.2%—than mammography (2.3%) or sonography (2.3%).26 The majority of breast cancers detected on MRI screening demonstrate the typical conspicuous morphologic and dynamic enhancing patterns expected for malignant lesions (Figure 27-2). However, in a trial studying patients at high risk of breast cancer, 20% of the breast cancers detected on MRI screening presented with non-masslike enhancement and 33% presented with an enhancing pattern typical for benign lesions.27 This lack of specificity associated with current morphologic and kinetic MRI features makes MRI detection of breast cancers in high-risk patients challenging. A study by Kuhl and coworkers28 compared clinical breast examination, mammography, sonography, and MRI in 529 study participants. The combination of mammography and MRI yielded the highest sensitivity (93%), which agrees with the findings from other published trials. In this trial, MRI and mammography had the same specificity (97%), as opposed to a higher specificity for mammography than MRI reported in prior studies. This improved specificity of MRI may be due to improved imaging techniques.28
While the role of MRI in breast screening programs continues to evolve, the full impact of modern MRI screening technology on mortality from breast cancer is still unknown. Patients for whom MRI is being considered should be informed of the current status of MRI in the detection of breast cancer, and appropriate management strategies are best determined on an individual basis. At the present time, no consensus exists as to the appropriateness of MRI as an adjunct to screening mammography in the general population.
Anatomy and Pathology
Anatomy
The breast is located in the superficial fascia and is attached to the skin by fibrous connective tissue and suspensory structures, called Cooper’s ligaments. Fibrous connective tissues interdigitate between the Cooper ligaments and the glandular tissue within the breast. Posterior to the breast tissue is the retromammary bursa, located between the deep layer of the superficial fascia and the deep layer of the pectoralis major muscle.29 The breast extends from the second or third rib to the sixth or seventh rib, or inframammary fold. The muscles that the breast rests on are the pectoralis major, serratus anterior, and external abdominal oblique muscles and the upper extent of the rectus sheath. The growth of the mammary gland is influenced by the levels of estrogen, progesterone, and adrenal corticoids; during pregnancy or lactation, growth of the mammary gland is also influenced by prolactin (from the adenohypophysis) and somatomammotropin (from the placenta).
Pathology
Breast cancer is a heterogeneous disease and has been traditionally categorized as in situ lesions (DCIS and lobular carcinoma in situ [LCIS]) or invasive cancers. The in situ or noninvasive disease encompasses a spectrum of lesions and is not as simple as the original definition implies30; in situ lesions develop in a linear progression from normal breast tissue to atypical ductal hyperplasia, DCIS, and invasive cancers. DCIS predominantly arises from the terminal ductal-lobular unit and may extend into the extralobular ducts.
The conventional pathologic classifications of invasive breast cancers by morphology include invasive ductal carcinoma, invasive lobular carcinoma, tubular carcinoma, mucinous carcinoma, medullary carcinoma, papillary carcinoma, metaplastic carcinoma, and other less common types. Tubular carcinoma accounts for 3% to 5% of all invasive breast carcinomas. It is a low-grade cancer with an excellent prognosis and usually occurs in older women.31 Mucinous or colloid carcinoma is characterized on histologic examination as having extracellular pools of mucin and low-grade tumor aggregates. Pure mucinous carcinoma also has a very favorable prognosis, with a 10-year survival rate of 90%.31 Medullary carcinoma is commonly seen on imaging as a circumscribed mass consistent with the distinctive smooth pushing border seen on pathology.32 Medullary carcinoma also has a favorable prognosis, but it is not observed as frequently as tubular and mucinous carcinomas. This type of carcinoma comprises a higher proportion of BRCA-related breast cancers than sporadic cases but still represents a minority of the breast cancers in women with BRCA mutations. Metaplastic carcinomas include tumors that show both epithelial and mesenchymal features. Although there are conflicting data on the biologic behavior of these tumors, many exhibit an aggressive clinical course with sarcoma-like behavior.33 Inflammatory breast cancer is a rare but aggressive disease with a poor prognosis; it is usually diagnosed clinically owing to the inflammatory symptoms. In addition to the type of breast cancer, the receptor or tumor marker status of the cancer (estrogen receptor [ER], progesterone receptor [PR], and human epidermal growth factor receptor 2 [HER2] receptor) is very important for determining the treatment options in patients with invasive breast carcinoma. The status of these receptors is also important for patients with in situ disease who are considering hormone therapy.
Even though genetic profiling has not been widely used in clinical practice, the research on this technology has resulted in a molecular classification of breast cancer with the hope of offering insight on the prognosis and response of tumors to specific therapy. Based on genetic profiling, breast cancers are divided into five subtypes: luminal A, luminal B, normal breast–like, HER2+, and basal-like carcinomas.34 Luminal A breast cancer is characterized by the high expression of ERs and PRs. These cancers tend to be of a lower grade and have a better prognosis than the other subtypes. Luminal B breast cancers not only express ERs and/or PRs but also overexpress HER2. They are more aggressive, with high tumor grade. The normal breast–like type or unclassified subtype is a triple-negative tumor with a molecular profile similar to that of basal-like carcinoma; however, normal breast–like carcinoma has a better prognosis than basal-like carcinoma. The HER2+ subtype includes ER-negative and ER-positive types and is often associated with DCIS. The basal-like breast carcinomas lack ER, PR, and HER2 expression and, therefore, are often called triple-negative tumors. These tumors show a high rate of p53 mutations and are common in BRCA mutation carriers. Triple-negative cancers tend to be more sensitive to chemotherapy but have a worse prognosis than other subtypes.2,35 However, not all triple-negative breast cancers are basal-like tumors. Some data suggest that 76% of triple-negative breast cancers are basal-like, and 77% of the basal-like tumors are triple negative.31 This molecular classification will likely be further modified as additional molecular features are discovered. Future classifications should help predict the sensitivity of particular tumors to targeted therapies, thereby improving the selection of patients for specific therapeutic approaches.
Key Points Pathology
• Breast cancer is a heterogeneous disease and can be in situ, invasive, or both.
• Invasive breast cancers are classified by morphology: ductal, lobular, tubular, mucinous, medullary, papillary, metaplastic, and others.
• Breast cancer subtypes are determined by gene profiling: luminal A, luminal B, normal, HER2+, and basal-like.
• ER, PR, HER2, and lymphovascular invasion status are important risk factors for relapse.
Clinical Presentation
The detection of cancer at an early stage, when the cancer is small, may reduce cancer-related mortality and improve survival. Cancers can be detected on palpation or examination of breast symptoms rather than with imaging screening, such as mammography or ultrasonography. In a national survey of 41,000 breast cancer patients, approximately 70% of the patients presented with a palpable breast mass.36 A more recent study of 592 breast cancer cases reported that the cancer was detected as a palpable mass and not at screening in 43% of the patients.37 The investigators evaluated the role of breast self-examination as a screening tool and reported that women who performed breast self-examinations did not have lower breast cancer mortality rates than women who did not practice breast self-examinations.37
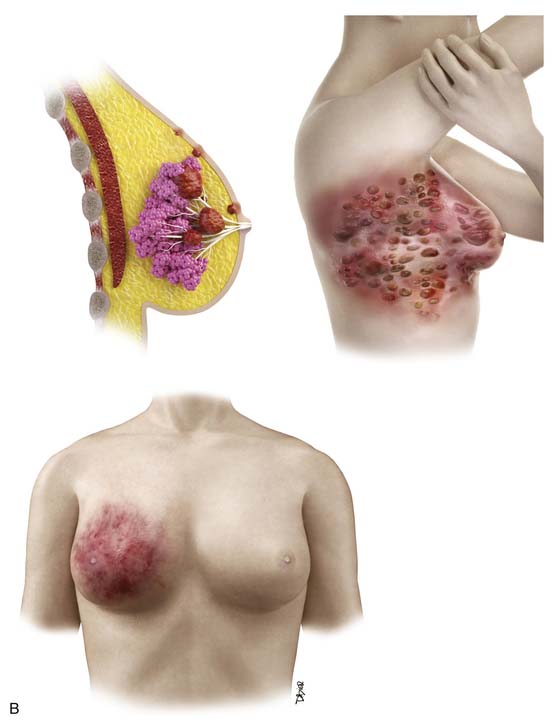
A clinical breast examination should evaluate breast symmetry and shape, palpable lumps, and skin changes. Some common clinical presentations of breast cancer include a breast mass, asymmetrical thickening or nodularity, nipple retraction, bloody nipple discharge, skin dimpling, and erythema. For a palpable breast mass, further evaluation on imaging and, possibly, a core needle or fine-needle aspiration biopsy would be needed. For asymmetrical thickening or nodularity, further evaluation with ultrasonography is needed and, possibly, mammography for women 30 years or older. For nonspontaneous nipple discharge and discharge from multiple ducts, the suspicion of breast cancer is low. These patients are advised to stop compression of the breast and elicitation of the nipple discharge. However, spontaneous nipple discharge with unilateral and single duct involvement, as well as discharge that is serous, sanguineous, or serosanguineous in nature, would require investigation with mammography, ultrasonography, MRI, or ductography to determine the etiology and rule out a malignancy (Figure 27-3). Skin changes in women older than 40 years would raise a concern of either infection or inflammatory breast cancer. Skin changes may include peau d’orange or skin thickening, Paget’s disease with scaling, eczema, or nipple excoriation. Other associated clinical symptoms of concern include lymphadenopathy and weight loss.
Staging Classification
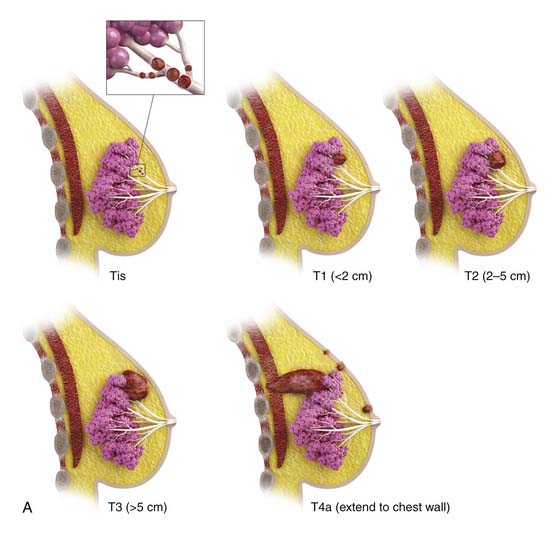
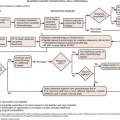
The most popular staging system for classifying breast cancer is the tumor-node-metastasis (TNM) system (Table 27-1), which includes the primary breast tumor size, the spread of cancer to the regional lymph nodes, and the spread of cancer to distant sites.38 The pathologic staging is based on the tumor size of the final pathology specimen. For multiple synchronous ipsilateral primary carcinomas, the largest tumor is used. Once the tumor size, node status, and presence of metastatic disease have been determined, one of the five breast cancer stages is assigned (Table 27-2). Stage 0 is assigned to precancerous lesions or carcinoma in situ with no local or distant metastasis; this stage is associated with a nearly 100% cure rate. Stage I is assigned to small cancers confined to the breast; patients with stage I disease have an excellent prognosis. Stage II cancers have regional lymph node metastases, and stage III breast cancers have large tumors or locally advanced disease at the time of initial diagnosis. Stages II and III are associated with a poor prognosis. Stage IV cancers have a distant metastasis and are associated with a poor survival (Figure 27-4).
Table 27-1 Tumor-Node-Metastasis Classification of Breast Cancer
| STAGE | CHARACTERISTICS |
|---|---|
| T | Primary Tumor |
| Tx | Primary tumor not assessed |
| To | Primary tumor not detected |
| Tis | DCIS, LCIS, Paget’s disease of nipple with no tumor. |
| T1 | Tumor 2 cm or smaller in greatest dimension |
| T1mic | Microinvasion ≤0.1 cm or smaller in greatest dimension |
| T1a | Tumor larger than 0.1 cm but 0.5 cm or less in greatest dimension |
| T1b | Tumor larger than 0.5 cm but 1 cm or less in greatest dimension |
| T1c | Tumor larger than 1 cm but 2 cm or less in greatest dimension |
| T2 | Tumor larger than 2 cm but 5 cm or less in greatest dimension |
| T3 | Tumor larger than 5 cm in greatest dimension |
| T4 | Tumor of any size with direct extension to chest wall or skin |
| T4a | Extension to chest wall, but not pectoralis muscle |
| T4b | Edema |
| T4c | T4a and 4b |
| T4d | Inflammatory carcinoma |
| N | Regional Lymph Nodes |
| pNx | Regional lymph nodes cannot be assessed |
| pN0 | No regional lymph node metastasis identified histologically |
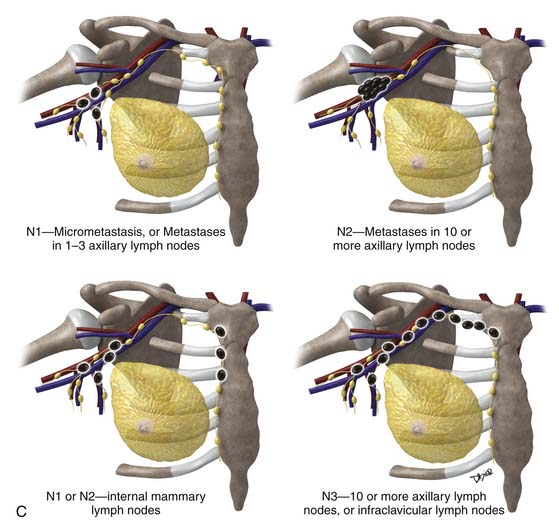
| pN1 | Micrometastasis, or Metastases in one to three axillary lymph nodes with or without internal mammary nodes |
| pN2 | Metastases in 10 or more axillary lymph nodes, or Infraclavicular lymph nodes (level III), or Clinically detected internal mammary lymph nodes, or More than three axillary lymph nodes and internal mammary nodes, or Ipsilateral supraclavicular lymph nodes |
| pN3 | Metastases in 10 or more axillary lymph nodes, or Metastases to infraclavicular lymph nodes (level III) |
| M | Distant Metastases |
| M0 | No distant metastases |
| cM0 (i+) | Circulating tumor cells or microscopic tumor cells in bone marrow No clinical or radiologic distant metastasis |
| M1 | Distant metastases |
DCIS, ductal carcinoma in situ; LCIS, lobular carcinoma in situ.
Table 27-2 American Joint Committee on Cancer Stage Grouping System Classification for Breast Cancer
| STAGE GROUPING | |
|---|---|
| Stage 0 | Tis N0 M0 |
| Stage I | T1* N0 M0 |
| Stage IIA | T0 N1 M0 T1* N1 M0 T2 N0 M0 |
| Stage IIB | T2 N1 M0 T3 N0 M0 |
| Stage IIIA | T0 N2 M0 T1* N2 M0 T2 N2 M0 T3 N1 M0 T3 N2 M0 |
| Stage IIIB | T4 N0 M0 T4 N1 M0 T4 N2 M0 |
| Stage IIIC | Any T N3 M0 |
| Stage IV | Any T Any N M1 |
Tis, in situ; T1.
From Edge SB, Byrd DR, Compton CC, et al. Breast. AJCC Cancer Staging Manual. 7th ed. New York: Springer; 2010:347-369.
Patterns of Tumor Spread
Local recurrence, regional recurrence, and distant metastases occur when cancer cells remain after treatment or spread beyond the breast and axillary lymph nodes; the spread of cancer cells represents a major source of morbidity in breast cancer patients. Breast cancer cells can spread via the lymphatic system to the regional lymph nodes, involving the low axillary lymph nodes (level I) first, followed by the mid- (level II) and high axillary lymph nodes (level III). Level I lymph nodes are lateral to the lateral border of the pectoralis minor muscle. Level II lymph nodes are between the medial and the lateral borders of the pectoralis minor muscle, as well as the interpectoral lymph nodes (Rotter’s nodes). Level III lymph nodes are nodes medial to the medial margin of the pectoralis minor muscle and inferior to the clavicle. Therefore, the standard approach to staging lymph nodes via axillary nodal dissection often involves removal of the low and midaxillary lymph nodes. Involvement of the internal mammary chain is observed in approximately 20% of patients, often with large and deep medially located breast cancer, and tends to be associated with extensive metastasis.
Tumor cells may spread to distant sites via the lymphatic or circulatory system; the four major sites of distant metastasis for breast cancer are bone, lung, brain, and liver. Bone is the most common site of metastasis from most subtypes of breast cancer,39 and the presence of tumor cells in the bone marrow is a strong predictor for distant metastases. Cutaneous metastasis is not common, but breast carcinoma is the most common primary malignancy to spread to the skin and accounts for 24% of all cutaneous metastases.40
Imaging
Several modalities available to image the breasts include mammography, ultrasonography, ductography, and MRI. Once a woman has been diagnosed with breast cancer, cross-sectional imaging with computed tomography (CT), positron-emission tomography (PET), MRI, or bone scintigraphy are often indicated to image the rest of the body, especially when there are suspicious findings on the patient’s history or physical examination or if serologic tests for liver or bone function show elevated serum levels. Most often, using a combination of imaging modalities is crucial for the accurate staging of breast cancer. Here, an overview of the role of imaging in the diagnosis and staging of this common disease is presented.
Primary Tumor
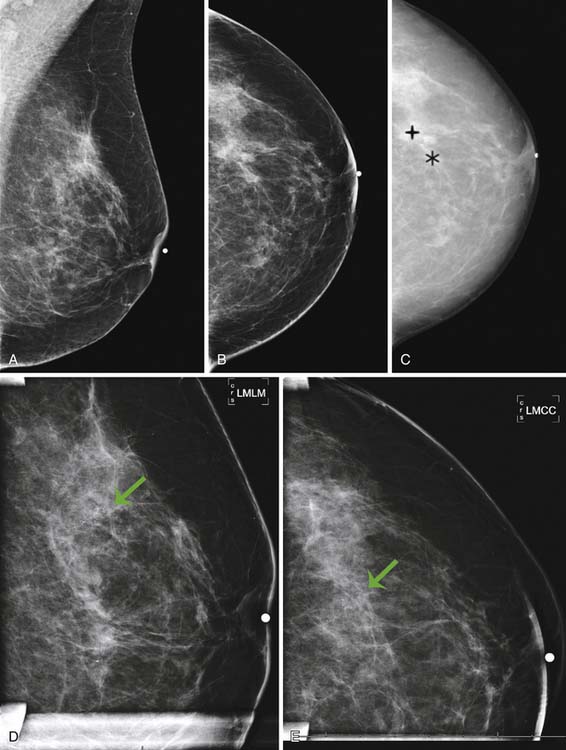
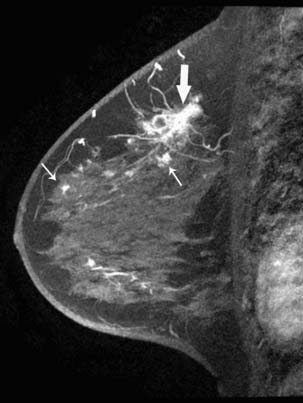
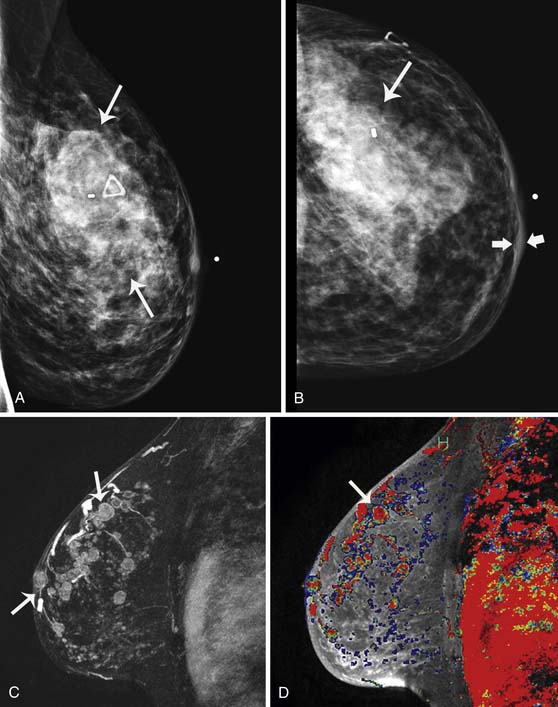
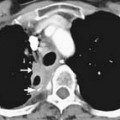
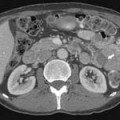
Breast imaging is used to detect primary or recurrent breast cancer, determine the extent of disease in the affected breast, evaluate regional lymph nodes, and identify distant metastatic disease. Cancer within the affected breast may be described as unifocal (Figure 27-5), multifocal (two or more lesions in the same quadrant [Figure 27-6]), or multicentric (lesions in two or more quadrants [Figure 27-7]). Currently, mammography, ultrasonography, and MRI are the primary modalities indicated in the detection and staging of breast cancer.
Mammography
Diagnostic mammography or problem-solving mammography is performed when a patient presents with a palpable finding or when a suspicious finding is detected with screening mammography. A radiopaque BB or skin marker is placed directly over the suspicious region before the mammogram is obtained. In addition to the mediolateral oblique and craniocaudal views, described earlier, additional views may be indicated to properly evaluate the abnormality. A 90-degree lateral view can assist in triangulating the abnormality. Spot compression views in craniocaudal and lateral projections are incorporated to evaluate a possible mass, asymmetrical density, or superimposition of normal breast parenchyma. Magnification views, commonly performed in craniocaudal and lateral projections, facilitate characterization of microcalcifications. Slowly, conventional analog technology is being replaced by digital mammography. However, standard mammography projections remain unchanged. With advances in digital acquisition and processing technology, digital tomosynthesis is being incorporated into mammography systems and may provide a solution to the problem of distinguishing overlapping structures in the breast, increasing the sensitivity and specificity of mammography for cancer detection and diagnosis.41 Clinical trials are currently under way to establish the efficacy of digital breast tomosynthesis and to define its role in future practice.
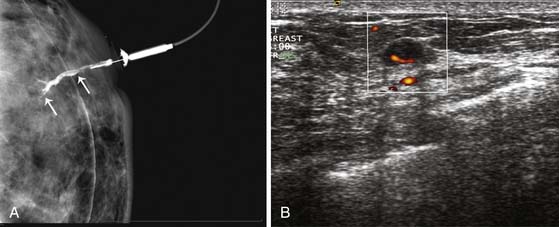
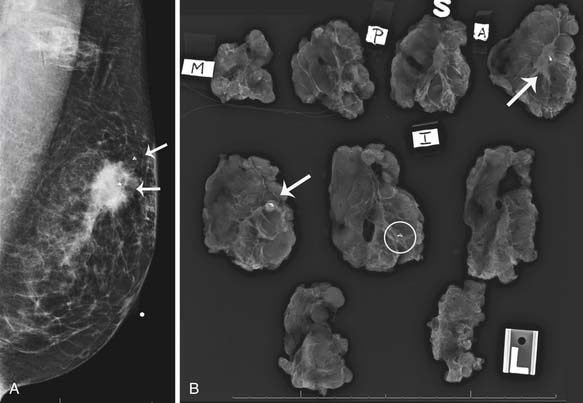
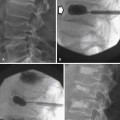
Mammography is also used to guide breast interventions, including a stereotactic-guided core biopsy using vacuum-assisted devices and an image-guided needle localization of a lesion prior to surgery. Immediately after resection of the targeted lesion, the biopsy or resected specimen can be imaged while the patient is still under anesthesia in order to verify the margins. This “specimen radiography” provides additional information in patients who had a complete response to preoperative chemotherapy because the only image-detectable finding is often the biopsy clip placed at the time of the initial biopsy or residual calcifications (Figure 27-8).