Tremendous advances in understanding of the circulatory system stemmed from the groundbreaking work of Claude Bernard, a French physiologist who performed cardiac catheterization of an equine heart in 1844. However, only with Wilhelm Konrad Röentgen’s revolutionary discovery of x-rays in Germany in 1895 did direct imaging of the heart in vivo became feasible. The first human in vivo cardiac catheterization was performed in 1929 by the German urologist Werner Forssmann, who put a urethral catheter into the right cardiac chambers through his own antecubital vein. Subsequently, the era of percutaneous invasive cardiology was introduced by Andreas Gruentzig in 1977.
In the clinical setting, coronary angiography is performed to visualize coronary endoluminal vascular anatomy in assessing for the presence (and burden) or absence of coronary artery disease. Information about the luminal obstructive pattern and collateral pattern can also be obtained. Conventional invasive coronary angiography remains the standard procedure in the evaluation of anatomic coronary disease, although it has limitations. In vivo structural and physiologic evaluations such as intravascular ultrasound (IVUS) and fractional flow reserve have expanded the clinical usefulness of invasive diagnostic cardiac catheterization.
This chapter provides a clinically oriented review of coronary anatomy as assessed with cardiac angiography. Normal coronary anatomy as assessed with conventional coronary angiography is reviewed, as are common coronary artery anomalies.
Normal Coronary Anatomy
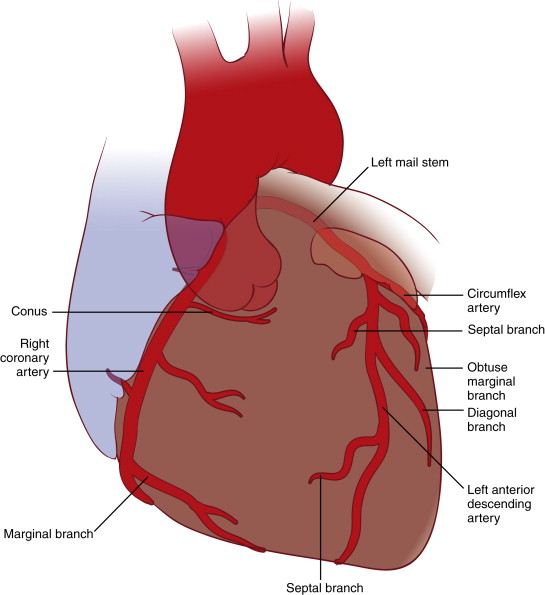
Two coronary ostia arise from the proximal aorta; the left main coronary artery (LMCA) comes out of the left coronary sinus, and the right coronary artery (RCA) originates from the right coronary sinus, located slightly lower than the LMCA ostium. The LMCA is further divided into the left anterior descending (LAD) coronary artery and the left circumflex (LCx) coronary artery. The LMCA, LAD, and LCx arteries and their branches comprise the left coronary system, and the RCA belongs to the right coronary system ( Fig. 14-1 ). Depending on the vessel sizes and collateral vessels, the posterior (inferior wall) side of the heart can be supplied by different arteries. Hence, the coronary blood flow system can be referred to as either left dominant (both the posterior descending and posterior left ventricular branches originate from the LCx artery; 15% of cases) or right dominant (both branches take off from the RCA; 60% to 85% of cases). In addition, codominance (balanced distribution) of the arteries (the posterior descending branch comes from the RCA, and the posterior left ventricular branch derives from the LCx artery) is found in less than 20% of cases. Normally, the arteries run through grooves of the epicardial surface of the heart.
Common Views for Coronary Angiography
Horizontally, the x-ray plane can be positioned in the midline (anteroposterior [AP] view), from the patient’s left side (left anterior oblique [LAO] view) or right side (right anterior oblique [RAO] view). Perpendicular angles are either cranial (downward view from the head of the patient) or caudal (upward view from the foot of the patient) ( Fig. 14-2 ). No fixed sequence of order in visualization is used because each patient may have different indications for coronary angiography. However, the angiographer usually begins with an evaluation of the left coronary system and then proceeds to right coronary angiography. When left heart catheterization is indicated (i.e., to assess left ventricular end-diastolic pressure), this should be carried out before contrast administration.

The following practical angiography sequence is routinely used:
- 1.
RAO caudal for the LMCA, proximal LAD artery, and LCx artery
- 2.
RAO cranial for the middle and distal portions of the LAD artery
- 3.
LAO cranial for the middle and distal portions in an orthogonal position
- 4.
LAO caudal (“spider view”) for the LMCA, proximal LAD artery, and LCx artery
- 5.
LAO caudal for the proximal RCA
- 6.
AP or LAO cranial for the distal RCA, posterior descending artery (PDA), and posterior left ventricular branch
Table 14-1 summarizes commonly used views for each coronary artery segment. However, additional views can be obtained in case of overlapping or unclear images.
| Coronary Artery Branches and Segments | At Origin | In the Course |
|---|---|---|
| LMCA | AP cranial, RAO caudal, LAO cranial, LAO caudal | AP cranial, LAO caudal |
| Proximal LAD | LAO cranial, RAO cranial, AP caudal | LAO cranial, LAO caudal, RAO cranial |
| Mid-LAD | LAO cranial, RAO cranial | LAO cranial, LAO caudal, RAO cranial |
| Distal LAD | AP cranial, RAO cranial | LAO cranial, LAO caudal, RAO cranial |
| Diagonal | LAO cranial, RAO cranial | RAO cranial |
| Proximal LCx | RAO caudal, LAO caudal | LAO caudal |
| OM | RAO caudal, LAO caudal | RAO caudal |
| Proximal RCA | LAO | LAO, right lateral |
| Mid-RCA | RAO, LAO | LAO, right lateral |
| Distal RCA | LAO cranial | LAO cranial |
| PDA | LAO cranial | RAO |
| Posterior left ventricular | RAO cranial | RAO cranial |
Segmental Nomenclature of the Coronary Artery System
The Coronary Artery Surgery Study (CASS) classification system of the coronary artery tree was developed by the CASS, Thrombolysis in Myocardial Infarction (TIMI), and Bypass Angioplasty Revascularization Investigation (BARI) study groups in 1992. The entire coronary arterial network is divided into 29 anatomic segments. Figure 14-3 illustrates three coronary arteries and their major branches. Numbers 1 through 8 denote the RCA and its branches. The LAD and LCx arteries are detailed in numbers 12 to 17, and 18 to 23, respectively.

Assessment of Coronary Artery Blood Flow
One of the most widely used methods to quantify the coronary angiographic blood flow is the TIMI flow grading system developed by the TIMI group in 1987. Although the trial itself was designed to compare the efficacy of intravenous streptokinase and recombinant-plasminogen activator in achieving reperfusion, this classification was found to be useful in quantifying the flow of diseased coronary arteries ( Table 14-2 ).
| Grade 0 (no perfusion) | No antegrade flow occurs beyond the point of occlusion. |
| Grade 1 (penetration with minimal perfusion) | The contrast material passes beyond the area of obstruction, but it “hangs up” and fails to opacify the entire coronary bed distal to the obstruction for duration of the cine run. |
| Grade 2 (partial perfusion) | The contrast material passes across the obstruction and opacifies the coronary bed distal to the obstruction. However, the rate of entry of contrast material into the vessel distal to the obstruction or its rate of clearance from the distal bed (or both) is perceptibly slower than its entry into or clearance from comparable areas not perfused by the previously occluded vessel (e.g., the opposite coronary artery or the coronary bed proximal to the obstruction). |
| Grade 3 (complete perfusion) | Antegrade flow into the bed distal to the obstruction occurs as promptly as antegrade flow into the bed proximal to the obstruction, and clearance of contrast material from the involved bed is as rapid as clearance from an uninvolved bed in the same vessel or the opposite artery. |
The complexity of coronary artery disease can be assessed using the SYNTAX score, which was developed in an effort to provide an evidence base for determination of best treatment options. An online calculator and tutorial with a detailed description can be found online at www.syntaxscore.com .
Problem Solving: Combining Cardiac Angiography with other Imaging Modalities
For the most part, coronary angiography is limited to detailing large epicardial artery blood flow. Assessing myocardial perfusion defects (ischemia or infarction) with other noninvasive imaging modalities plays a crucial role in guiding revascularization therapy. The American Heart Association suggested a 17-segment system of regional wall distribution map for 2-dimensional echocardiography ( Fig. 14-4 , A ) and for single photon emission computed tomography (SPECT) nuclear cardiology study (see Fig. 14-4 , B ).


Problem Solving: Angiographic Visualization of Individual Coronary Artery and its Branches
Because the coronary arteries form a globelike three-dimensional structure around the myocardium, each segment must be visualized at a specific angle. Moreover, because of the eccentricity of the obstruction, a different angle is often required to visualize the same segment of artery to obtain a clearer picture. Orthogonal views are desired to exclude significant stenosis, given the potential limitation of underestimation of eccentric stenoses and foreshortening of vessels.
Left Main Coronary Artery
The LMCA originates from the left coronary sinus. Studies showed that the average length of the LMCA measures approximately 3 to 6 mm, with a wide variation of 0 to 15 mm. The average diameter of the LMCA is approximately 4.5 mm, but it can vary from 3 to 6 mm, depending on gender and ethnicity. Table 14-3 shows anatomic variations in coronary artery diameter by gender. Several studies showed that the LMCA diameter is 75% to 91% smaller in women than in men. The LMCA runs behind the right ventricular outflow tract (RVOT), between the pulmonary trunk and the left atrium. At the distal end, it bifurcates to the LAD and LCx arteries. In approximately 30% of the population, the intermediate branch (ramus intermedius) exists between the LAD and LCx arteries. This intermediate artery usually follows the course of a high obtuse marginal (OM) branch of the LCx artery.
| Dodge et al | MacAlpin et al | Others | |
|---|---|---|---|
| LMCA (mm) | 4.5 ± 0.5 (men) 3.9 ± 0.4 (women) | 4.3 ± 0.6 (men) 3.5 ± 0.7 (women) | |
| Proximal LAD (mm) | 3.6 ± 0.5 (men) 3.2 ± 0.5 (women) | 3.5 ± 0.5 (men) 2.9 ± 0.4 (women) | 3.3 ± 0.5 (Kimball et al) |
| Mid-LAD (mm) | 2.3 ± 0.4 (men) 2.2 ± 0.5 (women) | 2.0 ± 0.4 (men) 1.8 ± 0.2 (women) | |
| Distal LAD (mm) | 1.1 ± 0.4 (men) 0.9 ± 0.3 (women) | ||
| Proximal LCx (mm) ∗ | 3.4 ± 0.5 (men) 2.9 ± 0.6 (women) | 3.1 ± 0.7 (men) 2.6 ± 0.6 (women) | |
| Proximal RCA (mm) † | 3.9 ± 0.6 (men) 3.3 ± 0.6 (women) | 3.4 ± 0.7 (men) 3.0 ± 0.5 (women) |
∗ Measured from the segment proximal to the first obtuse marginal branch.
† Measured from the segment proximal to the first acute marginal branch.
Standard views for the LMCA include the RAO caudal and AP views ( Fig. 14-5 , A and B ), in addition to the LAO caudal view. The LAO caudal view is very useful for separating the distal LMCA in cases of overlapping vessels (see Fig. 14-5 , C ).

Left Anterior Descending Coronary Artery
The LAD artery arises from the LMCA, crosses beneath the left atrial appendage, and travels down the anterior interventricular sulcus to feed the majority of the anterior wall of the left ventricle and ventricular septum. The average diameter of the proximal LAD artery is 3.5 to 3.6 mm in men and 2.9 to 3.2 mm in women. In general, women tend to have a 15% to 20% smaller diameter compared with men. The diameter of the LAD artery tapers gradually as it runs distally (see Table 14-3 ). Approximately 80% to 90% of population will exhibit left coronary dominance wherein the LAD artery wraps around the apex and runs in the inferior interventricular groove on the back of the heart ( Fig. 14-6 , A ). In contrast, only 40% to 50% of the LAD artery goes beyond the apex in case of right dominance because the RCA supplies the apical territory with its PDA branch (see Fig. 14-6 , B ).

Along the course of the LAD artery, side branches of two major types can be seen: the diagonal (D) and septal (S) (or perforating) branches ( Fig. 14-7 ). In general, the diagonal branches are larger, longer, and have more angular takeoff from the LAD artery. Diagonal arteries run across between the interventricular and AV grooves and supply the anterior left ventricular free wall, the anterolateral mitral papillary muscle, and part of the anterior right ventricular wall. The septal branches are usually smaller, shorter, and form almost perpendicular angles to the LAD artery. These perforating branches enter the septum and deliver blood to the apical and anterior two thirds of the interventricular septum. The first septal perforating branch perfuses the His bundle and the proximal left bundle branch.