div class=”ChapterContextInformation”>
6. Advanced Carotid Vessel Wall Imaging and Future Directions
Keywords
Carotid arteryAtherosclerosisMagnetic resonance imagingPlaque ruptureBlack-bloodMulti-contrastIschemic strokeCarotid endarterectomyCarotid artery stentingCryptogenic strokeIntroduction
Within the circulatory system, the extracranial carotid artery bifurcation is one of the most common sites for developing atherosclerotic plaques. In an imaging study of middle-aged males and females, carotid atherosclerosis was seen in 31% of the participants, which was more common than atherosclerosis in the aorta or coronary arteries and only behind iliofemoral atherosclerosis [1]. Most individuals with carotid atherosclerosis are not aware of its presence as the disease is often asymptomatic, even when there is already substantial luminal stenosis. However, prognostic evaluation of carotid plaque in high-risk individuals is clinically significant given its potential pathophysiological consequences, including intraluminal thrombosis, artery-to-artery thromboembolism, and in the late stage, severe luminal stenosis, all of which may lead to brain infarction and/or hypoperfusion. Because of the high prevalence of carotid atherosclerosis in the general population, carotid artery disease is a major source for neurological ischemic events, accountable for about 41,000 ischemic stroke incidences annually in the USA alone [2]. The burden of asymptomatic brain infarctions and/or hypoperfusion is often less recognized but expected to be >10-fold higher than that of ischemic strokes, which is increasingly recognized as a major contributor to cognitive impairment and dementia [3]. Furthermore, as a local manifestation of systemic atherosclerosis, carotid atherosclerosis is often evaluated for improving cardiovascular risk assessment and monitoring response to medical treatment, given that carotid lesions are less challenging to image and characterize compared to atherosclerotic lesions in other deep-seated, small-caliber arteries.
In contrast to luminal imaging techniques that grade the severity of carotid atherosclerosis according to its impact on lumen diameter or area, vessel wall imaging with magnetic resonance imaging (MRI) allows for direct visualization of carotid plaque morphology and tissue composition, as well as pathological activities at the cellular level when contrast agents are used. MRI of carotid atherosclerosis has been rapidly evolving in the past two decades. Images with diagnostic image quality at 0.6–0.8 mm spatial resolution can now be achieved on all major platforms by modifying commercially available pulse sequences. This wave of technical development in the past two decades has been primarily driven by an unmet clinical need in the selection of patients with carotid disease for carotid revascularization. Since the early 1990s, several landmark clinical trials evaluated the efficacy of carotid endarterectomy (CEA) in a broad spectrum of patient populations. The North American Symptomatic Carotid Endarterectomy Trial (NASCET) compared CEA to medical treatment alone in patients with transient ischemic attack or nondisabling ischemic stroke within the previous 6 months. There was clear evidence of a net benefit in patients with high-grade stenosis (70–99%) but not in patients with moderate or mild stenosis [4]. The number needed to treat to prevent 1 ischemic stroke in 5 years was 6 and 15 in the groups with high and moderate stenosis, respectively. Nonetheless, the risk under medical therapy alone in patients with <70% stenosis was still considerable at about 20% over 5 years in the mild-to-moderate stenosis group. Compared to studies on symptomatic patients, clinical trials that evaluated the efficacy of CEA in asymptomatic patients documented a more moderate benefit using stenosis severity to guide patient selection [5]. These clinical trials highlighted the limitations of luminal stenosis-based carotid disease risk assessment. Two decades later, it has become more challenging to decide on the best treatment for individual patients based on luminal stenosis due to the changing landscape in clinical management of carotid artery disease, including improvements in optimal medical therapy, reduced perioperative complication rates of carotid revascularization, and new approaches to carotid artery stenting (CAS). Since carotid vessel wall imaging may allow for more precise risk estimation than luminal stenosis assessment by characterizing key pathophysiological processes, it has potential to revise existing guidelines on indications for carotid revascularization.
Evidence on the Capabilities of Carotid Vessel Wall Imaging
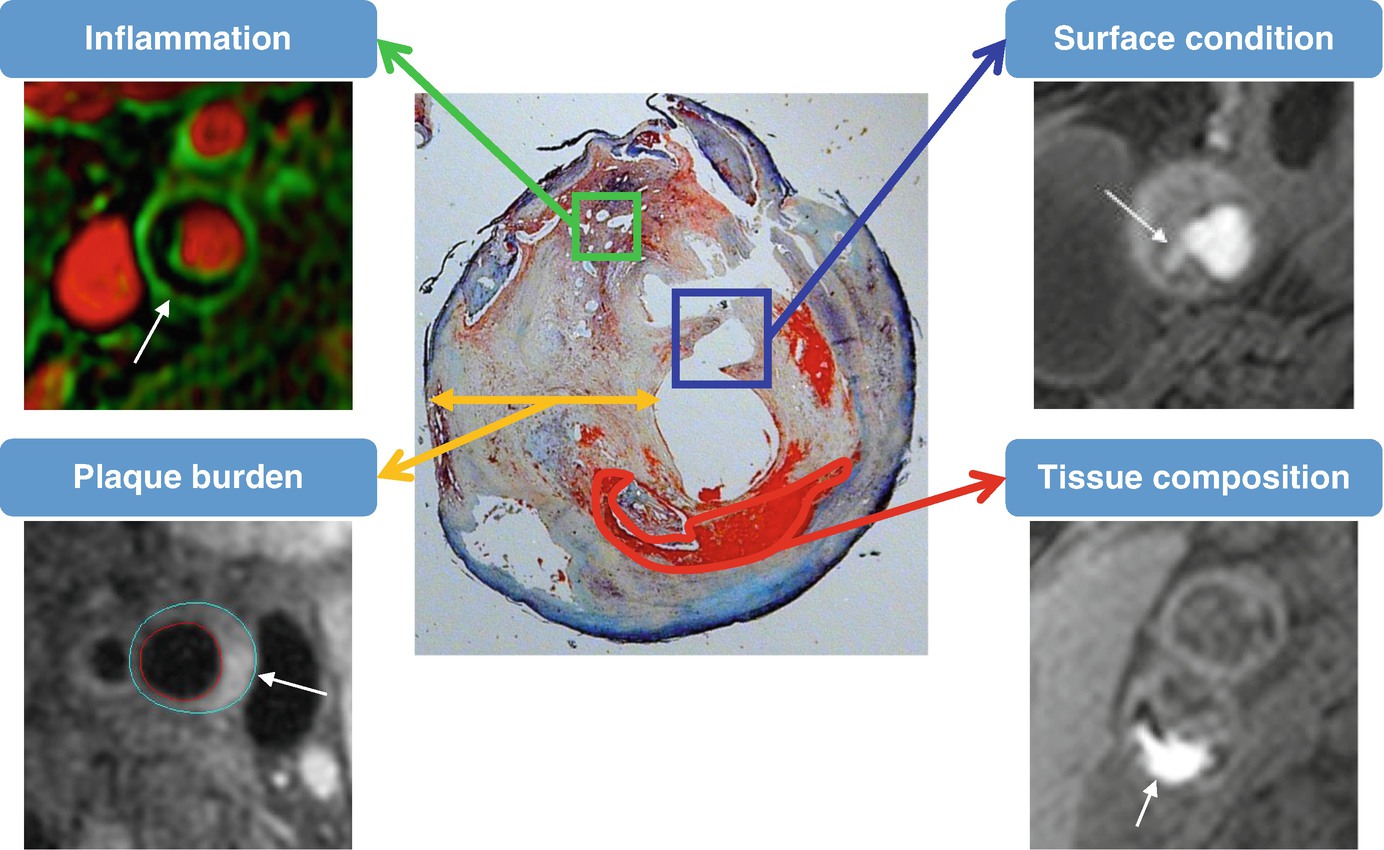
Carotid plaque morphology: accessible information with MRI. Carotid plaques are heterogeneous at various levels. With different protocols, MRI allows us to noninvasively evaluate carotid plaque burden, tissue composition, surface condition, and inflammatory activities
Plaque Burden
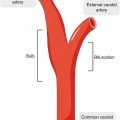
The carotid vessel wall expands at the origin of the internal carotid artery to form the carotid bulb, where atherosclerosis tends to build up due to the presence of low wall shear stress. During plaque development, there can also be various degrees of expansive remodeling of the vessel wall [10]. As such, a significant amount of atherosclerosis could already exist before there is appreciable luminal stenosis. Large plaque burden is known to be associated with increased risk for plaque rupture [11]. As a marker of systemic atherosclerosis, carotid plaque burden was strongly associated with coronary calcium score and may improve the prediction of cardiovascular events beyond conventional risk factors [12, 13].
Accurate measurement of carotid plaque burden requires clear delineation of lumen and outer wall boundaries. High spatial resolution is necessary to minimize partial volume averaging [14]. Duivenvoorden et al. [15] compared different in-plane resolutions of carotid MRI varying from 0.65 mm to 0.20 mm at 3 mm slice thickness performed on the same subjects. Mean wall area measured on images with lower resolution was larger than those measured on images with higher resolution. When a phased-array surface coil is available, images are typically acquired with 2 mm slice thickness and 0.5–0.6 mm in-plane resolution in 2D carotid MRI and with 0.6–0.8 mm isotropic resolution in 3D carotid MRI [16–18], which have been shown to provide good signal-to-noise within clinically acceptable scan times. In addition to high spatial resolution, sufficient blood and fat suppression are also essential in achieving diagnostic image quality. Blood suppression in cardiovascular MR can be implemented using inflow saturation, double inversion recovery (DIR), or motion sensitization. As the flow pattern near the carotid artery bifurcation can be very complex, inflow saturation is rarely adequate. DIR is flow direction dependent and often used in 2D carotid MRI. However, flow artifacts may still arise from recirculating flow which has not outflowed from the imaging slice at the second inversion pulse. Motion-sensitized driven equilibrium (MSDE) preparation has been shown to provide more complete flow suppression compared to inflow saturation and DIR [19]. MSDE preparation allows flow direction-independent blood suppression in a large imaging slab and, thus, has been frequently used in 3D carotid MRI.
Mean/maximum wall area and mean/maximum wall thickness are common measurements of carotid plaque burden, which are usually obtained by segmenting lumen and total vessel area on cross-sectional images. Multiple studies have evaluated the precision and accuracy of these measurements by carotid MRI. Despite minor methodological differences, previous studies consistently showed excellent inter-observer and inter-scan reproducibility of carotid wall measurements [16, 20–22]. The coefficient of variation for repeated scans, an indicator of measurement errors, was <10% for most carotid wall measurements, which was smaller compared to similar measurements by B-mode ultrasound [15]. Of note, measurement precision is influenced by image quality and spatial resolution. At the same image quality, larger carotid plaques are expected to be associated with smaller measurement errors, which may have implications for planning pharmaceutical trials using imaging endpoints [23]. Furthermore, wall area measurements typically have smaller measurement errors than wall thickness measurements.
The accuracy of MRI measurements of carotid plaque burden has also been established. In patients who underwent CEA, Luo et al. [24] demonstrated a high agreement between in vivo MRI measurements of carotid plaque burden and corresponding ex vivo MRI measurements (Pearson’s correlation coefficient >0.90). The correlations between minimum lumen area, maximum wall area, and wall volume were weak, suggesting that these measurements provide different information of carotid plaque burden.
Plaque Tissue Composition
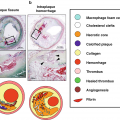
Plaque tissue composition plays an essential role in the pathogenesis of atherothrombotic complications. Ruptured plaques frequently harbor large necrotic cores, intraplaque hemorrhages, and speckle calcifications [25]. As substantial heterogeneity in tissue composition exists among carotid plaques with the same level of luminal stenosis, it is expected that noninvasive imaging of carotid plaque composition would allow for improved prediction of clinical risk. Common plaque components, besides fibrous tissue, include extracellular lipids, necrotic tissue, calcification, and intraplaque hemorrhage. MRI is unique for imaging plaque composition in that it allows differentiation between all common plaque components. Indeed, its capability of accurately classifying all common plaque components has led to multiple studies demonstrating that carotid MRI is able to not only detect lipid core presence but also quantitatively measure lipid core area or percent area [26, 27]. Furthermore, MRI is currently considered the only imaging modality that is capable of detecting intraplaque hemorrhage, a characteristic feature of high-risk carotid plaque.
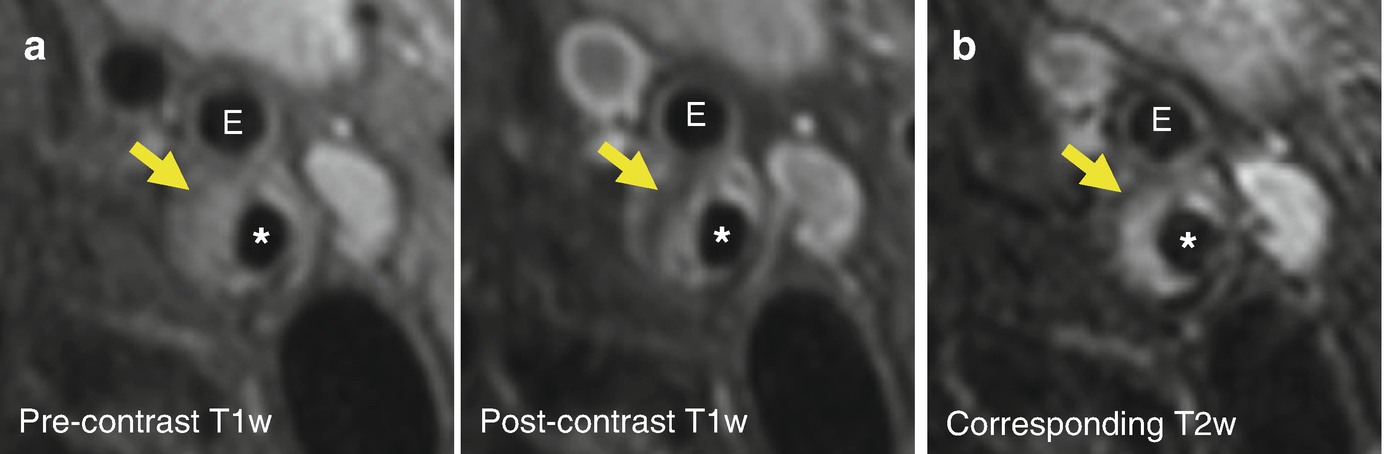
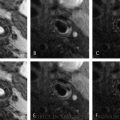
Lipid-rich necrotic core detection by combining pre- and post-contrast images. (a) Because of presence of neovasculature and expanded extravascular space, fibrous tissue in carotid plaque enhances after gadolinium contrast injection. The lipid-rich necrotic core (arrow) is detected as areas with no or little enhancement on post-contrast T1w-TSE images when compared with pre-contrast T1w-TSE. (b) Cholesterols and cholesterol esters constitute the main lipid content of carotid plaque, which shorten T2 of water molecules. Thus, the lipid-rich necrotic core is detected as hypointense areas on T2w-TSE, which is used as an alternative when post-contrast images are not available. ∗indicates lumen of internal carotid artery; E, external carotid artery; T1w-TSE, T1-weighted turbo spin echo; T2w-TSE, T2-weighted turbo spin echo
Intraplaque hemorrhage is a common finding in advanced carotid plaques. The degradation of red blood cells produces methemoglobin, which reduces the T1 relaxation time of surrounding water molecules and generates a hyperintense signal on T1-weighted images. Moody et al. [34] studied 63 patients scheduled for CEA using a heavily T1-weighted magnetization-prepared 3D gradient echo (MP-RAGE) sequence. T1 hyperintensity was observed in 40 (63.5%) cases, with an 84% sensitivity and 84% specificity for intraplaque hemorrhage seen on histology. Several other studies have since evaluated the performance of heavily T1-weighted MP-RAGE sequence with optimized contrast and/or improved spatial resolution for detecting intraplaque hemorrhage [35–37]. Notably, Ota et al. [38] showed that MP-RAGE provided better tissue contrast between intraplaque hemorrhage and background tissue and achieved a higher agreement with histological analysis when compared to T1-weighted fast spin echo and TOF. When MP-RAGE is not available, the source images of the pre-contrast scan in contrast-enhanced MR angiography and TOF images can be used to detect intraplaque hemorrhage [39], which may be particularly handy in the clinical setting.
Luminal Surface Condition
The typical vulnerable plaque in the coronary artery is described as thin-cap fibroatheroma. Virmani et al. [25] measured fibrous cap thickness of ruptured coronary plaques and found that mean cap thickness at the site of rupture was 23 ± 19 um, with 95% of the cases measuring <65 um. Therefore, the fibrous cap overlying the necrotic core is considered thin in coronary arteries if it is <65 um in histology. Compared to coronary plaques, carotid plaques can rupture when the fibrous cap is thicker or less infiltrated by inflammatory cells, which is likely explained by the different hemodynamic conditions between carotid and coronary arteries. Redgrave et al. [40] studied fibrous cap thickness in 428 CEA plaques. The median minimum cap thickness was 150 um (interquartile range: 80, 210), and the median representative cap thickness was 300 um (interquartile range: 200, 500). The optimal cutoff values for discriminating ruptured from nonruptured plaques were minimum cap thickness <200 um and representative cap thickness <500 um. Further, fibrous cap rupture leads to fissure or ulceration, which are important features to identify as they increase the ipsilateral risk of cerebral embolism and the risk of cardiovascular events in general [41]. Another luminal surface condition that is of particular relevance is calcified nodule, which is seen more frequently in carotid plaques than in coronary plaques [42].
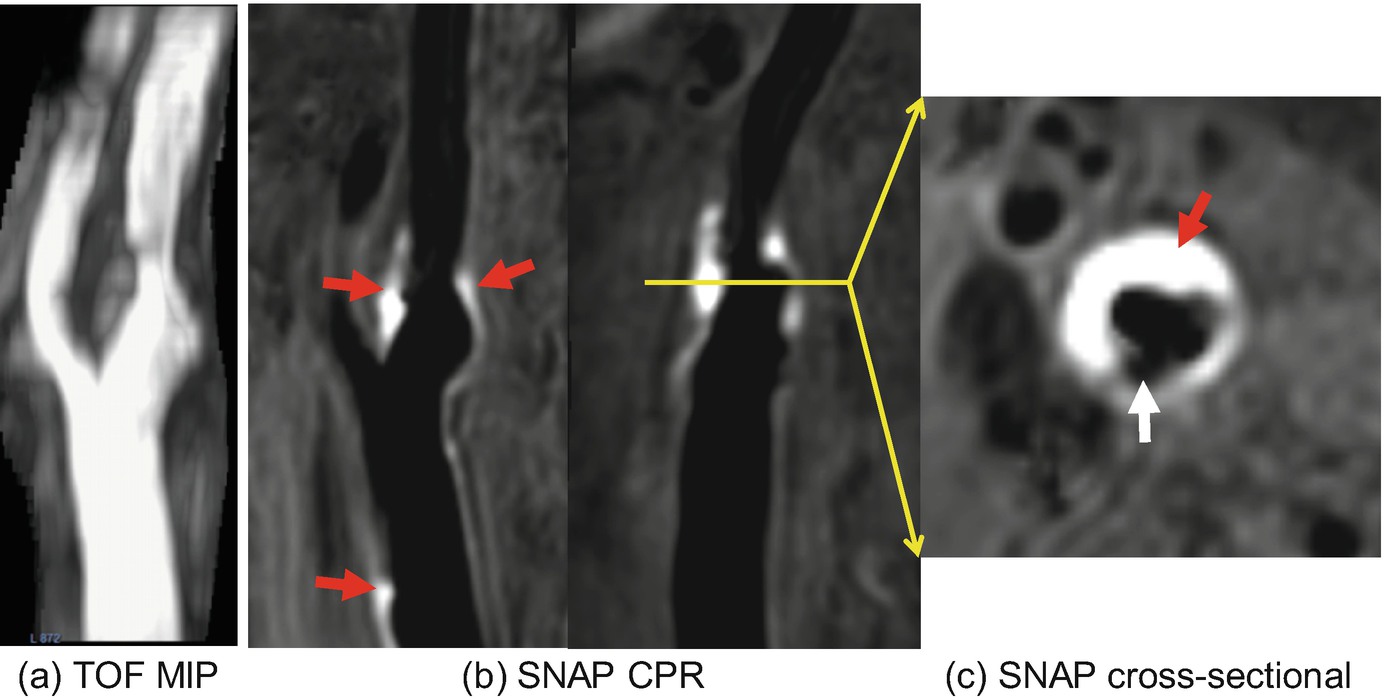
Characterizing the fibrous cap has typically relied on endovascular imaging techniques such as optical coherence tomography. Because of limited spatial resolution of carotid MRI, it is challenging to directly measure the thickness of fibrous cap. Nonetheless, it is possible to classify carotid plaques into those with thick, thin, or ruptured fibrous cap based on carotid MRI findings. Thick fibrous cap appears as a dark band between white lumen and gray wall on 3D TOF [43]. On T2-weighted or contrast-enhanced images, fibrous cap is distinguishable from the underlying necrotic core and considered to be thick if it is readily visible. On the other hand, absence of the dark band between the white lumen and gray wall on 3D TOF and lack of fibrous cap signal on T2-weighted or contrast-enhanced images indicate the presence of thin fibrous cap. Fibrous cap rupture is considered if there is juxtaluminal hyperintense signal on 3D TOF, which indicates the presence of fissure or ulceration. In a study of 22 patients undergoing CEA, classification of fibrous cap by 3D TOF showed a high agreement with histological findings (89% agreement; kappa, 0.83 [95%CI, 0.67, 1.0]) [43]. Calcified nodule appears hypointense on all MRI contrast weightings and protrudes into the lumen. As calcified nodules may be indistinguishable from carotid lumen on black-blood images, its detection usually requires comparing lumen morphology between bright- and black-blood images.
Plaque Inflammation and Neovasculature
Atherosclerosis has been recognized as an inflammatory disease. Although most studies focused on coronary atherosclerosis or animal models, there is also clear evidence on the role of inflammation in carotid atherosclerosis progression. T lymphocytes and macrophages were abundant in symptomatic carotid plaques and closely associated with plaque rupture [6]. Redgrave et al. [7] and Peeters et al. [44] found that inflammatory cells and proinflammatory cytokines tended to decrease with time after ischemic stroke but persisted after transient ischemic attack, while smooth muscle cell content increased with time after ischemic stroke, suggesting a possible healing process after ischemic stroke but not after transient ischemic attack. Plaque neovascularization has been described in the early stages of plaque development and may alleviate hypoxia as the carotid wall thickens [45, 46]. However, it is likely a major contributor to plaque progression and rupture given its associations with intraplaque hemorrhage and plaque inflammation [47–49]. Electron microscopy studies revealed the compromised structural integrity and leucocyte infiltration of intraplaque microvessels [50], which supports plaque neovasculature as a route for intraplaque hemorrhage and plaque inflammation.
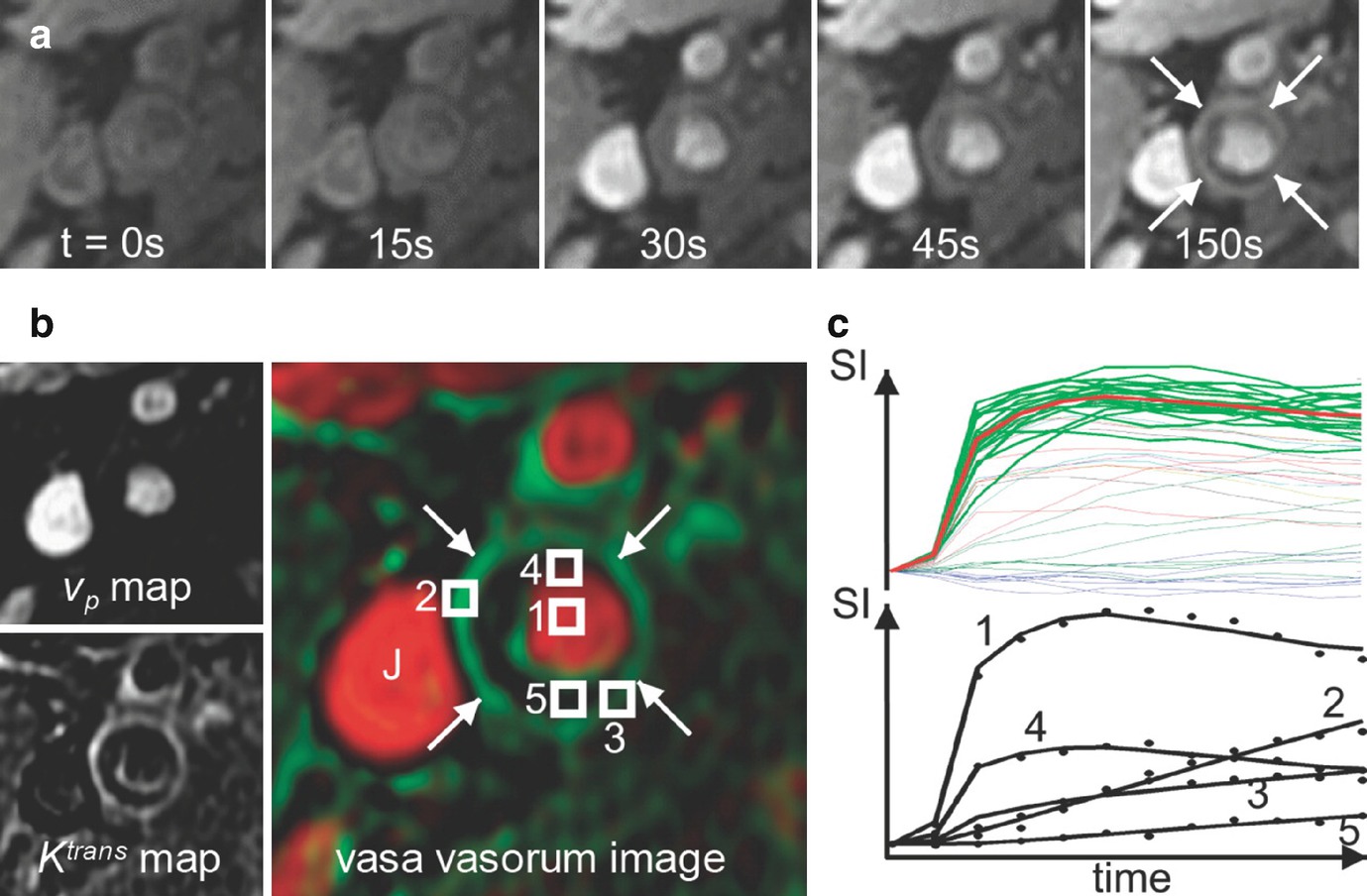
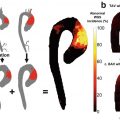
Dynamic contrast-enhanced MRI for probing plaque inflammation and neovasculature. (a) Original DCE-MR images of different time points show the dynamic enhancement process of a carotid plaque (arrows). (b) Pharmacokinetic modeling generates Ktrans and vp maps, which are fused in the color-coded vasa vasorum image. J, jugular vein. (c) Top graph shows signal-intensity-versus-time curves for all pixels in the 2 cm2 region for the set of blood curves extracted by the clustering algorithm (green lines) and their average (red line). Bottom graph shows typical fitting results for the kinetic model with corresponding points in (b), including a pixel in the lumen (1), adventitial pixels with high (2) and low (3) Ktrans, a pixel with partial volume of the lumen (4), and an interior plaque pixel (5). (Adapted with permission from Kerwin et al. [55])
Another approach to plaque inflammation is to use ultrasmall superparamagnetic iron oxide particles (USPIOs) as MRI contrast agent to directly image macrophages. Kooi et al. [62] studied the potential of USPIO-enhanced MRI for measuring plaque inflammation in 11 patients undergoing CEA. After at least 24 hours post USPIO injection, histological and electron microscopical analyses of plaques demonstrated that USPIOs were primarily found in plaque macrophages and could lead to signal changes on T2∗-weighted images. Trivedi et al. [63] found that 24–36 hours post USPIO injection was the optimal time window for detecting signal changes, with which USPIO-enhanced MRI may be a sensitive way to detect carotid plaque inflammation [64].
Evidence on the Applications of Carotid Vessel Wall Imaging
As novel information on carotid atherosclerotic plaque becomes increasingly assessible with vessel wall MRI techniques, there have come along new opportunities to optimize clinical diagnosis and management in patients with carotid artery disease or other atherosclerotic cardiovascular diseases. It is worth mentioning that the clinical value of vessel wall MRI usually depends on the strength and nature of the association between an imaging feature and the risk of atherothrombosis. Once an imaging biomarker is successfully developed, it is essential to elucidate its relationship with clinical events or their surrogates, including the evolution of imaging biomarkers prior to or after clinical events. Imaging biomarkers that are strongly implicated in the pathogenesis of clinical events may be useful for identifying the culprit lesion in clinical diagnosis, selecting patients at higher risk for stroke for CEA or CAS, and monitoring disease progression or therapeutic response at the lesion level.
To Identify the Culprit Lesion
In patients who present with neurological ischemic events, carotid atherosclerosis is currently considered to be the culprit lesion if the degree of luminal stenosis exceeds 50%, as specified in the TOAST classification [65]. While stenosis severity is an indicator of the likelihood of a carotid plaque to be accountable for ipsilateral thromboembolic events, the presence of stenosis itself is not a surrogate for atherothrombosis. Clinically, it is not uncommon to encounter cases in which only mild carotid stenosis (<50%) is found ipsilateral to brain infarct(s). In a retrospective study, Cheung et al. [66] studied 217 patients referred for carotid vessel wall imaging due to neurological ischemic symptoms who had <50% carotid stenosis on Doppler ultrasound or TOF MR angiography. Intraplaque hemorrhage, detected as hyperintense signals on MP-RAGE, was found in 13% (31 out of 233) of carotid arteries ipsilateral to neurological ischemic symptoms compared to 7% (14 out of 201) of contralateral carotid arteries (p < 0.05, Fisher’s exact test).
Freilinger et al. [67] interrogated the hypothesis that complicated, nonstenotic carotid plaques may contribute to clinical events previously classified as cryptogenic strokes. Thirty-two consecutive patients with ischemic stroke of undertermined etiology in the anterior circulation after extensive clinical workup were scanned with a multi-contrast carotid MRI protocol to detect complicated plaques (AHA Type VI) in the ipsilateral or contralateral carotid artery, defined as the presence of fibrous cap rupture, intraplaque hemorrhage, or mural thrombus. The mean time interval between qualifying stroke and carotid MRI was 5.8 ± 4.1 days. AHA Type VI plaques were found in 12 (37.5%) of carotid arteries ipsilateral to cryptogenic stroke, whereas none was found on the contralateral side (p = 0.001, McNemar’s test). The most common diagnostic feature was intraplaque hemorrhage (75%), followed by fibrous cap rupture (50%) and luminal thrombus (33%). This proof-of-concept study provided evidence that a substantial proportion of anterior circulation ischemic strokes previously classified as cryptogenic may be due to artery-to-artery thromboembolism from nonstenotic carotid plaques.
Similar findings have been reported by Gupta et al. [68] and Singh et al. [69]. Gupta et al. [68] used TOF for detecting intraplaque hemorrhage, which is less sensitive than MP-RAGE but more readily available in routine MR angiography studies of cervical arteries. A total of 119 (35.3%) patients were classified as having stroke of undetermined origin according to the TOAST criteria among 337 consecutive patients diagnosed with ischemic stroke. Twenty-seven cases, which had only unilateral anterior circulation infarct(s) and available TOF images during inpatient admission, were selected for analysis. Six (22.2%) patients were found to have high-intensity signals in the nonstenotic carotid artery ipsilateral to brain infarct(s), while none had high-intensity signals consistent with intraplaque hemorrhage on the contralateral side (p = 0.01, McNemar’s test). The study by Singh et al. [69] examined 35 consecutive patients with embolic strokes of undetermined origin with MP-RAGE. Ipsilateral and contralateral intraplaque hemorrhage was found in seven (20.0%) and three (8.6%) patients (p = 0.005 for difference), respectively. The three patients with intraplaque hemorrhage contralateral to stroke also had intraplaque hemorrhage on the ipsilateral side.
Despite these promising results, there remains a pressing need for large-scale, multicenter studies with standardized imaging protocols to determine the diagnostic features that can be incorporated into clinical diagnosis workup. Intraplaque hemorrhage is not rare even in asymptomatic patients. Silent fibrous cap rupture is also much more common than clinical events, which may stay unhealed for years. Residual luminal thrombus may provide the “smoking gun” evidence for recent thromboembolism from carotid atherosclerosis. However, the reliable detection of luminal thrombus remains challenging.
To Select Patients at Higher Risk for Stroke for Carotid Revascularization
In the clinical decision-making of carotid revascularization versus medical therapy alone, future risk of carotid-source ischemic stroke needs to be evaluated and compared to surgical risk including periprocedural stroke and myocardial infarction on an individual basis. As mentioned in the introduction section, multiple randomized clinical trials comparing CEA versus medical therapy alone have shown an unmet need for identifying which individual patients are at high risk for future stroke and, therefore, could benefit from prophylactic surgical intervention, among asymptomatic patients as well as symptomatic patients with mild-to-moderate carotid stenosis. These clinical trials were conducted during a period when clinical assessment of carotid atherosclerosis was largely limited to measuring luminal stenosis on angiography, which provided limited information on plaque characteristics. As carotid vessel wall imaging makes it possible to identify and quantify a variety of plaque characteristics, certain plaque features that are associated with neurological ischemic events have been shown in prospective, longitudinal studies to predict future risk of ipsilateral ischemic stroke. To date, evidence on the independent predictive value of plaque characteristics is most abundant for carotid plaque composition and luminal surface condition. Thus, vessel wall imaging information may help further classify carotid artery disease beyond stenosis severity.
In one of the earliest investigations using carotid MRI, Takaya et al. [70] followed 154 patients with asymptomatic 50–79% stenosis for an average of 38.2 months. Baseline plaque characteristics, including thin/ruptured fibrous cap (38% of cases; HR, 17.0; 95% CI, 2.2, 132), intraplaque hemorrhage (28% of cases; HR, 5.2; 95% CI, 1.6, 17.3), larger %lipid core area (HR, 1.6 per 10% increase; 95% CI, 1.2, 2.0), and greater maximal wall thickness (HR, 1.6 per 1 mm increase; 95% CI, 1.1, 2.3), were predictive of subsequent ipsilateral stroke or transient ischemic attack. Kwee et al. [71] showed that these plaque characteristics, including thin/rupture fibrous cap (HR, 5.8; 95% CI, 1.9, 17.3), intraplaque hemorrhage (HR, 3.5; 95% CI, 1.1, 11.9), and lipid core (HR, 3.2; 95% CI, 1.1, 9.5), were also predictive of recurrent stroke in 126 symptomatic patients with 30–69% stenosis. Compared to lipid core and/or fibrous cap status, there has been more abundant evidence on the prognostic value of intraplaque hemorrhage, thanks to its straightforward detection on heavily T1-weighted MRI. Three independent meta-analyses have been published on this topic, which highlighted the great potential of MRI-detected plaque characteristics for identifying high-risk patients [72–74]. Recently, Gupta et al. [75] conducted a cost-effectiveness analysis concerning asymptomatic carotid stenosis (50–89%), which compared two management strategies: (1) intensive medical therapy in which patients undergo CEA if there is substantial stenosis progression (≥90%) and (2) intensive medical therapy plus MRI-based patient stratification in which those with intraplaque hemorrhage on carotid MRI undergo CEA. MRI-based patient stratification was found to be cost-efficient for identifying asymptomatic patients with carotid stenosis who are likely to benefit from CEA.
Given these promising results, vessel wall imaging will likely play a major role in stratifying patients with mild-to-moderate stenosis to optimize outcomes of carotid artery disease in the near future. MRI of intraplaque hemorrhage can be readily incorporated into clinical protocols, and presence of intraplaque hemorrhage was strongly associated with increased risk of ischemic stroke, particularly in symptomatic patients [73]. Further optimizing the approach to imaging-based patient stratification is of high clinical significance since the majority of total ischemic strokes occur in patients with mild-to-moderate stenosis due to the vast population affected. However, prospective data from large-scale, multicenter studies remain scarce. While patients without intraplaque hemorrhage appear to have minimal risk for future stroke, the heterogeneous outcomes in patients with intraplaque hemorrhage remain to be understood.
To Choose Between CEA and CAS
CEA is a well-established technique that provides long-lasting prevention of ipsilateral stroke in patients with high-risk carotid artery disease. The perioperative risk for major surgical complications, including stroke, myocardial infarction, and death, is small but not negligible. CAS is less invasive than CEA and does not require general anesthesia [76]. Several randomized clinical trials have compared CEA and CAS in patients without high surgical risk. The composite event rate, which included both perioperative major events and long-term ipsilateral strokes, was similar between CEA and CAS. However, the overall stroke rate was higher with CAS (OR, 1.50; 95% CI, 1.22, 1.84), which was attributed to a higher perioperative risk of embolic stroke during CAS compared to CEA [77]. Current clinical guidelines by the Society for Vascular Surgery recommend CEA as the first-line treatment for most patients who require carotid revascularization. CAS is an alternative to CEA for selected anatomic or medical reasons. CAS mechanically disrupts carotid plaque during stent deployment, which is considered a major source for distal emboli as supported by intraoperative transcranial Doppler monitoring. As such, carotid plaque composition (and its thrombogenicity) may serve as a risk factor for perioperative embolization in the setting of carotid artery stenting.
Carotid plaques with intraplaque hemorrhage have large lipid-rich necrotic core, which is highly thrombogenic. Yoshimura et al. [78] tested the hypothesis that high-intensity signal on preoperative TOF-MRA, indicative of large intraplaque hemorrhage, is associated with increased risk of postoperative ipsilateral ischemic lesions on diffusion-weighted images. A total of 112 patients undergoing CAS were studied. High-intensity signal was present in 38 (33.9%) patients. After CAS, new ischemic lesions on diffusion-weighted images ipsilateral to CAS were found in 51 patients, which were more frequently seen in patients with high-intensity signal plaque (65.8% versus 35.1%, p = 0.002). Using the MP-RAGE sequence, Chung et al. [79] also examined whether intraplaque hemorrhage is a significant risk factor for cerebral embolism during CAS. Intraplaque hemorrhage was detected in 43 (45.7%) patients. New ipsilateral ischemic lesions on postoperative diffusion-weighted images were found in 9 (20.9%) and 16 (31.4%) patients with and without intraplaque hemorrhage (p = 0.35). There have been a number of other studies on this topic [80–83]. All evaluated intraplaque hemorrhage as a risk factor for periprocedural cerebral embolization, whereas some also evaluated other variables such as plaque volume, remodeling index, and tumor necrosis factor α. The techniques that were used to detect intraplaque hemorrhage varied substantially between studies. Recently, Brinjikji et al. [84] performed a meta-analysis on the association between intraplaque hemorrhage on pre-surgery MRI and clinical outcomes after CAS. Nine studies with a total of 491 patients were included. The composite outcome of stroke, myocardial infarction, and death within 30 days was 8.1% (13/160) in the intraplaque hemorrhage group and 2.1% (5/239) in the non-intraplaque hemorrhage group (OR, 4.45; 95% CI, 1.61, 12.30). The rate of new ipsilateral ischemic lesions on diffusion-weighted imaging was 49.7% (75/161) in the intraplaque hemorrhage group and 33.6% (81/241) in the non-intraplaque hemorrhage group (OR, 2.01; 95% CI, 1.31, 3.09) [84].
Overall, existing data support that high-risk carotid plaques, when disrupted during CAS, are more likely to lead to distal embolization and postprocedural stroke. How this pathophysiological association can be used clinically to optimize treatment selection for individual patients, such as switching to CEA or a different type of protection device (e.g., proximal protection, flow reversal), remains to be explored.
To Monitor Therapeutic Response
Imaging measurements of carotid atherosclerosis are often used to evaluate the efficacy of medical therapies. Because of the large sample size and long follow-up that are needed to power a clinical trial using cardiovascular events as the primary endpoint, the development of new anti-atherosclerosis therapies is usually prolonged and expensive. Surrogate endpoints by vessel wall imaging may greatly expedite the drug development process by dramatically reducing sample size and shorten study duration that are needed to demonstrate drug efficacy. As a noninvasive, ionizing radiation-free technique that provides a spectrum of quantitative measurements from plaque morphology to inflammatory activity, carotid MRI is ideally suited for monitoring therapeutic response. Importantly, MRI measurements of carotid atherosclerosis have shown excellent test-retest reproducibility in previous studies. Duivenvoorden et al. [15] compared common carotid mean wall thickness by MRI and common carotid intima-media thickness by ultrasound. Measurements were repeated 3 times within 3 weeks in 45 subjects, consisting of 15 healthy younger volunteers, 15 older volunteers, and 15 patients with 30–70% carotid stenosis. The coefficient of variation, which indicates measurement errors, was 6.9% for mean wall thickness by MRI and 12.8% for intima-media thickness by ultrasound. Other carotid wall measurements, such as mean wall area, showed similar measurement errors as mean wall thickness. Because of the smaller size, plaque components have larger measurement errors compared to wall thickness or area measurements. Nonetheless, plaque composition may tend to change earlier and/or more dramatically than plaque size under treatment, as studies have documented significant changes in carotid plaque composition before significant changes in carotid plaque size were observed [85, 86].
A number of carotid MRI studies have evaluated the effects of statin therapy on carotid wall. In one of the earliest investigations, Corti et al. [87] studied 32 carotid plaques and found a reduction in wall area but not in lumen area after 1-year simvastatin treatment. The ORION study was the first to evaluate changes in carotid plaque composition under statin therapy using multi-contrast vessel wall MRI. In the ORION study, Underhill et al. [85] studied 33 subjects with 16–79% carotid stenosis by ultrasound and elevated LDL cholesterol. After 2 years of statin therapy, there was a reduction in lipid core volume but not in carotid wall volume. Zhao et al. [85] performed a serial imaging study over 3 years in 33 subjects with documented necrotic core at baseline, which shed light onto the time course of plaque lipid depletion under lipid-lowering treatment. There was a significant reduction in necrotic core size in Year 1, whereas the reduction in wall volume was not apparent until Year 2.
Beyond cost saving via reducing sample size and shortening study duration, imaging-based clinical trials provide some other benefits, including clarifying the mechanisms of experimental agents and revealing heterogeneous responses to treatment at the individual level. Interestingly, different plaque phenotypes responded differently to the same lipid-lowering strategy. Intraplaque hemorrhage plaques were less likely to have lipid depletion than non-intraplaque hemorrhage plaques under intensive statin therapy, indicating a need for more aggressive or targeted treatment in patients with intraplaque hemorrhage [88]. Eventually, understanding atherosclerotic plaque progression or regression under various medical treatments may facilitate the pursuit of optimal medical therapy for individual patients. To this end, highly robust methods will be needed. Certain measurement errors may be acceptable (do not affect pathophysiological associations) in research that investigates disease mechanisms at the group level but may be unacceptable in the management of individual patients.
Future Perspectives
Validation of New Carotid Vessel Wall Imaging Techniques
Despite the established capabilities and expanding applications of traditional vessel wall imaging techniques, carotid MRI is an evolving field with tremendous opportunities in the coming years. New imaging techniques have been developed to overcome certain limitations of the traditional techniques so that imaging biomarkers of carotid plaque morphology and activities may be measured more accurately and reproducibly. As such, comparison with traditional techniques may not be sufficient. Further validation against CEA specimens is helpful to demonstrate if these new techniques bring about new or more detailed information on carotid plaques compared to traditional vessel wall MRI techniques.
Diffusion-weighted vessel wall imaging has been proposed as an alternative to T2-weighted imaging for measuring the lipid-rich necrotic core, but traditional diffusion-weighted imaging sequences may not be ideal for vessel wall MRI [89–91]. Xie et al. [92] developed a diffusion-weighted turbo spin echo sequence and demonstrated in a pilot study that the in vivo ADC value of lipid-rich necrotic core was significantly lower than that of fibrous tissue (0.60 ± 0.16 × 10−3 mm2/s vs. 1.27 ± 0.29 × 10−3 mm2/s, p < 0.01). Moreover, high-resolution T2 mapping has been developed as a more quantitative approach to lipid core quantification in carotid arteries [93–96], in which lipid core can be measured by pixel-level classification of T2 maps. Chai et al. [97] studied the performance of T2 maps for quantifying lipid core by using histological analysis. Dual T2 thresholds were used to detect lipid cores with and without intraplaque hemorrhage. A high correlation was seen between T2 mapping and histology in measuring percent lipid core area (r = 0.83, p < 0.001).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree