- •
Cardiac magnetic resonance (CMR) is the current golden standard for accurate assessment of ventricular volumes and global and regional function, thus providing information on prognosis.
- •
Extracellular accumulation of gadolinium, imaged at 10 to 30 minutes after injection, identifies areas of myocardial fibrosis commonly seen in cardiomyopathies, generally subendocardial or transmural in a coronary distribution in the case of ischemic cardiomyopathy.
- •
Transmural extent of LGE can predict recovery of myocardial function after revascularization in patients with coronary artery disease.
- •
Negative stress CMR with either adenosine or dobutamine stressors has excellent negative predictive value for subsequent coronary events.
- •
Noncoronary segmental distribution and midwall or epicardial involvement of LGE are atypical of myocardial infarction. This allows discrimination between ischemic and nonischemic etiologies of cardiomyopathy, which can assist in clinical decision making.
- •
Takotsubo cardiomyopathy is characterized by left ventricular apical dilation and regional hypocontraction, absence of first-pass perfusion abnormality, and minimal or absent concomitant subendocardial LGE.
- •
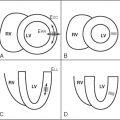
Because of its higher spatial and temporal resolution, CMR can offer an improved assessment for the presence and extend of ventricular noncompaction, compared with echocardiography.
- •
CMR can accurately quantify severity of valvular regurgitation and stenosis using phase contrast imaging. In the same study CMR can assess the physiologic impact of the valvular heart disease by providing quantitation of large vessel morphology and ventricular volumes and functions.
- •
CMR is the most sensitive technique for detecting clinically unrecognized small subendocardial myocardial infarctions that do not result in wall motion abnormality or ECG changes, thereby identifying patients at risk of cardiac events.
- •
CMR infarct imaging can provide direct information on the amount of irreversibly injured myocardium and viable myocardium, including the extent of periinfarct zone and microvascular obstruction, which are strong predictors of cardiovascular outcome in patients after myocardial infarction (MI).
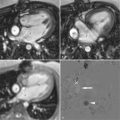
A 54-year-old woman without significant risk factors for coronary artery disease presented to our services complaining of a 4-week history of exertional dyspnea (NYHA class III) and pedal edema. The patient did not have any history of alcoholism, family history of heart disease, or sudden cardiac death. ECG showed sinus rhythm and a left bundle branch block pattern of unknown duration. Transthoracic echocardiography showed a dilated left ventricle and severe global hypokinesis with reduced ejection fraction of 25%. A coronary angiography was performed that revealed no significant coronary stenosis. CMR was performed ( Figure 10-1 ).

Comments
Determining the etiology of heart failure is essential for directing the correct treatment and predicting survival of patients who presented with new cardiac failure. CMR can often differentiate ischemic from nonischemic etiologies by the pattern of distribution of LGE. Indeed, in patients with idiopathic nonischemic cardiomyopathy, there is often a distinct pattern of LGE presenting as patchy longitudinal midwall LGE that spares the subendocardium and subepicardium and does not conform to a coronary vascular territory, as in our current case. The transmural extent of this midwall LGE has been implicated in the pathogenesis of ventricular arrhythmias in patients with nonischemic dilated cardiomyopathy. More recently LGE has been shown to prognosticate cardiac events in patients with this condition.
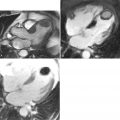
A 48-year-old woman without coronary risk factors was admitted to the emergency room following 30 minutes of severe retrosternal discomfort associated with diaphoresis. ECG showed widespread T-wave inversion in anterolateral leads. Emergency cardiac catheterization revealed unobstructed epicardial coronary arteries with LV distal akinesis resembling apical ballooning but without any intracavitary thrombus formation. CMR was performed the following day to assess the degree of ventricular dysfunction ( Figure 10-2 ).

Comments
Left ventricular (LV) apical ballooning (Takotsubo cardiomyopathy) is a unique clinical entity characterized by transient anteroapical hypokinesis in the setting of elevated adrenergic tone, ECG abnormalities mimicking acute coronary syndrome, modest elevation of cardiac enzyme levels, and a lack of angiographically significant epicardial coronary stenoses. Although diagnosis is usually suspected by a lack of significant coronary lesion on angiography, CMR is useful in this context by providing precise evidence of LV structural changes, exclusion of concomitant significant obstructive coronary disease, assessing for any extent of myocardial necrosis, and noninvasive monitoring for recovery of ventricular function.
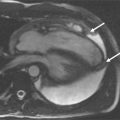
A 56-year-old man with a history of anterior myocardial infarction one year ago was referred to CMR for assessment of new onset chest pain, ventricular function, and myocardial viability ( Figure 10-3 ). During the hospitalization for his acute MI, coronary intervention of the proximal LAD was complicated with luminal dissection and as a result there was incomplete coronary revascularization of the infarct-related artery despite attempts to perform coronary stents.


Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree