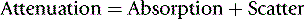Chapter 10 Effects of radiation
 A beam of X-ray photons is gradually attenuated as it passes through matter by being absorbed and scattered.
A beam of X-ray photons is gradually attenuated as it passes through matter by being absorbed and scattered. The most important interactions that result in attenuation are photoelectric absorption and Compton scatter.
The most important interactions that result in attenuation are photoelectric absorption and Compton scatter. Photoelectric absorption is more likely to occur in dense materials with atoms of high proton number.
Photoelectric absorption is more likely to occur in dense materials with atoms of high proton number. Photoelectric absorption is responsible for differential absorption in the body’s tissues that results in a radiographic image.
Photoelectric absorption is responsible for differential absorption in the body’s tissues that results in a radiographic image. Compton scatter occurs alongside photoelectric absorption and is the dominant process above a setting of 75 kV.
Compton scatter occurs alongside photoelectric absorption and is the dominant process above a setting of 75 kV. Attenuation in the body’s tissues with the transfer of energy and subsequent ionisation may result in deterministic or stochastic biological effects.
Attenuation in the body’s tissues with the transfer of energy and subsequent ionisation may result in deterministic or stochastic biological effects. Radiation protection regulations aim to reduce the risk of stochastic effects and avoid deterministic effects.
Radiation protection regulations aim to reduce the risk of stochastic effects and avoid deterministic effects. Effective dose measured in sieverts (Sv) takes into account different types of radiation and their damaging potential on different tissues and organs in the body.
Effective dose measured in sieverts (Sv) takes into account different types of radiation and their damaging potential on different tissues and organs in the body. X-ray photons produce fluorescence in phosphors, a property utilised in some image recording systems.
X-ray photons produce fluorescence in phosphors, a property utilised in some image recording systems.ATTENUATION
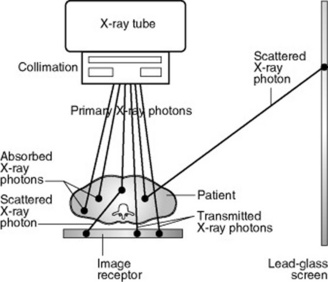
Attenuation is partly due to some X-ray photons being totally absorbed and partly to the energy of some X-ray photons being partially absorbed while the remainder is scattered in various directions (Fig. 10.1). Some X-ray photons are transmitted through the material unchanged, without interacting with any atoms.
ATTENUATION AND THICKNESS OF MATERIAL
The number of X-ray photons transmitted compared to the number attenuated in any particular type of material depends on the thickness of that material. In general, the thicker the material, the greater the attenuation. However, it is not a linear relationship where the same numbers of X-ray photons are attenuated in an equal thickness of material, but an equal percentage is attenuated in equal thickness. For example, 20% of photons may be attenuated in the first centimetre of material, then 20% of what is left in the second centimetre, and 20% of the remainder in the third centimetre, etc. This is called an exponential relationship and the percentage attenuated in each thickness is known as the linear attenuation coefficient (LAC or μ) for the specific material. This relationship is used in practice during quality control checks on X-ray equipment to measure the half value thickness/layer (HVT or HVL) of an X-ray unit. The measurement gives the thickness of aluminium that will attenuate 50% of the X-ray photons at a specific kV setting. This gives an indication of the penetrating power of the beam: the thicker the aluminium required to attenuate half of the beam, the more penetrating it is and it can be related (using published tables) to the total filtration present in the beam (see pp. 100, 107).
PHOTOELECTRIC ABSORPTION
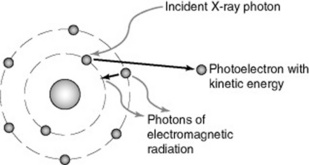
Photoelectric (PE) absorption occurs when an X-ray photon interacts with a bound electron, usually in the inner shell of an atom, when its energy exceeds the binding energy of the electron (Fig. 10.2). The atom may be in the patient (an atom of calcium in bone, for example) or it might be an atom of carbon in the carbon-fibre tabletop; or an atom of lead in the lead-glass screen.
THE IMPLICATIONS OF PHOTOELECTRIC ABSORPTION IN PRACTICE
The main implications to consider are:
The radiographic image
Bone has a fairly high density and contains atoms of calcium and phosphorus, giving an effective proton number (Zeff) of approximately 12. Soft tissues, such as muscle and fat, are lower in density and contain atoms of lower proton number (particularly carbon, hydrogen and oxygen), giving an effective proton number of approximately 7. Air is primarily nitrogen and oxygen, giving a similar effective proton number to soft tissue, but has very low density (Table 10.1).
Table 10.1 The principal constituents of human tissue1
| Material | Density (kg m−3) | Effective proton number |
|---|---|---|
| Bone | 1700 | 12.3 |
| Soft tissue (muscle) | 1000 | 7.6 |
| Soft tissue (fat) | 900 | 6.5 |
| Air | 1 | 7.8 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree