Intracranial arteriovenous shunts (AVSs) in children can be divided into pial arteriovenous malformations, vein of Galen malformations, and arteriovenous fistulae (AVF). Dural AVF and dural sinus malformations are rare entities within this group. The relative immaturity of the anatomy and physiology of the neonatal and infant brain results in the inability of the hydrovenous system to compensate in the face of such disorders. Thus, the clinical presentation reflects this difference in the underlying anatomy, physiology, and disorder between children and adults. In this article, we briefly review the presentation, natural history and management of these entities.
Key points
- •
Divided into arteriovenous malformations and arteriovenous fistulae.
- •
Presentation is different from adults: eg, heart failure in neonates.
- •
Commonest cause of haemorrhagic stroke in children.
- •
Digital subtraction angiography is the gold standard method of investigation.
- •
High projected life-time risk of haemorrhage warrant more aggressive treatment strategy.
Introduction
Intracranial arteriovenous shunts (AVSs) in children can be divided into pial arteriovenous malformations (AVMs), vein of Galen malformations (VGAMs), and arteriovenous fistulae (AVF), including single-hole fistulae. Dural AVF and dural sinus malformations (DSMs) are rare entities within this group.
The relative immaturity of the anatomy and physiology of the neonatal and infant brain results in the inability of the hydrovenous system to compensate in the face of such disorders. The AVSs in this population are of a distinct angioarchitecture often containing fistulous components with direct arteriovenous (AV) connections. AVMs can be multifocal and also often exhibit a complex pattern of venous drainage that can affect the entire venous system, often associated with venous malformations.
Thus, the clinical presentation reflects this difference in the underlying anatomy, physiology, and disorder between children and adults. Most intracranial AVSs in adults are pial AVMs or dural AVFs. The clinical presentation of heart failure is rare in adults; however, pediatric AVSs often present with heart failure in the neonatal period. Other differences in children include their association with other vascular abnormalities and genetic influences.
Introduction
Intracranial arteriovenous shunts (AVSs) in children can be divided into pial arteriovenous malformations (AVMs), vein of Galen malformations (VGAMs), and arteriovenous fistulae (AVF), including single-hole fistulae. Dural AVF and dural sinus malformations (DSMs) are rare entities within this group.
The relative immaturity of the anatomy and physiology of the neonatal and infant brain results in the inability of the hydrovenous system to compensate in the face of such disorders. The AVSs in this population are of a distinct angioarchitecture often containing fistulous components with direct arteriovenous (AV) connections. AVMs can be multifocal and also often exhibit a complex pattern of venous drainage that can affect the entire venous system, often associated with venous malformations.
Thus, the clinical presentation reflects this difference in the underlying anatomy, physiology, and disorder between children and adults. Most intracranial AVSs in adults are pial AVMs or dural AVFs. The clinical presentation of heart failure is rare in adults; however, pediatric AVSs often present with heart failure in the neonatal period. Other differences in children include their association with other vascular abnormalities and genetic influences.
Pial AVMs
Cause
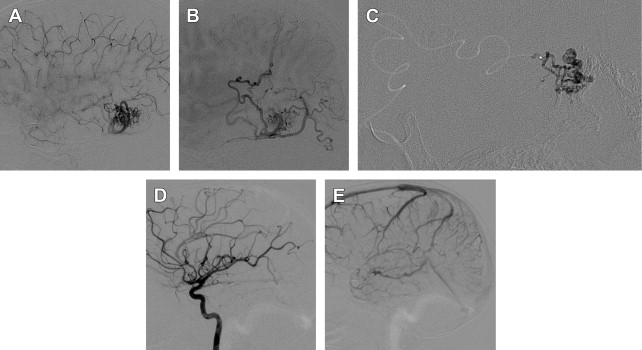
Pial AVMs ( Fig. 1 ) are the commonest cerebrovascular lesion in children. The lesion is thought to develop between the third and eighth week of fetal life. They are usually congenital lesions that arise from the abnormal development of the arteriolar-capillary network that exists between the arterial and venous circulations with an organ. Only 18% of AVMs that become symptomatic do so before the age of 15 years. The mortality from hemorrhage in a child is 25%. Intracranial pial AVMs may be associated with facial AVMs or cerebrofacial metameric syndromes (CAMS; or Bonnet-Dechaume-Blanc or Wyburn-Mason syndrome) as well as genetic conditions such as hemorrhagic hereditary telangiectasia (HHT; or Osler-Weber-Rendu syndrome) and neurofibromatosis type I. Single-hole AVFs, once considered a subset of AVMs, are thought to be a distinct vascular entity according to their angioarchitecture and thus are discussed separately in this article.
Presentation
Pial AVMs are rarely seen in the first year of life. They may present in infancy, but most present in the second and third decade of life up to the age of 15 years. If the contention that they are congenital is correct, this pattern implies that they undergo postnatal angioarchitecture evolution leading to presentation when the child is older.
Although presentation in the newborn is infrequent, AVMs may manifest as congestive heart failure (50%), followed by hemorrhage (37.5%). In infants, hemorrhage (30%) and macrocrania (27%) are more common than heart failure (23%). The commonest presentation of an AVM in childhood is hemorrhage (75%), either intraparenchymal or intraventricular, followed by seizures, which constitute approximately 15% of the presenting population. AVMs are the commonest cause of hemorrhagic stroke in children (30%–55%) and, as such, any child with spontaneous intracranial hemorrhage should be presumed to have an AVM until proved otherwise. Associated aneurysms are found in about 16% of children with AVMs.
Natural History
Enlargement of an AVM in children occurs with growth as a result of increased flow through poorly differentiated vessels and the potential recruitment of new arterial supply. Children have a higher rate of hemorrhage from an AVM at initial presentation compared with adults, which may reflect the underlying angioarchitectural differences in the age groups but equally may reflect the lower frequency of intracranial imaging for pediatric nonhemorrhagic symptoms.
The annual risk and rate of hemorrhage of a previously nonruptured AVM is probably similar to that of adults, ranging between 2% and 4%. Although children are more likely to present with hemorrhage, similar to adults, the rate of subsequent hemorrhage in children with AVM is approximately 90% lower than in adults with AVM. However, despite the annualized risk of hemorrhage being similar to that of adults, the cumulative risk is greater in children given the greater number of years left to live.
Diagnosis
Computed tomography (CT) is useful in the context of a ruptured AVM to assess the location and size of the hematoma, the presence of hydrocephalus, and mass effect. However, CT scan often fails to identify the precise source of bleeding. Magnetic resonance (MR) imaging is useful for localization of the AVM, its size, presence of hematoma, or posthemorrhagic changes. Functional MR imaging and MR tractography may be useful in an AVM located in an eloquent part of the brain, particularly in preparation for treatment. CT and MR angiography are useful to determine the afferent vessels of the AVM and the venous outflow of the malformation.
Digital subtraction angiography is the gold standard for the diagnosis of a cerebral AVM; no imaging modality other than digital subtraction angiography can exclude the presence of an intracranial AVS. It has a vital role in determining the angioarchitecture and hemodynamic features of the AVM and is essential to plan treatment. Angiography may be used during surgery to localize the lesion, distinguish arteries from veins, and ensure complete removal.
Treatment
The main goal of AVM treatment is to prevent intracranial hemorrhage and, to achieve this, the AVM must be completely obliterated. Although complete cure is desirable, it should be considered in the context of the anticipated natural history of the AVM in any particular child versus the risk of treatment. Clear indications for treatment of an AVM include hemorrhage, rapid neurologic deterioration, or the presence of an intranidal or feeding artery aneurysm. In children, consideration should be given to treatment of an asymptomatic AVM, because lifelong risk of hemorrhage is high.
Different therapeutic approaches include surgical resection, endovascular embolization, gamma knife radiosurgery, and proton beam therapy. These approaches are used as single treatment modalities or in combination, determined by the size, location, and vascular anatomy of the lesion. The treatment approach should be determined by factors that include the age of the patient, the clinical presentation, and the angioarchitecture of the AVM. Management is guided by a multidisciplinary team with experience in the treatment of neonates, infants, and children with intracranial AVMs. Grading systems have been used to guide treatment and assess outcome. The most widely used is the Spetzler-Martin classification, which is a surgical grading system. The Spetzler-Martin grade is determined by the size of the AVM, eloquence of the involved brain, and pattern of venous drainage.
Transarterial endovascular embolization with liquid embolic agents (see Fig. 1 ) has gained popularity in the last 2 decades, largely as a result of improvements in angiographic image quality, endovascular access products, and embolic materials, becoming a favored method in children in many centers. The choice of embolic material depends on the angioarchitecture of the AVM and operator expertise and preference. Each material has advantages and disadvantages. Liquid embolic agents are of 2 main types:
- •
Cyanoacrylate (glue)-based materials that polymerize on contacting hydroxyl ions in blood. Commonly used preparations include Histoacryl (n-butyl-2 cyanoacrylate [NBCA]), and TRUFILL NBCA liquid embolic system. Polymerization time can be modified by the addition of oil-based contrast agents like Lipiodol (labeled Ethiodol in the United States), which also confers radiopacity to the embolic to allow fluoroscopic visualization. Lipiodol is an ethyl ester of iodized fatty acids of poppy seed oil. GLUBRAN 2 (General Enterprise Marketing, Viareggio, Lucca, Italy) is comonomer of NBCA and metachryloxysulfolan monomer. The longer radical chain allows a lower temperature of polymerization and slightly slower polymerization than Histoacryl, which may result in lower toxicity and less inflammatory reaction. Glubran is approved for use in Europe and carries the CE mark.
- •
Precipitated polymers: Onyx is a nonadhesive liquid embolic agent composed of ethylene vinyl alcohol (EVOH) copolymer dissolved in dimethyl sulfoxide (DMSO), and suspended micronized tantalum powder to provide contrast for visualization under fluoroscopy. Onyx is available in 2 product formulations: Onyx 18 (6% EVOH) and Onyx 34 (8% EVOH). Onyx 18 travels more distally and penetrates deeper into the nidus because of its lower viscosity compared with Onyx 34. Onyx is delivered by slow controlled injection through a microcatheter into the brain AVM under fluoroscopic control. The DMSO solvent dissipates into the blood, causing the EVOH copolymer and suspended tantalum to precipitate in situ into a spongy, coherent embolus. Onyx immediately forms a skin as the polymeric embolus solidifies from the outside to the inside, while traveling more distally in the lesion.
Glue can be useful in direct AVF, where flow is rapid and high-concentration embolic polymerizes rapidly at the AV junction without escape into the venous system. Glue may be superior in occlusion of intranidal aneurysms and may be delivered through smaller flow-direct catheters in more remote locations. Small glue volumes can be injected because of rapid polymerization with limited nidal penetration, usually requiring repeated pedicle catheterizations, which may result in lower cure rates in larger nidal AVMs compared with Onyx. Glue injections unfold over seconds and are more difficult to control compared with Onyx injections, which unfold over minutes with time to pause, repeat angiography, and reflect. Onyx generally achieves better nidal penetration with larger volume of nidus occluded on each injection. Onyx offers potential complete cure of even large brain AVMs and has a higher rate of occlusion, but postprocedural hemorrhage rates may be higher. Onyx delivery requires DMSO-compatible microcatheters that generally are more rigid compared with the best glue-delivery microcatheters, thus making catheterization of tortuous small arterial pedicles more difficult. DMSO is toxic, and injection of large volumes should be avoided in small, sick children.
Outcome
Children in general have high compensatory capacity with good outcome compared with adults. Outcome following hemorrhage depends on the location and size of hemorrhage, with mortalities in hospital series around 8% to 50% and moderate to severe cognitive deficits at around 23%. Overall AVM obliteration has been reported in up to 40% of patients undergoing endovascular embolization, often taking multiple procedures. Embolization cure rates are better in small malformations (<1 cm) and those with single pedicles, including the fistula-type lesions, than in large lesions with more pedicles.
AVFs of childhood
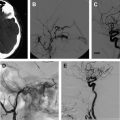
Pediatric AVFs are characterized by a direct AV connection with no intervening network of vessels, and therefore typically exhibit rapid flow, often causing high-output cardiac failure. The pediatric AVFs include VGAM, pial AVF, DSM, and infantile high-flow dural fistula ( Figs. 2–7 ).
VGAM
Definition and cause
VGAM (see Figs. 2 and 3 ) can be defined as a congenital AV shunt, centered on the tela choroidea of the third ventricle, supplied predominantly by choroidal arteries, and usually draining into an aberrant falcine sinus. In VGAMs, the thalamostriate and internal cerebral veins do not communicate with the vein of Galen but instead drain into the posterior and inferior thalamic veins, and either join a subtemporal vein or, more commonly, the lateral mesencephalic vein to open into the superior petrosal sinuses, which gives the characteristic angiographic epsilon shape or epsilon vein on the lateral angiogram. The straight sinus is usually absent, with a falcine sinus draining the malformation to the superior sagittal sinus.
In some instances it may be difficult to distinguish between a VGAM and a thalamic or tectal AVM draining into a dilated vein of Galen, and a medially located pial AVF draining into the vein of Galen. In general, 2 types of angioarchitecture are described in VGAMs: choroidal and mural. The former corresponds with a primitive condition involving all of the choroidal arteries and an interposed network before opening into the large venous pouch. The latter type corresponds with direct AVF within the wall of the median vein of the prosencephalon. The fistulas may be single but are often multiple, converging into a single venous chamber or into multiple venous chambers located along the anterior aspect of a venous pouch or along 1 dilated choroidal vein before reaching the venous pouch.
VGAM is rare, but is the most common intracranial AV shunt presenting in the neonatal period. Our institution receives around 1 to 2 new VGAM referrals per month, but it is difficult to be certain what catchment population this comes from, or what proportion reaches us. Disproportionate attention is given to VGAM, because the logic for treatment of all other high-flow AVSs of the neonatal period is based largely on our experience with VGAMs.
Paget first explicitly dealt with the embryology of this condition, referring to the possibility of an early embryonic AV shunt developing in the tela choroidea of the third ventricle, and postulating as to its consequences. Raybaud and colleagues in 1989 stated that the ectatic vein in VGAM was the embryonic precursor, the median vein of the prosencephalon, and not the vein of Galen itself.
Presentation
Antenatal diagnosis of VGAM is increasingly made using ultrasound and MR imaging. It is puzzling that it has never been shown on a 20-week gestational age antenatal scan, because the embryology suggests that it must be present at this stage. Antenatal diagnosis of VGAM raises some difficult considerations; some develop in utero parenchymal brain damage, and have a poor prognosis. Most have normal brain in utero, and present with either antenatal or perinatal heart failure.
Gold in 1964 described the following clinical stages in patients with VGAMs: (1) neonates with cardiac insufficiency, (2) infants and young children with hydrocephalus and seizures, and (3) older children or adults with headaches and subarachnoid hemorrhage. Amacher later added a further clinical group that included neonates and infants with macrocephaly and minimal cardiac symptoms. The most common mode of presentation in a neonate is heart failure caused by extremely low-resistance flow through the AV shunt. These children are often gravely ill, and may progress to multiorgan failure and ischemic brain damage within hours to days, despite optimal support in an intensive care unit. Infants and children may also present with hydrocephalus or macrocephaly, often associated with the development of jugular bulb stenosis and venous hypertension.
Ventricular drainage in VGAM should be avoided if possible. Johnston and colleagues, in a literature review, noted that of 11 shunted infants 7 died and only 1 had no deficit. In an additional group of 6 shunted children, only 2 had no deficit. In the series by Zerah and colleagues, only 1 of 17 infants had an uneventful shunting procedure. Wherever possible in this group, we advocate embolization, aiming to cure the AV shunt, and deferral of ventricular drainage wherever possible. Third ventriculostomy has been used successfully in this setting, although preferably after embolization to reduce the rate of the AV shunt.
Older children or adults sometimes present with previously undiagnosed (or untreated or partially treated) VGAM. Most present with hemorrhage, or signs of increased intracranial pressure caused by venous hypertension. Treatment is technically difficult, and has a higher complication rate than in children.
Natural history
The natural history data are not robust; the diagnosis is often never made, and the literature confuses VGAMs with other disorders. Most neonates in heart failure die untreated. Infants and children presenting with hydrocephalus have a more indolent course, but often evolve toward chronic venous ischemia resulting in developmental delay, seizures, and parenchymal hemorrhage. Adults typically present with hemorrhage or signs of increased intracranial pressure. Spontaneous thrombosis of VGAM is probably rare; in the series by Lasjaunias and colleagues, 5 out of 120 patients (4%) developed spontaneous thrombosis, with 2 neurologically normal.
Diagnosis
Antenatal diagnosis is usually by identification of an anechoic structure in the region of the cistern of the vein of Galen with demonstrable flow within it on Doppler ultrasound, although ultrasound is poor at assessing the brain parenchyma. Fetal MR imaging is useful, both to characterize the VGAM and to assess the brain parenchyma. Many neonates are not diagnosed promptly; any neonate with unexplained high-output cardiac failure should have imaging of the head to exclude a VGAM. MR imaging is optimal in this regard, because it allows both diagnosis and assessment of brain parenchyma, although CT and CT angiography (CTA) are more often used in the clinical setting. Older children and adults are best imaged by MR imaging.
However, the angioarchitecture of the lesion is best shown and appreciated by catheter angiography. Catheter angiography should only be performed as a prelude to treatment; iliac artery occlusion is common in angiography in neonates, and becomes an issue in subsequent treatment. The dominant arterial supply usually comes from the posterior choroidal arteries, although their morphology is distorted from the usual adult pattern. The pericallosal and anterior choroidal arteries also often contribute. In older children, subependymal vessels passing through the thalamus and midbrain also often contribute to the AV shunt.
Although the distinction in angioarchitecture is well described as choroidal and mural types according to Lasjaunias and colleagues, in our experience there is a spectrum ranging from a single AV shunt to innumerable arterial feeders from multiple sources. In VGAM, the dilated median vein of the prosencephalon is thought not to communicate with the deep venous system of the brain, although there are reports of thalamic hemorrhage after embolization; in this regard we have observed at least 2 cases in which we performed retrograde catheterization of the thalamic veins off the sac.
Treatment
Neonates
Whether presenting antenatally, or more commonly perinatally, it is not clear when these children are best treated (see Fig. 7 ); Lasjaunias traditionally decided by a scoring system, aiming to avoid treatment of neonates wherever possible, to allow treatment when the child is larger and it is technically easier and safer. At Great Ormond Street Hospital (GOSH) for Children, our approach has changed over time, influenced by both the death of neonates and infants from heart failure while awaiting treatment, and excellent outcomes in children who were regarded as too systemically unwell to treat by other operators. Our current approach is to perform embolization on any VGAM with cardiac failure as soon as possible after delivery, provided there is no imaging evidence of severe parenchymal damage to the brain.
The initial aim of treatment is to relieve heart failure, rather than cure the malformation. Subsequent staged embolization aims for cure by 6 months. It is self-evident that the very sick children with low scores tend to have worse outcomes, but at GOSH for Children we offer treatment to even very sick children provided there is not evidence of diffuse parenchymal damage. We have seen cured patients growing up as normal children.
The first step is catheter angiography (see Fig. 2 ). We generally use a femoral approach, using a 4-Fr sheath, and a short Berenstein diagnostic catheter. Bilateral internal carotid and left vertebral high-frame-rate angiography is performed, which consumes most of the available contrast dose in a neonate, and most of the subsequent intervention is performed under fluoroscopy, with occasional microcatheter angiography.
In our practice at GOSH for Children, all neonates are treated by transarterial embolization using concentrated Histoacryl rendered radiopaque with tantalum. This technique is quick, and reliably relieves heart failure in these gravely ill children. All neonates return to the intensive care unit after embolization, and almost all need a combination of positive inotropes and vasodilators, aiming for management of pulmonary arterial hypertension, and careful control of systemic pressure. They are generally kept heavily sedated and intubated in the intensive care unit for 48 hours after embolization.
Although no clear statement can be made, the optimal target is probably around 50% reduction in cardiac output in the first stage of embolization. The fraction of cardiac output passing through the AV shunt in a VGAM has exceeded 90% in all neonates in whom we have measured, a state that is clearly incompatible with life, and for which no animal model exists. Sudden closure of a shunt of this magnitude often precipitates severe left ventricular failure, and occasionally intracranial hemorrhage. Such an abrupt physiologic alteration needs to be carefully managed; before, during and after the procedure, and requires the input of pediatric cardiology, intensive care unit specialists, anesthesia, and experienced pediatric interventional neuroradiologists.
Infants and children
Those children who are not treated in the neonatal period usually present, sometimes repeatedly, with hydrocephalus, macrocephaly, seizures, or developmental delay, probably mediated by increased intracranial venous pressure ( Fig. 3 ). The logical approach would be an attempt at reduction of intracranial venous pressure. In contrast, the optimal solution is cure of the AV shunt, although this should probably be staged, and is not always possible because by this age there are often innumerable transthalamic and transtectal arterial feeders. Cerebrospinal fluid diversion should be avoided wherever possible.
Every conceivable approach has been used for VGAM; transarterial occlusion with coils, glue, or Onyx, retrograde transvenous embolization with coils, and, in our center, retrograde embolization using glue and stent placement of stenosis of the jugular bulb. Stereotactic radiosurgery and conventional neurosurgery have also been attempted. It is not clear which of these approaches is best in any set of circumstances and hence a multidisciplinary approach is essential.
Outcomes
The poorer the state of the child at the outset of the disease process, the lower the probability of a good outcome. Our reluctance to use the rigid Lasjaunias clinical criteria for treatment stems from our personal experience. At our institution, we have seen children deemed too sick to treat by these criteria who end up normal, and children deemed too well to treat end up dead. Our approach in VGAM has evolved to mirror that in any other pediatric neurovascular disorder: a multidisciplinary decision involving pediatric neurology, neurosurgery, interventional neuroradiology, and pediatric intensive care specialists is made, taking into account the radiological state of the brain, the clinical state of the child, and the wishes of the parents. The only absolute exclusion to treatment is evidence of severe parenchymal damage to the brain (see Fig. 7 ). Severe physiologic derangement (high shunt volume, renal failure, hepatic failure, heart failure on maximal inotropes, requirement of high-frequency ventilation and so forth) makes periprocedural management more difficult, but tends to polarize outcomes into death or good outcome, provided the brain is intact at the time of embolization. Overall, neonates presenting to GOSH for Children now have around a 60% chance of being neurologically intact, 15% chance of mild disability, 10% chance of severe disability, and 15% chance of death. This probability includes many patients who would be refused treatment by conventional criteria.
High-flow Pial AVF
Definition and cause
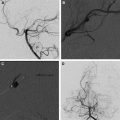
Pial AVFs of the brain (see Fig. 4 ) are sometimes termed single-hole AVFs and account for approximately 3.3% to 4.7% of all intracranial AVMs. They are characterized by direct connections between arteries and veins with arterial supply derived from cortical or pial arteries, and are frequently associated with large to giant venous pouches, which can occur as a solitary fistulae, but frequently are multiple, which is a characteristic finding of this entity. Most of these are situated in the supratentorial neuroparenchyma and are more common in the frontal and temporal lobes.
Natural history
The exact incidence of a pial AVF in children is not known because of its rarity. Most published papers have included this type of angioarchitecture within the overall population of the cerebral AVMs or even the VGAMs. As within most vascular anomalies in this age group, there is a male predominance and strong genetic association in pediatric cases in which there are multiple intracranial AVFs. Conditions such as HHT and encephalocraniocutaneous lipomatosis are well-recognized associations in the literature.
Presentation
Presentation is usually within the first 5 years of life with the mean age in one series at 23 months. Symptoms attributable to pial AVFs are usually the consequence of high-flow AV shunting, leading to cardiac failure, hemorrhage, macrocrania, seizures, focal neurologic deficits, edema, or mass effect from the venous varices. When presenting with hemorrhage, AVF can also be associated with false arterial or venous aneurysms; these should be vigilantly identified, because this is an indication and a focal target for treatment. Venous outflow obstruction in the form of pial venous outflow stenosis, dural sinus stenosis and thrombosis seems to be associated with hemorrhagic presentations, similar to the experience with AVMs.
Diagnosis and treatment
The diagnosis follows the imaging algorithm of pial AVM as detailed earlier using CT, CTA, MR imaging, MR angiography, and catheter angiography methods.
The goal of treatment in pial AVFs in young children, even when asymptomatic, is the rapid control of the lesion (AV shunt), because of the worse neurocognitive prognosis and mortality when managed conservatively. Surgery has been reported to be successful in a small number of cases of neonates and infants presenting with a symptomatic nongalenic AVF. However, our experience is that endovascular embolization is a safe and often curative treatment ; the technique used is identically to that described with concentrated glue and coils to occlude the fistula.
DSM and Infantile Dural AVF
Definition and cause
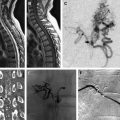
These disorders are collectively rare (see Figs. 5 and 6 ) and the true incidence and prevalence are difficult to quantify accurately. We receive around 1 per year; around 10% of the neonatal VGAM caseload. DSMs are thought to be a consequence of uncontrolled development of the posterior sinuses, including transverse sinus, sigmoid sinus, and/or confluence of sinuses, rather than of embryologic nondevelopment.
Presentation
DSMs generally present in the neonatal and infancy periods, although they usually become symptomatic on the average at 5 month of age. The most frequent clinical presentation is macrocrania followed by seizures, psychomotor delay, and intracranial hemorrhage. The other clinical presentations include congestive cardiac failure, cranial bruit, facial vein dilatation, and intracranial hypertension (intracranial hypertension being associated with macrocrania).
Natural history
Anatomically there are 2 types of DSM :
- 1.
DSM involving the adjacent posterior sinuses, with giant pouches and slow-flow mural AV shunting. Partial thrombosis of the sinus may occur and can also be observed in utero. The dural sinus pouch at birth communicates with the other sinuses and drains normal cerebral veins.
- 2.
DSM of the jugular bulb malformation with otherwise normal sinuses that appears as a sigmoid sinus-jugular bulb diaphragm and is associated with a petromastoid-sigmoid sinus high-flow AVF, which is usually a single-hole type. DSMs usually have a benign course, remain asymptomatic for a long time, and are incidentally discovered. Their treatment by embolization is technically easy, and the outcome of most patients is complete exclusion of the shunt and favorable neurologic outcome.
Associated multiple slow-flow AV shunts are consistently shown within the dural wall of the DSM contributing to the hydrovenous restriction by venous congestion of the brain. The brain at birth has to drain solely through the diseased sinus before cavernous capture. Early and rapid spontaneous thrombosis of a DSM lake and secondary dysmaturation of the jugular outlets further compromise cerebral venous drainage and can lead to venous infarction and lethal intraparenchymal hemorrhage. Unilateral DSMs, which have lateralized from the torcular, have a more favorable outcome as 1 normal draining venous sinus is maintained. It is important to have the ipsilateral cerebral hemisphere drain via an alternate normal venous pathway, either by cavernous capture or by a persistent medial occipital sinus bypassing the thrombosed sinus into the ipsilateral jugular vein or into the contralateral sinus via the superior sagittal sinus.
Diagnosis and treatment
Diagnosis on antenatal ultrasonography or MR imaging is possible but requires recognition of the disease condition by an experienced sonographer or neuroradiologist. Following antenatal diagnosis, MR imaging of the brain should be performed to determine the location of the DSM and quantify the extent of brain damage. If the DSM is located on the midline and large, early angiography is recommended to assess the anatomy of the cerebral veins. The physiologic issues and decision making are similar to other AVSs but the techniques used are more variable.
The therapeutic options thus depend on the angioarchitecture of each individual case and the state of maturation or dysmaturation of the venous sinuses. When partial or no cavernous capture and no pial reflux are seen, there is an option of treating with heparin and embolization of the shunts with glue, in anticipation of cavernous capture to take place with minimal or no consequences for the hydrovenous equilibrium of the maturing brain. If there is a significant shunt causing pial reflux, embolization is necessary to prevent venous hypertension and cerebral ischemic damage. The goal is to reduce pial reflux by occluding the shunts while maintaining patency of the sinuses. If there is complete occlusion of a mural AVS, this will likely lead to complete sinus thrombosis, resulting in an absence of a venous outlet for the brain and leading to poor outcome.
The prognosis, and specifically the mortality, associated with dural AVSs reported in the literature are extremely high, on the order of 38% in all ages and 67% in the neonatal subgroup. However, the perception of a DSM at the torcular being lethal has not been verified by our experience. Although these lesions are difficult to manage, we have achieved outcomes broadly similar to those of VGAM. Pial AVF and infantile dural AVF have a better prognosis.
Cranial AV shunts key facts:
- •
Divided into AVM and AVF
- •
Presentation is different in adults (eg, heart failure in neonates)
- •
Commonest cause of hemorrhagic stroke in children
- •
Digital subtraction angiography is the gold standard method of investigation
- •
High lifetime risk for significant mortality and morbidity warrants more aggressive treatment strategy
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree