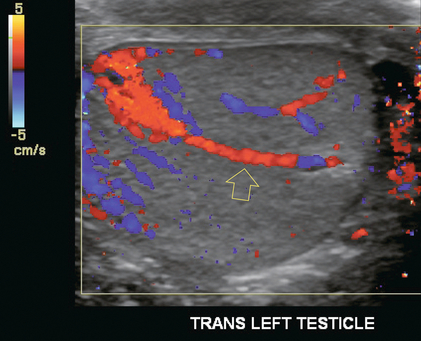
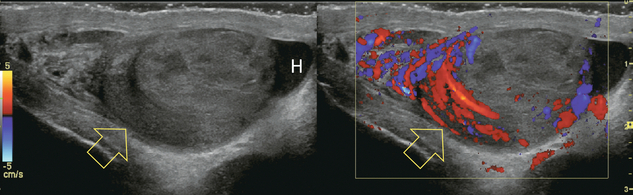
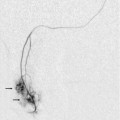
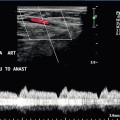
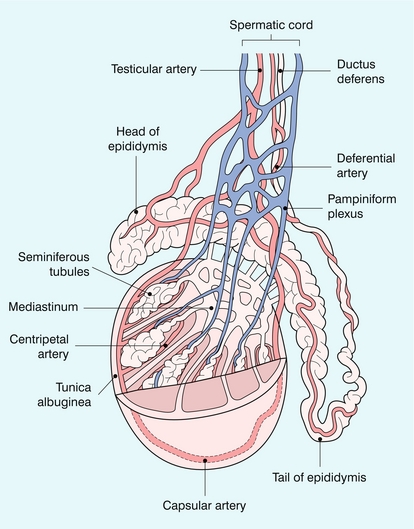
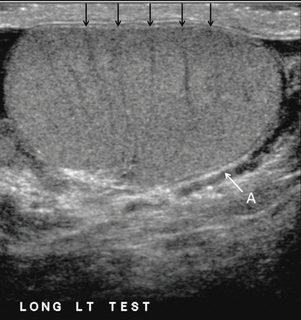
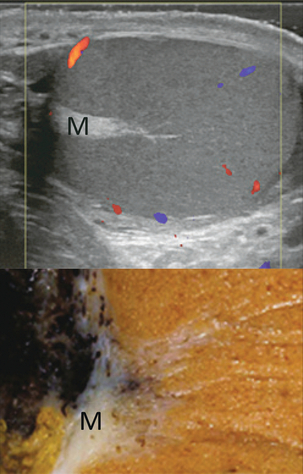
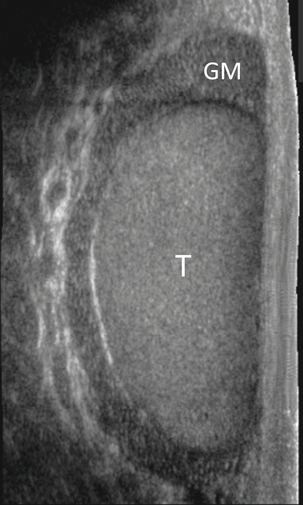
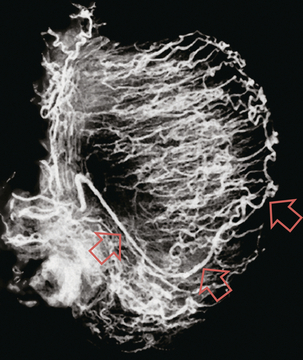
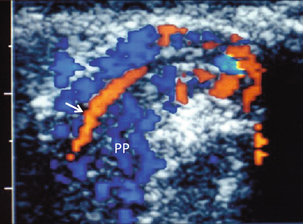
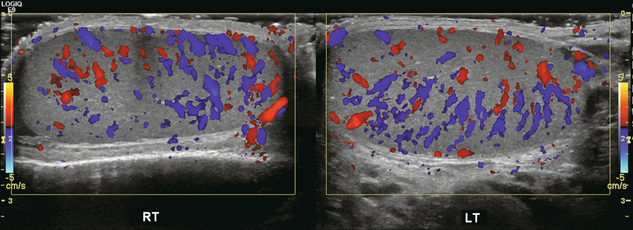
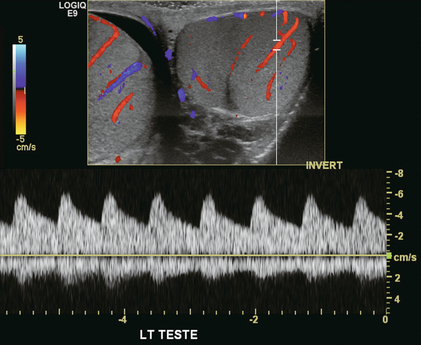
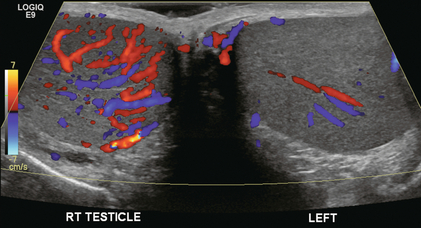
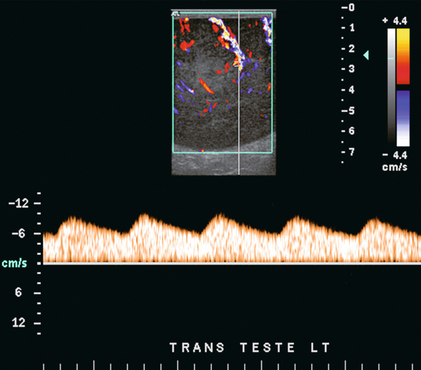
Chapter 13 The normal adult testis is an egg-shaped gland which is approximately 3–5 cm in length and 2–4 cm in width and thickness with a volume of 4 cm3. Testicular size varies with age and stage of sexual development. The surface of the testicle is covered by the tunica albuginea, a thin, dense, inelastic fibrous capsule. Just within the tunica albuginea is the tunica vasculosa, through which the branches of the testicular artery course before entering into the gland (Fig. 13-1). Numerous thin septations (septula) arise from the tunica albuginea and create 250–400 cone-shaped lobules containing the seminiferous tubules (Fig. 13-2). These tortuous tubules course towards the mediastinum of the testis (Fig. 13-3) and progressively merge to form larger ducts known as tubuli recti. These, in turn, join with each other to form a network of epithelium-lined spaces embedded in the fibrostroma of the mediastinum called the rete testis. These continue as 10–15 efferent ductules which pass into the head of the epididymis. FIGURE 13-1 The testicular artery supplies flow to the epididymis and the testicle. As the testicular artery courses through the spermatic cord, it is surrounded by the pampiniform plexus of veins. The capsular artery, a branch of the testicular artery, courses just beneath the tunica albuginea. The centripetal arteries are branches of the capsular artery and course between the septa supplying the testicular parenchyma. FIGURE 13-2 Longitudinal view of a normal testicle. The testicle is of relatively uniform echogenicity. On this high-resolution image, the testicular septa can be perceived as fine lines dividing the parenchyma into the individual lobules (black arrows). Note the echogenic tunica albuginea surrounding the testicle (A). FIGURE 13-3 Transverse view of the left testicle compared to a specimen in the region of the mediastinum testis. The mediastinum testis is an echogenic fibrous structure. It is the point of confluence of the efferent ductules and the septa. Note its triangular echogenic appearance on the ultrasound (M) and how well it correlates with the dense white fibrous stroma on the specimen. The epididymis is a comma-shaped structure that runs along the posterolateral aspect of the testis (Fig. 13-4). The head of the epididymis is located next to the upper pole of the testis and receives the efferent ductules. The ductules eventually converge through the body and tail and form the vas deferens which continues on in the spermatic cord. Along with the vas, the cord also contains the testicular artery, cremasteric artery, differential artery, the pampiniform plexus of veins, the genitofemoral nerve and lymphatic channels. FIGURE 13-4 Longitudinal view of a normal right scrotum. The slightly hypoechoic epididymis wraps as a comma shape around the testis (T). The larger more prominent area is known as the globus major or the head of the epididymis (GM). The right and left testicular arteries originate from the aorta just below the renal arteries. They course through the deep inguinal ring to enter the spermatic cord, accompanied by the cremasteric and deferential arteries, which supply the soft tissues of the scrotum, epididymis and vas deferens. The testicular artery penetrates the tunica albuginea along the posterior aspect of the testis and gives off capsular branches which course through the tunica vasculosa. These capsular branches then give rise to the centripetal arteries which carry blood from the capsular surface, centrally towards the mediastinum along the septula (Figs 13-5 and 13-6). Branches of the centripetal arteries then course backward towards the capsular surface; these are known as recurrent rami. In about 50% of testes, a more robust artery can be seen passing directly from the testicular artery at the mediastinum into the parenchyma, known as the transtesticular artery (Fig. 13-7). It is occasionally accompanied by a vein. Small anastomoses do exist between the testicular artery, cremasteric, and differential arteries. Branches of the pudendal artery may also supply the scrotal wall. FIGURE 13-5 Microangiogram of the testicle. A prominent capsular branch (open arrows) courses from the testicular artery under the capsular surface. Centripetal arterial branches emanate from it and pass into the lobules of the testicle. Image courtesy of Dr. Thomas Winter. FIGURE 13-6 3-D rendered power Doppler acquisition of a testicle. With very sensitive Doppler settings, the network of centripetal arteries is seen fanning across this normal testicle. Testicular venous outflow courses mostly through the mediastinum testis, into the spermatic cord, and eventually up through the inguinal canal. As the veins exit the scrotum through the spermatic cord, they form a web-like network that surrounds the testicular artery, known as the pampiniform plexus (Fig. 13-8). This plexus functions as a heat exchange mechanism, pulling warmth away from the testicular arterial inflow, thereby helping to maintain spermatogenesis at a lower, more optimal temperature. FIGURE 13-8 The pampiniform plexus of veins (PP) is a web-like collection of vessels surround the testicular artery (arrow) as it passes through the spermatic cord. On the left side, the testicular vein usually drains into the left renal vein; on the right side, drainage is directly into the inferior vena cava just below the right renal vein. The testicular veins normally have valves that prevent retrograde flow of venous blood to the scrotum, but if they are absent or become incompetent; this predisposes to development of a varicocele. This results in compromise of the heat exchange mechanism and, therefore, is a frequent cause of infertility.1 Ultrasound examination of the scrotum is not complete without the application of colour Doppler. The procedure is a mandatory part of the imaging evaluation to confirm the presence (or absence) of uniform, symmetric vascular perfusion of the testes and epididymides (Fig. 13-9). The settings for colour Doppler scanning must be optimised for low-volume and low-velocity flow. If colour noise is excessive at low-flow settings and interferes with the examination, colour artefact can be decreased by properly adjusting gain and pulse repetition frequency settings. Temporal resolution can be improved by minimising the overall image size and zooming in to the area of interest. Additionally decreasing depth, limiting the number of focal zones and limiting the size of the colour box will also improve temporal resolution. Use of appropriate technical parameters should assure demonstration of intratesticular vascularity in all normal adult patients. In the prepubertal age group, intratesticular flow volume is less and therefore more difficult to identify. The application of power Doppler may be helpful. Spectral Doppler can assess arterial and venous waveforms and quantify velocities (Fig. 13-10), but its application in the scrotum is relatively limited, except for a few conditions such as partial torsion or extreme swelling secondary to inflammation predisposing to ischaemia. In those cases spectral Doppler can best identify the high resistance to arterial inflow caused by venous outflow compromise or parenchymal congestion. FIGURE 13-9 Transverse colour Doppler ultrasound image of both testicles using a split screen format. Note the symmetry of echotexture and the uniformity and symmetry of colour flow. FIGURE 13-10 Spectral Doppler waveform of a normal left testicular artery. The resistive index is normal (approx. 60%). Colour Doppler should reveal bilaterally symmetric and relatively uniform flow through both testes and epididymides (Fig. 13-9). A fan-like array of the centripetal arteries should be present through the testicles (Fig. 13-6). Spectral Doppler tracings of testicular arterial inflow demonstrate relatively low resistance (Fig. 13-10); this is in contrast to the cremasteric and deferential arteries which have relatively high resistance to flow. The normal testicular artery resistive indices in adults range from 0.46 to 0.78, with a mean of 0.64. Similar findings are reported in the intratesticular arteries of postpubescent boys, with resistive indices ranging from 0.48 to 0.75 (mean, 0.62). In prepubertal boys, however, resistance is higher to the point that diastolic arterial flow may not be detectable. Supratesticular arteries to the vas deferens or cremaster muscle, on the other hand, have higher impedance with low-diastolic flow and resistive indices ranging from 0.63 and 1.0, with a mean of 0.84. Pulsed Doppler is relatively insensitive in detecting arterial flow in prepubescent patients. In contrast, power Doppler has been shown to reveal arterial flow in 92% of testes in pre-pubescent patients, while colour Doppler demonstrates flow in 83% of cases. In postpubescent patients, both Doppler imaging techniques demonstrated flow in 100% of cases.2,3 The cause of the infection varies with age.4 In adult patients less than 35 years of age, Chlamydia trachomatis and Neisseria gonorrhoeae (sexually transmitted organisms) are the most common agents. In prepubertal boys and in men over 35 years of age, the disease is most frequently caused by Escherichia coli and Proteus mirabilis. Cytomegalovirus is the most common agent in the immunocompromised patient. In most normal paediatric patients, a bacterial pathogen is not isolated and the inflammation is presumed to be viral in nature. Those patients who have an underlying urogenital congenital anomaly are prone to infection from Gram-negative bacteria.5 The sonographic appearance of epididymo-orchitis varies depending on the stage of the process. The sensitivity of greyscale sonography for detecting epididymo-orchitis is reported to be about 80%. In the early, acute stage, the epididymis and/or testicle will be enlarged and hypoechoic. With the onset of tissue breakdown and haemorrhage, the echogenicity begins to increase. There may be reactive thickening of the scrotal wall. A hydrocele may be present and it may contain debris. If allowed to progress, microabscesses may develop and the appearance becomes more complex and variable. Further progression can lead to frank intra- or extratesticular abscess, ischaemia and eventually necrosis. Scarring associated with chronic orchitis typically results in a small hyperechoic testis.6 The diagnosis of infection and inflammation typically hinges on the identification of hyperaemia by colour Doppler – an asymmetric appearance with more robust flow (an increased number and prominence of discernible vessels) in association with an enlarged, painful, hypoechoic epididymis and/or testis (Fig. 13-11). Several studies have demonstrated sensitivity and specificity for the diagnosis of scrotal inflammatory disease by colour Doppler sonography approximating 100%. Early in the inflammatory process, vasodilatation and hyperemia results in a low-resistance flow pattern on spectral Doppler (Fig. 13-12).7 FIGURE 13-11 Transverse colour Doppler image of the scrotum including both testicles. This paediatric patient presented with right scrotal pain. Colour Doppler reveals marked hyperaemia of the right testicle and obvious asymmetry in perfusion. The diagnosis is right orchitis. FIGURE 13-12 Spectral Doppler tracing obtained in an area of focal orchitis. Flow during diastole is relatively brisk, resulting in a resistive index of less than 50%. This is seen relatively early in the inflammatory process before the accumulation of significant oedema and/or pus. In cases of severe epididymitis, periepididymal swelling may obstruct testicular venous outflow, leading to testicular ischaemia or infarction. An enlarged, heterogeneous testicle with reduced or absent colour flow and a contiguous abnormal epididymis may be seen on greyscale imaging (Fig. 13-13). Hyperaemia of the epididymis helps to differentiate testicular ischaemia following inflammation from that caused by torsion. A high-resistance waveform, along with decreased or reversed diastolic flow, may be seen on the spectral Doppler tracing and suggests venous infarction8 (Fig. 13-14).
Doppler Imaging of the Scrotum
Testicular Anatomy
TESTICULAR ARTERIAL ANATOMY

TESTICULAR VENOUS ANATOMY
Technique
DOPPLER TECHNIQUE
NORMAL ULTRASOUND AND DOPPLER FINDINGS
Acute Scrotal Pain
INFLAMMATORY DISEASE
Doppler Imaging of the Scrotum