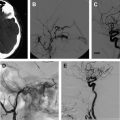
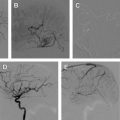
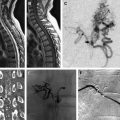
Carotid occlusive disease is one of several etiologic factors for stroke. Of all strokes, an estimated 88% are ischemic in nature. Less than 20% of these are caused by atheroma in the carotid bifurcation. Traditionally, carotid artery stenosis has been treated with carotid endarterectomy (CEA); however, carotid artery balloon angioplasty and stent placement has enjoyed significant technological advances over the last decade and can now offer a comparable treatment alternative to CEA. In this review, the authors concentrate their discussion on the treatment of carotid atherosclerotic disease with particular attention on the endovascular treatment.
Key points
- •
Patients who have severe (>70%) symptomatic carotid stenosis have up to a 20% risk of an ipsilateral stroke over the following 3-month time period, with 30% to 35% risk of ipsilateral stroke over 2 to 3 years’ time when treated with optimum medical management.
- •
The degree of carotid stenosis by ultrasound should be reported based on the Society of Radiologists in Ultrasound’s consensus conference in 2003.
- •
Carotid artery balloon angioplasty and stent placement (CAS) has a higher incidence of perioperative stroke when compared with carotid endarterectomy (CAE); however, there is a decreased incidence of myocardial infarction, infection, and cranial nerve injury.
- •
Patients that are ideal for CAS include patients that have a high surgical risk, such as patients with prior neck irradiation, aberrant neck anatomy, contralateral recurrent laryngeal nerve injury, prior ipsilateral CEA, significant coronary artery disease, high cervical stenotic lesion location, and tracheostomy.
- •
Patients with type II and III aortic arches, tortuous common carotid artery (CCA), or young patients with otherwise normal anatomy and a low-lying cervical lesion location precluding mandibular disarticulation are best served with CEA.
Introduction
In the United States, 150,000 patients die and 600,000 suffer significant morbidity each year as a result of a cerebrovascular accident (CVA), making stroke the second most common cause of death in the United States. An estimated 88% of these are ischemic strokes, with 15% attributed to extracranial carotid occlusive disease. Endovascular therapy for the treatment of carotid atherosclerotic disease has advanced over the last decade and is now considered to be a viable alternative to carotid endarterectomy (CEA) in appropriately selected patients. Carotid artery balloon angioplasty and stent placement (CAS) during its infancy was associated with higher rates of perioperative ischemic complications when compared with CEA. However, lower rates of other perioperative complications were noted, including myocardial infarctions and infection. As a result of increased experience with the procedure and improvements in technology specifically designed for CAS, the rates of periprocedural ischemic complications have diminished. Today, the rate of complications from CEA versus CAS are nearly equivocal, making the choice of therapy based more on clinical presentation, medical comorbidities, and carotid artery anatomy. In this review, the authors concentrate their discussion on the treatment of carotid atherosclerotic disease with particular attention on the endovascular treatment.
Introduction
In the United States, 150,000 patients die and 600,000 suffer significant morbidity each year as a result of a cerebrovascular accident (CVA), making stroke the second most common cause of death in the United States. An estimated 88% of these are ischemic strokes, with 15% attributed to extracranial carotid occlusive disease. Endovascular therapy for the treatment of carotid atherosclerotic disease has advanced over the last decade and is now considered to be a viable alternative to carotid endarterectomy (CEA) in appropriately selected patients. Carotid artery balloon angioplasty and stent placement (CAS) during its infancy was associated with higher rates of perioperative ischemic complications when compared with CEA. However, lower rates of other perioperative complications were noted, including myocardial infarctions and infection. As a result of increased experience with the procedure and improvements in technology specifically designed for CAS, the rates of periprocedural ischemic complications have diminished. Today, the rate of complications from CEA versus CAS are nearly equivocal, making the choice of therapy based more on clinical presentation, medical comorbidities, and carotid artery anatomy. In this review, the authors concentrate their discussion on the treatment of carotid atherosclerotic disease with particular attention on the endovascular treatment.
Clinical presentation
Carotid artery stenosis in patients is usually discovered after an ischemic event (either a transient ischemic attack [TIA] or a permanent stroke). The remainder of carotid artery occlusive disease is usually discovered after an initial abnormality is revealed on physical examination (ie, carotid bruit), which is then confirmed on imaging studies. The most common ischemic stroke syndrome is a middle cerebral artery occlusion resulting in contralateral hemiparesis (arm more than leg) and hemisensory loss as well as aphasia depending on hemispheric dominance. Other TIA symptoms can include ocular symptoms, such as amaurosis fugax, which is most commonly described as a shade being pulled over one’s eye on the ipsilateral side. Unfortunately, in many patients’, carotid atherosclerotic disease is discovered after a large stroke has occurred.
Epidemiology and natural history
Approximately 88% of strokes have an ischemic cause, with approximately 15% being secondary to carotid occlusive disease. Risk factors for stroke of all types include increasing age, male sex, hypertension, smoking history, diabetes, obesity, hypercoagulable states, African American race, and presence of carotid artery atherosclerosis. The prevalence of carotid stenosis (>50%) in patients 65 years of age or older is estimated at 5% to 9%. In patients without symptoms of cerebral ischemia (TIA, stroke, amaurosis fugax) with carotid stenosis (>60%), the risk of stroke has been cited between 2% and 5% annually, with a 2.2% per year risk of ipsilateral stroke when treated with maximum medical management. Alternatively, patients who have severe (>70%) symptomatic carotid stenosis have up to a 20% risk of an ipsilateral stroke over the following 3-month time period, with a 30% to 35% risk of ipsilateral stroke over 2 to 3 years time when treated with optimum medical management. For symptomatic patients with moderate stenosis (50%–69%), the risk of stroke on medical management is 15% to 20% over a period of 3 years.
The likelihood of stroke has more recently been shown to be higher in patients with an echolucent plaque on ultrasound, suggesting that plaque morphology in addition to the degree of stenosis plays a role in the risk of stroke. An ulcerated atherosclerotic plaque on imaging studies also has been shown to have a higher likelihood of becoming symptomatic when compared with a nonulcerated, smooth atherosclerotic plaque. Furthermore, patients who have suffered a stroke of any cause are also 2 to 3 times as likely to have a myocardial infarction. It has also been shown that the degree of carotid stenosis is directly related to the amount of cerebral collateralization. This finding implies that a hemodynamically significant carotid lesion causing cerebral hypoperfusion recruits arterial collateral vessels to the territory affected. The annual risk of a subsequent stroke in any cerebral territory following an occlusion of the carotid artery is high at 5.5% according to a meta-analysis of 20 follow-up studies in patients with TIA or a minor ischemic stroke; however, interestingly, the risk of subsequent ipsilateral stroke following such an occlusion is only 2.1% per year. The hypothesis being that the development of collaterals in the ipsilateral hemisphere is facilitated by worsening vessel stenosis caused by the incriminating carotid plaque over a period of time, which then offers protection against further ischemia once the carotid artery becomes occluded. Recent long-term data have shown that this risk of stroke is highest in the first 18 months following occlusion and then drops off dramatically to an average annual rate of only 2.4% for all types of stroke at the 10-year follow-up. Interestingly, cerebral leptomeningeal collateralization increased the risk of recurrent ipsilateral ischemic events. The implication being that in the first 18 months following carotid vessel occlusion, the collateral circulation is not completely established and any hypoperfusion episode can potentially precipitate an ischemic event; however, beyond this period the risk drops off significantly because of the establishment of a new equilibrium in the cerebral perfusion. The apparent fallacy demonstrated in the long-term follow-up study from Netherlands showing leptomeningeal collateral presence predicting increased risk of subsequent ischemia merely reveals the delicate balance of cerebral perfusion in the midst of progressively occluding carotid vessel.
Anatomy and pathophysiology
In most patients, the right common carotid artery (CCA) arises from the brachiocephalic artery, whereas the left CCA typically arises directly from the aortic arch. At approximately the level of the superior border of the thyroid cartilage or the inferior edge of the hyoid bone, the carotid artery bifurcates into the internal carotid artery (ICA) and the external carotid artery (ECA). In one cadaveric study, 73% of specimens’ bifurcations were above the thyroid cartilage and 63% were below the hyoid bone, which typically correlates to approximately the midportion of the vertebral body of C3. At the bifurcation, there is a dilation referred to as the carotid bulb. On average, the outer diameter of the proximal ICA is 8.1 mm, whereas the carotid bulb is 12.8 mm. Distal to the bifurcation, the ICA then narrows to an average diameter of 6.1 mm. These average numbers are helpful when choosing stent and balloon sizes during endovascular cases as well as when using Fogarty balloons and shunts in CEA.
The cervical ICA proceeds into the carotid foramen, entering the petrous portion of the temporal bone. After exiting the petrous temporal bone, the ICA swings anteriorly in the cavernous sinus before then bending posteriorly as it exits the cavernous sinus to become an intracranial vessel. The ICA then continues to terminate into the middle cerebral artery and the anterior cerebral artery.
Atherosclerotic changes of the carotid artery typically extend from the carotid bulb into the ICA. There is a dramatic change in vessel diameter from approximately 8 mm in the CCA to a 4–5 mm ICA.
Atherosclerotic lesions tend to occur at areas experiencing low endothelial shear stress which are local wall stresses that are generated by patterns of blood flow, such as the carotid bifurcation region. Low shear stress promotes atherosclerosis by a variety of mechanisms, including impairment of endothelial function by downregulation of the endothelial isoform of nitric oxide synthase (e-NOS) and upregulation of endothelin-1, increased endothelial uptake of low-density lipoprotein (LDL), promotion of oxidative stress, and increased plaque thrombogenicity. Low endothelial shear stress also enhances the proatherogenic effect of chronic inflammation because it allows for the attachment and infiltration of inflammatory cells via the activation of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB) and subsequent upregulation of adhesion molecules, chemokines, and proinflammatory cytokines. Low endothelial shear stress has also been implicated in the transition of stable atherosclerotic lesions to vulnerable plaques resulting in acute coronary syndromes. As the diameter of the vessel narrows because of plaque buildup, it can cause reduction of blood flow and, hence, ischemic changes. However, more importantly, most TIAs and stroke occur from ulceration of the atherosclerotic plaque with distal embolization into the intracranial circulation. Finally, with plaque rupture, a vigorous local coagulation cascade may be initiated and a platelet plug may form and occlude the entire ICA at the area of the lesion or send distal emboli into the intracranial circulation.
Diagnostic imaging features
Many diagnostic imaging modalities may be used to help confirm the diagnosis of carotid occlusive disease. Carotid duplex ultrasonography is often the first test performed when the clinician suspects carotid stenosis. The duplex ultrasound examination includes a quantitative measurement of the degree of arterial narrowing using a brightness mode (B-mode technique) as well as a spectral analysis to determine the measurement of blood flow velocity across the lesion. This examination provides 2 pieces of information for the ultrasonographer to interpret. The degree of carotid stenosis should be reported based on the Society of Radiologists in Ultrasound’s consensus conference in 2003:
- •
Normal: ICA peak systolic velocity (PSV) less than 125 cm/s and no plaque or intimal thickening is visible
- •
Less than 50% stenosis: ICA PSV less than 125 cm/s and plaque or intimal thickening is visible
- •
50% to 69% stenosis: ICA PSV is 125 to 230 cm/s and plaque is visible
- •
Greater than 70% stenosis to near occlusion: ICA PSV greater than 230 cm/s and visible plaque and lumen narrowing are seen
Ultrasound is, however, a limited assessment of only the extracranial carotid vessels and results can vary based on artifact induced by calcific plaque within the lesion. Carotid ultrasound has been shown to often overestimate the degree of stenosis when compared with the gold standard measurements of diagnostic carotid angiography. Ultrasound technology has continued to improve over the last 30 years; however, it remains inferior to computed tomography angiography (CTA) and magnetic resonance angiography (MRA). Therefore, after this noninvasive study is performed, a confirmatory study, such as a CTA or MRA, is needed to determine the further management of patients.
CTA is performed by intravenous injection of iodinated contrast. Axial images are obtained as the contrast density peaks in the arterial phase. Sagittal, coronal, and multiplanar 3-dimensional (3D) images can be formatted based on the axial images obtained to help measure the intraluminal diameter at the narrowest portion of the proximal ICA and across the area of the ICA that is above the stenosis that is thought to be normal. The degree of stenosis is then calculated based on the North American Surgical Carotid Endarterectomy Trial (NASCET) criteria as a percentage. CTA accuracy exceeds 95% and is limited largely by technical factors alone. Patient motion and poor cardiac function can decrease the accuracy of CTA because of the motion artifact and suboptimal luminal contrast related to compromised cardiac function. Furthermore, any venous contrast contamination can make the images more difficult to interpret. Additionally, the contrast load administered is not insignificant and must be taken into consideration in patients with renal insufficiency or those on hemodialysis. Patients with iodinated contrast allergies may also require special consideration and might need a steroid preparation before the administration of contrast.
MRA offers another noninvasive approach to measure the degree of carotid stenosis. The 2 main MRA techniques to assess carotid stenosis are (1) 2-dimensional and 3D multiple overlapping thin slab angiography (MOTSA) time-of-flight (TOF) without the use of gadolinium contrast and (2) contrast-enhanced MRA with rapid injection of gadolinium to produce axial images through the neck from the aortic arch to include the circle of Willis. These images, much like CTA, can then be formatted into 3D images and the stenosis can be calculated in a similar manner according to NASCET criteria as described later. MR using TOF sequences is especially useful in patients with either contrast allergies or renal insufficiency because contrast (gadolinium) is not required to obtain these images with the caveat that flow-dependent TOF MRA readily lends itself to artifacts related to flow phenomena exaggerating stenosis. Turbulence can create artifacts on the TOF images that can overestimate stenotic lesions. Additionally, intracranial images can be obtained at the same time to assess for prior cerebral vascular accident (CVA) or additional intracranial pathologies; the intracranial (as well as the extracranial) circulations can also be assessed, making it a superior test to carotid ultrasound. However, MR is contraindicated in patients with metal implants, such as pacemakers. The sensitivity and accuracy of MRA is up 92% to 97%, making it an acceptable and sensitive noninvasive test.
Diagnostic cerebral angiography remains the gold standard to determine the degree of carotid artery stenosis of both the extracranial and intracranial circulation and to determine the optimal method of treatment. This invasive study carries an approximately 0.5% risk of significant morbidity and mortality, with the most notable complications being arterial dissection, stroke, and retroperitoneal hematoma. Additionally, the amount of iodinated contrast used varies but could represent a relative contraindication in patients with renal insufficiency, much like with CTA.
The degree of stenosis can be calculated in 2 ways: (1) using the maximal ICA stenosis/ICA normal caliber above the stenosis ratio or (2) using the maximal ICA stenosis/ICA outer to outer wall caliber ratio at the site of the stenosis. The former was used in the NASCET criteria and the latter in the European Carotid Surgery Trial (ECST) criteria. The other 2 methods are the common carotid criteria (CC) and the Carotid Stenosis Index (CSI) method ( Fig. 1 ). There are some discrepancies and inconsistencies in the measurements obtained by the NASCET and ECST criteria. The criteria are based on indirect ratios where there is estimation of the outer wall diameter of the ICA at the level of the stenosis. Both the above methods are based on angiographic measurements, which make it impossible to directly measure the outer wall of the ICA at the level of the stenosis. These variables in the measurements result in wide disparity in stenosis estimates by NASCET or ECST criteria. Studies show that measurement of 50% stenosis by NASCET is equivalent to 70% by ECST. The numerator both in NASCET and ECST is the direct measurement of the stenosis at its narrowest location. As for the denominator NASCET uses the diameter of the normal distal ICA, while ECST estimates the unseen outer wall diameter of the ICA at the carotid bulb stenosis. The diameter of the carotid bulb is usually double that of the normal ICA. This is the main reason for a different percentage stenosis measurement of the same carotid lesion using the two criteria. The CSI method of stenosis measurement is based on multiplying the CCA measurement with a factor of 1.2. The factor of 1.2 was based on studies of anatomic estimates of the ICA diameter from the CCA diameter, which was found to be 1:1.19 (±0.09). Percent stenosis is then calculated as in the other methods by comparing this with the diameter of the ICA stenosis (D) using the formula (1 − D/N) × 100%, with N = 1.2 × CCA diameter. With the advent of improved CTA technology, there have been studies that have validated a more direct estimation of the stenosis by using better visualization of the vessel wall and the contrast opacified lumen of the ICA stenosis. Most clinicians favor using the NASCET criteria for determining the degree of stenosis as a more accurate representation of actual ICA stenosis because the ICA often has a smaller caliber from the CCA at baseline. The reason the common carotid method is advocated by some investigators is that CCA and CSI criteria were created to preclude certain ambiguities in the NASCET and the ECST criteria. This calculation is based on the use of CCA as a denominator and the relative ease of determining the precise diameter on angiographic images without superimposed vessels or branches and its alleged disease-free state. The reader is advised to have a detailed understanding of the background of the various methods of estimating carotid stenosis to make an informed decision in selecting patients for the treatment of either CAS or CEA.
Treatment options are considered based on patients’ medical comorbidities, if they are symptomatic or asymptomatic, and the percent of ICA stenosis. At the authors’ institution, symptomatic patients with a stenosis of greater than 70% are offered treatment. Symptomatic patients with a 50% to 65% stenosis are also considered for treatment. The authors currently only consider treatment in patients who are asymptomatic if the operator has a less than 3% complication rate, the degree of stenosis is greater than 80%, and patients have a life expectancy of greater than 3 years. Currently, the treatment options include CAS and CEA. CEA is the traditional treatment approach; however, CAS has gained some acceptance based on the results of the Carotid Revascularization Endarterectomy Versus Stenting Trial (CREST). In the next few paragraphs, the authors discuss the technical details of CAS followed by CEA. Finally, the authors discuss the pros and cons of both techniques.
Endovascular technique
Technique
The procedure is performed under conscious sedation. A radial arterial line is placed to monitor patients’ blood pressure during and after the procedure. To minimize thromboembolic complication during stent placement and subsequent in-stent thrombosis caused by platelet aggregation, patients are placed on dual antiplatelet agents before the procedure. There is a lack of consensus on the standard dose regime. The authors recommend a loading dose of 325 mg aspirin with 300 mg clopidogrel bisulfate (Plavix) followed by a daily dose of 81 mg aspirin and 75 mg clopidogrel bisulfate (Plavix) for one week before the procedure. Aspirin function and P2Y12 inhibition assays are checked to ensure adequate platelet inhibition before the procedure. If patients are found to have inadequate platelet inhibition, the administration of an alternative antiplatelet agent, such as prasugrel (Effient), tricagrelor (Brilinta), or ticlopidine (Ticlid), should be considered.
Patients are placed supine on the angiographic table. Both groins are prepped and draped in the usual sterile fashion. The right common femoral artery is punctured retrograde under ultrasound guidance with a micropuncture kit using a modified Seldinger technique. The authors perform a complete cerebral angiogram with contrast injection in both common carotid arteries and one vertebral artery with projections over the neck and head before treatment. This procedure is to evaluate the intracranial vasculature and to assess the collateral circulation of the circle of Willis.
An 8F sheath is then placed at the arteriotomy site if the procedure is performed using a guide catheter. If the GORE flow reversal device (Gore Medical, Flagstaff, AZ) is used, a 9F sheath must be placed in the artery and a 6F sheath placed in the opposite common femoral vein. The Cook shuttle system (Cook Medial, Bloomington, IN) uses a 6F 90- or 100-cm length sheath directly introduced through the arteriotomy. After adequate femoral access is secured, patients are given a bolus of heparin (70–100 mg/kg) to achieve activated clotting times of approximately 250 to 300 (or 1.5–2.0 × baseline). The brachial artery or common carotid artery can be accessed in a similar fashion, if there is a contraindication to using the femoral artery. The details of these techniques are beyond the scope of this article.
The 8F guide catheter can be directly advanced through the sheath over a 0.035 inch Terumo Glidewire (Terumo Medical, Somerset, NJ) or coaxially over a 125-cm 6.5F Cook vertebral artery catheter (Cook Medical, Bloomington, IN) into the common carotid artery under biplane fluoroscopy. A digital subtraction angiogram (DSA) is obtained in the standard projection. A 30° right or left anterior oblique angulation in the anterior posterior (AP) plane is often beneficial to splay out the bifurcation of the respective CCA.
Under road map guidance, the guide catheter is navigated over the wire just proximal to the lesion, while taking care not to cross the stenotic lesion with the wire. If additional wire purchase is required to navigate the CCA, the wire should be placed into the ECA and not the ICA to avoid any disruption of the plaque. The lesion is then crossed with an umbrella type of cerebral protection device placed into the distal cervical segment of the ICA.
There are various cerebral protection devices commercially available, but they all use 1 of 2 principles: (1) proximal flow reversal or (2) a distal embolic protection umbrella to arrest any platelet aggregates/thromboemboli from transgression into the intracranial circulation. Table 1 is a list of all the currently available protection devices.
| Device | Company | Type | Crossing Profile (F) | Guiding Catheter (F) | Diameter (mm) |
|---|---|---|---|---|---|
| GuardWire | Medtronic Vascular, Santa Rosa, CA | Distal balloon | 2.1–2.7 | 6 | 2.5–6.0 |
| Angioguard XP | Cordis Corp, Bridgewater, NJ | Filter | 3.2–3.7 | 7–8 | 3.0–7.5 |
| Spider | Ev3, Plymouth, MN | Filter | 2.9 | 6–7 | 3.0–7.0 |
| FilterWire EZ | Boston Scientific Corp, Natick, MA | Filter | 3.2 | 6 | 3.5–5.5 |
| EmboShield Nav6 | Abbott Vascular, Santa Clara, CA | Filter | 2.9–3.3 | 6 | 2.5–7.0 |
| RX Accunet | Abbott | Filter | 3.5–3.7 | 7–8 | 3.25–7.0 |
| FiberNet | Medtronic | Filter | 2.4–2.7 | 6–7 | 3.5–7.0 |
| Mo.Ma Ultra | Medtronic | Proximal Balloon | na | 9 | 6.0 ECA 13.0 CCA |
| Flow Reversal | W.L. Gore & Associates, Flagstaff, AZ | Proximal Balloon | na | 9 | <6.0 ECA 12.0 CCA |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree