- •
Cardiac magnetic resonance (CMR) is ideally suited for imaging the pericardium because of its large field of view and ability to differentiate soft tissue based on fat and water composition.
- •
By CMR normal pericardium is considered less than 3 mm thick and a pericardium greater than this thickness is indicative of pathology.
- •
Normal pericardium appears as a rim of low signal intensity on T1- and T2-W imaging.
- •
In acute pericarditis T1- and T2-W spin echo (SE) sequences may generate images in which the pericardium has a moderate to high intensity signal.
- •
Calcified pericardium will appear dark on images generated using T1-W SE, T2-W SE, fat saturated T2-W gradient echo (GRE), and steady-state free precession (SSFP) imaging.
- •
On T2-W SE imaging typical pericardial cysts have a homogenous high intensity signal.
- •
Metastatic melanoma to the pericardium will have a high intensity signal on T1-W SE imaging owing to paramagnetic metals bound by melanin.
- •
In patients with isolated congenital absence of the pericardium CMR imaging with SSFP, T1-W SE and T2-W SE all will demonstrate a lack of pericardium and axial imaging will show that the heart is markedly rotated to the left.
The pericardium is a fibrous sac that surrounds the majority of the heart as it only partially covers the left atrium. The pericardium is anchored in the midline anteriorly to the sternum by superior and inferior ligaments and is also anchored to the diaphragm. The pericardial sac is composed of two layers, the visceral and parietal layers. The visceral pericardium lies on the surface of the myocardium whereas the parietal pericardium is a continuation of the visceral pericardium that is folded on top of the visceral pericardium. The space between these two layers of pericardium is referred to as the pericardial space. The surfaces of the visceral and parietal pericardium that define the pericardial space are lined with a single layer of mesothelial cells. The cells of the parietal pericardium secrete a transudative fluid into the pericardial space. Normally the pericardial space contains 15 to 50 ml of fluid. Because of its relatively low compliance the rapid accumulation of even a couple hundred milliliters of fluid can have significant physiologic consequences. On the other hand, if fluid accumulates gradually the compliance of the pericardium will increase slowly, allowing the build-up of a significant volume of fluid.
The normal function of the pericardium is to prevent the spread of infection or inflammation by direct extension from adjacent structures in the mediastinum. It my also help reduce friction between the beating heart and neighboring structures in the chest, although patients with isolated congenital absence of the pericardium do not usually develop pathology secondary to the heart abutting adjacent structures. Pathologic processes that affect the pericardium can be considered in the following categories: pericardial effusion, acute pericarditis, constrictive pericarditis, pericardial masses, and other rare conditions.
Cardiac magnetic resonance (CMR) imaging is a noninvasive imaging modality that is ideally suited for imaging the pericardium because of its large field of view and ability to differentiate soft tissue based on fat and water composition. Transthoracic echocardiography (TTE) is the workhorse of noninvasive cardiac imaging because of its portability allowing bedside exams, relatively low cost allowing widespread use, and excellent temporal resolution allowing independence from ECG gating for image acquisition. However, TTE is dependent on having an acceptable acoustic imaging window. Furthermore, the field of view is limited with TTE. It can be challenging to image posterior structures in large patients. Finally TTE cannot readily differentiate soft tissue planes unless a layer of fluid or air separates the planes. Discerning tissue type is almost impossible with TTE unless the anatomy is normal or a known variant. The pericardium itself is not well visualized with TTE.
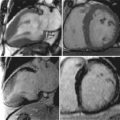
With CMR normal pericardium appears as a rim of low signal intensity on T1- and T2-W imaging as the pericardium is largely a fibrous structure with low water content. Normal pericardium is less than 3 mm thick. Autopsy studies describe the normal pericardium to be between 0.4 and 1.0 mm thick. Sechtem et al. have reported that the average normal pericardium thickness by CMR was 1.9 ± 0.6 mm. This discrepancy may be caused by fibrous tissue that was not measured at autopsy and nonlaminar motion of the pericardial fluid resulting in an artificially thick line. By CMR normal pericardium is considered less than or equal to 3 mm thick. A pericardium seen on CMR greater than 3 mm thick is indicative of pathology.
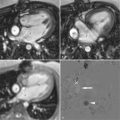
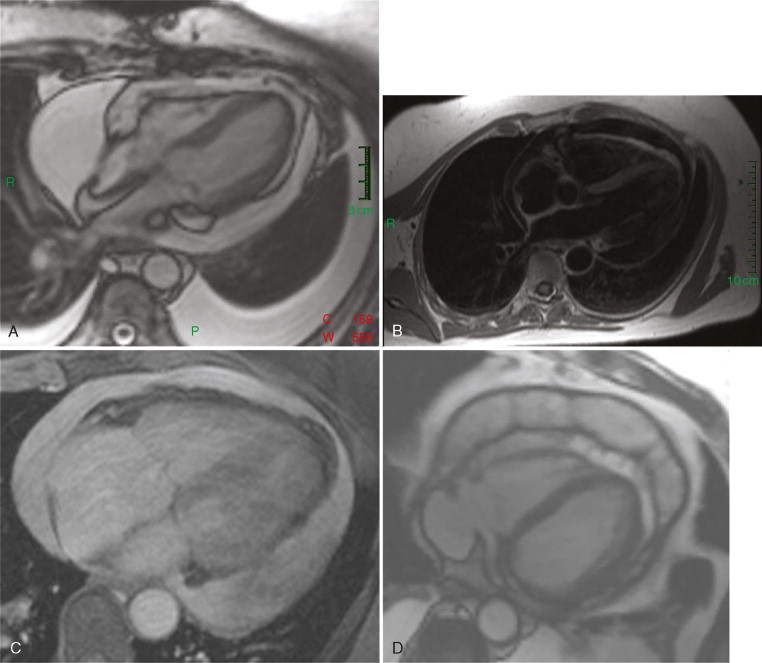
A 68-year-old woman with a history of breast cancer presented to the emergency department with a 1-week history of progressive dyspnea and lightheadedness. Her physical exam was remarkable for tachypnea, tachycardia with a blood pressure of 86/50, Kussmaul’s sign, and a pulsus paradoxus of 25 mm Hg. She was given IV fluids and pericardiocentesis was performed. Two days later her symptoms began to recur and a CMR was ordered ( Figure 9-1 ).
Comments
The differential for the etiology of pericardial effusion is broad. Common cases include neoplasm, infection (bacterial, including tuberculosis, and viral), uremia, and cardiac injury from myocardial infarction or trauma. If a pericardial effusion is sufficiently large to impair cardiac filling causing a reduction in cardiac output, then tamponade physiology is present. This is a clinical diagnosis and CMR should not be used to make this diagnosis but to corroborate it. However, if the presence of a pericardial effusion is suspected in a hemodynamically stable patient, then CMR may have diagnostic utility.
TTE is the most frequently utilized imaging modality for the visualization of pericardial effusion. This is largely because of the convenience of a bedside exam and the relatively high sensitivity and specificity of diagnosing pericardial effusion with TTE. However, loculated pericardial effusions may be difficult to appreciate on TTE. Furthermore in the immediate period after cardiac surgery, air in the pleural space may reduce or eliminate acoustic windows. CMR provides a large field of view allowing localization of loculated effusions ( Figure 9-1 D ). Furthermore CMR is not limited by air within structures of interest.
Spin echo (SE) and gradient echo (GRE) sequences can be utilized to differentiate pericardial effusions. Hemorrhagic effusions are characterized by high intensity signal on T1-weighted SE images and by low intensity signal on GRE images. Nonhemorrhagic effusions are characterized by a low signal intensity of T1-weighted SE and a high signal intensity of GRE images. Malignant effusions may be accompanied by a heterogeneously thick pericardium that may have a nodular appearance.
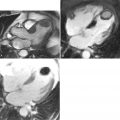
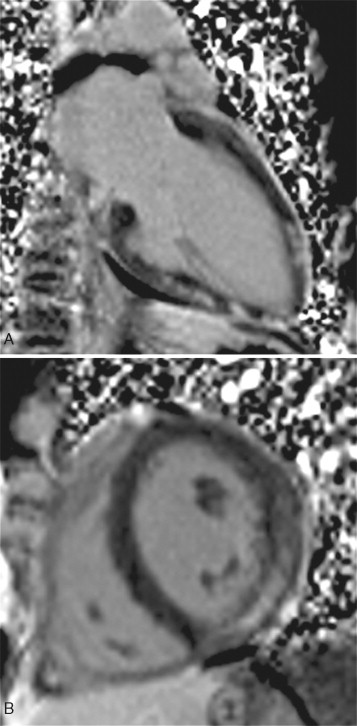
A 64-year-old man in the cardiac care unit (CCU) with congestive heart failure (CHF) secondary to viral myocarditis developed severe chest pain. The pain was relieved by sitting upright and leaning forward and exacerbated by lying supine. The ECG was remarkable for diffuse ST elevation and PR depression. The patient was referred for CMR imaging ( Figure 9-2 ).
Comments
Acute pericarditis is the syndrome of the signs and symptoms caused by pericardial inflammation that lasts less than 2 weeks. The differential diagnosis for the etiology is large ( Table 9-1 ). Many cases of acute pericarditis may actually be myopericarditis as the inflammation involves the myocardium as well as the pericardium. Dressler syndrome is the classic manifestation of this process. Twenty-four to seventy-two hours after a large transmural myocardial infarction a patient may experience local inflammation on the epicardial surface of the infracted myocardium and the adjacent pericardium, which produces the clinical signs and symptoms of acute pericarditis. Viral myocarditis may also manifest as myopericarditis. The hallmark of this condition is elevated biomarkers such as Troponin I, in the context of the signs and symptoms of pericarditis.
|
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree