CT and MR Contrast-Enhanced Tissue Perfusion Imaging: Basic Methodology, Postprocessing, Reliability Testing
Kohsuke Kudo, MD
▪ Introduction
Tissue perfusion can be measured using several methods, such as positron emission tomography (PET),1,2 single photon emission computed tomography (SPECT),3,4 computed tomography (CT),5,6 and magnetic resonance (MR) imaging.7,8 Among these techniques, PET and SPECT are the current standards of reference for in vivo assessment of the cerebral blood flow (CBF) and metabolism. However, high costs and limited accessibility have restricted the widespread clinical use of these modalities.
Compared to nuclear medicine methods, dynamic CT and MR perfusion techniques using bolus injection of contrast material have a number of practical advantages. First, these techniques can be applied immediately after unenhanced brain CT or MRI, which are performed to evaluate acute ischemic change, hemorrhage, and occlusion of arteries; CT and MRI are the choice modalities when acute stroke is suspected. Second, the examination time is considerably shorter for CT and MR perfusion (acquisition times typically range from 60 to 90 seconds). Third, the spatial resolution is much higher in these techniques compared with PET or SPECT. Finally, no special equipment is needed, except for a power injector, which is usually available in the scanner room in most hospitals. The potential limitation of short scan range in CT perfusion has now been overcome with advances in multidetector row CT (MDCT) including moving table techniques (toggling table or shuttle mode)9,10 and area detector CT (ADCT).11 Another drawback of CT perfusion is the radiation exposure. Attempts have been made to reduce the radiation dose, and the use of lower tube voltages (i.e., 80 kVp) and currents has become standard in many protocols.12,13 Quantitative analysis with both CT and MR perfusion is a common practice now, and there are several reports that have confirmed good correlations with PET or SPECT.14,15,16 and 17 Accuracy and reliability tests have been conducted using digital phantoms,18,19 and extensive refinement of analysis algorithms has been carried out.
CT perfusion imaging is also performed using cold Xe (Xe-CT),20,21 in which Xe acts as a diffusible tracer. However, it has several disadvantages such as the sedative effect of Xe and the requirement for special equipment (an inhalation system for Xe). Therefore, the clinical use of this technique is limited. MR perfusion is also known as perfusion-weighted imaging (PWI), and there are two types of MR perfusion that use dynamic contrast injection, that is, dynamic susceptibility contrast (DSC)7,8 and dynamic contrast-enhanced (DCE) PWI.22 DSC uses T2 or T2* contrast and is mainly used for blood flow measurement, while DCE uses T1 contrast and its main application is permeability analysis. In addition, arterial spin labeling (ASL) is now available in most clinical MR scanners.23,24 As ASL uses an intrinsic tracer of labeled blood, it is completely noninvasive and does not require contrast injection.
This chapter discusses basic principles, methods, and postprocessing issues for dynamic contrast-enhanced CT and DSC-MR perfusion. These techniques share the same methods for analysis as they use the same principles. Therefore, there are many common characteristics including behavior of the tracer, analysis algorithms, and limitations, albeit several differences also exist. CT perfusion has several advantages over MR perfusion, including more reliable quantitative results, higher spatial resolution, less image distortion, and better suitability for emergency scans. In contrast, MR perfusion has higher image contrast and wider scan coverage than has CT, and involves no radiation exposure (Table 5.1).
▪ Methodology
In order to measure blood flow dynamics, a “tracer” that flows in the bloodstream is required. Iodine and gadolinium (Gd) contrast materials are used in CT and MR perfusions, respectively, as these are commonly used in clinical practices. These contrast materials are macromolecules that do not pass through the blood-brain barrier (BBB). Therefore, they act as nondiffusible intravascular tracers (remaining in the blood) in normal brain tissue in which the BBB is intact, while diffusible tracers (freely diffuse across vascular wall into extravascular space) are used in PET and SPECT. As the kinetics of these tracers are different, so are the principles and analysis method.
When an intravascular tracer is used, rapid injection of the tracer and repetitive scans during its first passage are required for measuring the hemodynamics; this is based on the indicator dilution theory.25,26 Many hemodynamic parameters are derived from the measured dynamic curve. For quantitative analysis, the concentration of the tracer should be used, rather than the CT number or the MR signal itself. Therefore, CT numbers and MR signals are converted into tracer concentrations. In contrast to the linear relationship between CT number and iodine concentration, the concentration of Gd contrast is related to ΔR2*.7,27 Therefore, the following equation is used for the conversion.
TABLE 5.1 Characteristics of CT and MR Perfusion | |||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||
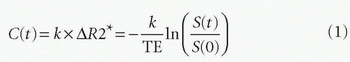
where C(t) is tracer (contrast) concentration at a given time t, k is a constant, and S(t) is the MR signal. This constant value, k, depends on the scan sequence, parameters, magnetic field strength, and tissue properties.27 Therefore, MR perfusion has potential difficulties in the accurate quantification of hemodynamic parameters. After this conversion, the time-concentration curve (TCC), C(t), is derived, and TCC is further used for quantitative analysis. TCC is recommended for semiquantitative analysis, but the time signal curve, S(t), can be also be used, especially for time-related parameters (see below), because such parameters are not affected by this conversion of contrast.
▪ Semiquantitative Parameters
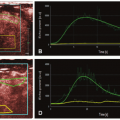
Semiquantitative parameters are derived directly from the TCC of tissue without any complex calculations (Fig. 5.1).28 The timerelated parameters are bolus arrival time (BAT), time to peak (TTP), first moment (FMT), and the full width at half maximum (FWHM), and these are sensitive to hemodynamic delay due to ischemia. Maximum slope (MS) and area under the curve (AUC) reflect CBF and cerebral blood volume (CBV), respectively, but not quantitatively. Maximum concentration (Cmax) also reflects CBV. All these parameters are affected by injection speed, amount of contrast material and saline chase, cardiac output, carotid stenosis, etc. For example, when the injection speed is low, BAT, TTP, and FMT become larger, and MS becomes smaller. AUC and Cmax are directly related to the amount of contrast. Therefore, they cannot be compared among subjects and even for studies on the same subject. The absolute values of these parameters are less meaningful, and relative values should be used for comparison.
▪ Quantitative Parameters
Quantitative analysis is performed by using the TCC of both tissues and arteries (C(t) and Ca(t), respectively). For both CT and MR perfusion, deconvolution is the most common method for quantitative analysis (Fig. 5.1).8,29,30 The following relationship has been shown for C(t) and Ca(t).
where R(t) is the tissue residue function and? denotes the convolution operation. R(t) represents the fraction of the injected tracer remaining in the vasculature at time t, and is calculated through deconvolution. If the contrast bolus remains in the artery for an infinitely short time, the shape of Ca(t) would be an impulse. In such an ideal situation, the observed C(t) becomes equal to R(t) (Fig. 5.2). However, in a practical situation, Ca(t) becomes a curve owing to the finite duration of the venous injection of contrast and mixture in the lungs and heart. Therefore, deconvolution is needed to obtain R(t) from measured Ca(t) and C(t), even with relatively low injection rates (3 to 5 mL/s), which are clinically possible.30 After deconvolution, CBF, CBV, and mean transit time (MTT) are derived from R(t). In practice, Ca(t) is called the arterial input function (AIF), which is determined by measuring the TCC of a large artery such as the middle cerebral artery (MCA) or the anterior cerebral artery (ACA), by the region of interest (ROI). However, the ROI usually has partial volume averaging, which causes the underestimation of AIF.27,31 Therefore, correction for partial volume averaging should be carried out before deconvolution with a scaling factor, which is determined by AIF and the venous output function (VOF).32 In brain, the VOF, Cv(t), is usually determined by the ROI of the superior sagittal sinus (SSS), which is sufficiently large to avoid partial volume averaging.
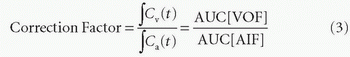
CBF refers to the blood volume flowing through a unit tissue volume for a unit of time (mL/100 g/min) and is one of the most important physiologic parameters, which is closely related to maintaining electronic functions and cellular metabolisms.33,34 and 35 For example, a study of monkeys showed that reversible motor dysfunction (paralysis) occurs for CBF below 23 mL/100 g/min, and complete hemiplegia occurs for CBF less than 8 to 9 mL/100 g/min. Furthermore, an electroencephalogram showed decreased action potential below 20 mL/100 g/min and electronic failure at 15 to 16 mL/100 g/min.34 In addition to these electronic failures, morphologic damage is also induced by failure of the integrity of the cell membrane due to depletion of adenosine triphosphate. This membrane failure is time dependent, as it occurs at 6 to 8 mL/100 g/min for 1-hour ischemia36 and at 12 mL/100 g/min for 2-hour.37 Therefore, quantification of CBF is theoretically important for the evaluation of tissue viabilities in acute stroke.
In a deconvolution method, CBF is determined as the peak height of R(t); however, CBF is also quantified by a MS model that assumes no venous output during the contrast bolus.5,38

CBV refers to the blood volume in a unit tissue volume, which is independent of flow (mL/100 g), and, thereby, is related to the vascular bed. When cerebral perfusion pressure (CPP) decreases due to stenosis or occlusion of a proximal artery, an increase in CBV occurs as the first compensation mechanism to maintain
CBF in ischemic tissue, and this mechanism is called “vascular reserve” (Figs. 5.3 and 5.4).34 After maximum dilatation of the vascular bed, CBF decreases by a further reduction in CPP. In the infarct core, a severe decrease of CBV is observed. In tumor cases, CBV is used for the estimation of vascular bed, which sometimes reflects malignancy (neovascularity).39 CBV is quantified as the AUC of R(t), but it is also estimated as the ratio of AUC of tissue and large vessel (VOF), which does not have partial volume averaging.8
CBF in ischemic tissue, and this mechanism is called “vascular reserve” (Figs. 5.3 and 5.4).34 After maximum dilatation of the vascular bed, CBF decreases by a further reduction in CPP. In the infarct core, a severe decrease of CBV is observed. In tumor cases, CBV is used for the estimation of vascular bed, which sometimes reflects malignancy (neovascularity).39 CBV is quantified as the AUC of R(t), but it is also estimated as the ratio of AUC of tissue and large vessel (VOF), which does not have partial volume averaging.8
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree