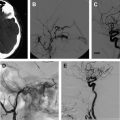
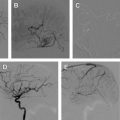
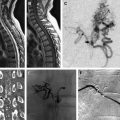
Cerebral vasospasm following aneurysmal subarachnoid hemorrhage (SAH) is a delayed, reversible narrowing of the intracranial vasculature that occurs most commonly 4 to 14 days after aneurysmal SAH and can lead to permanent ischemic injury. Angiographic spasm occurs in up to 70% of patients following SAH, and approximately half become symptomatic. Estimates of patients who are disabled by vasospasm, or die because of it, range from 5% to 9%, with vasospasm accounting for 12% to 17% of all fatalities or cases of disability after SAH. This article discusses the multiple medical and endovascular therapies used to prevent or treat vasospasm.
Key points
- •
Calcium channel blockers, magnesium, 3-hydroxy-3-methylglutaryl coenzyme A (HMG CoA) reductase inhibitors (statins), fasudil, thrombolytics, endothelial receptor antagonists, and tirilazad have all been used in attempts to prevent and treat vasospasm.
- •
Hypervolemia, hypertension, and hemodilution therapy (triple-H) has long been a mainstay of medical therapy in patients with aneurysmal SAH.
- •
Endovascular treatment with intra-arterial infusion of a vasodilator or balloon angioplasty is indicated in patients with symptomatic vasospasm refractory to medical therapy to prevent neurologic deficits referable to the vascular territory of the angiographic vasospasm.
- •
Papaverine, verapamil, nimodipine, and nicardipine have been used as intra-arterial vasodilators to treat patients with symptomatic vasospasm refractory to medical therapy.
- •
Two different balloon technologies have been used for treatment of vasospasm: coronary balloons and more compliant intracranial balloons.
Introduction
Cerebral vasospasm following aneurysmal subarachnoid hemorrhage (SAH) is a delayed, reversible narrowing of the intracranial vasculature that occurs most commonly 4 to 14 days after aneurysmal SAH and can lead to permanent ischemic injury ( Fig. 1 ). Although it has been the focus of much research and clinical effort, vasospasm remains difficult to treat and is responsible for significant morbidity and mortality in patients with ruptured cerebral aneurysms. Angiographic spasm occurs in up to 70% of patients following SAH, and approximately half become symptomatic. In the past, mortality rates from vasospasm have been reported to range from 30% to 70%, with 10% to 20% of patients experiencing severe neurologic deficits.
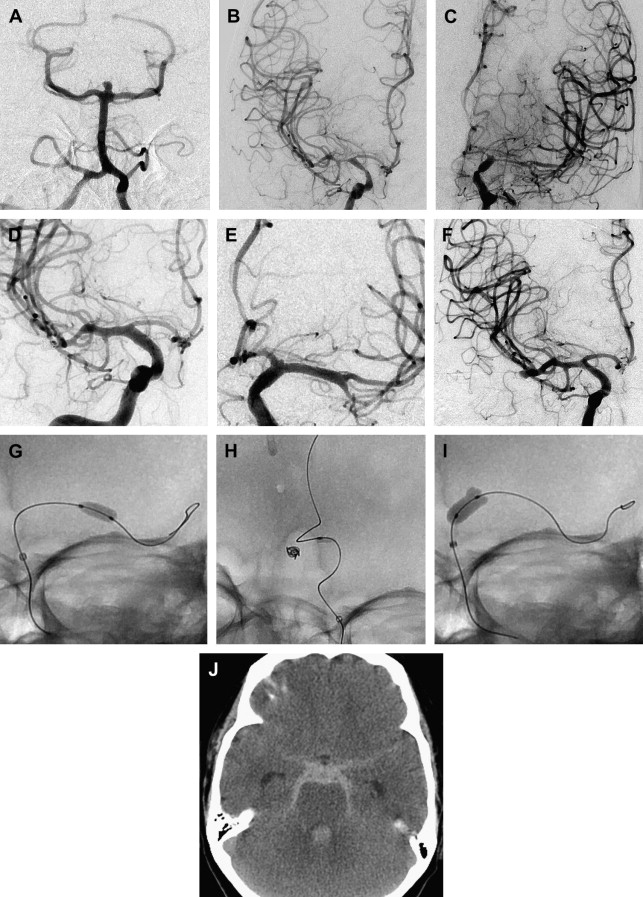
With advancements in diagnostic and interventional technology, estimates of patients who are disabled by vasospasm, or die because of it, range from 5% to 9%, with vasospasm accounting for 12% to 17% of all fatalities or cases of disability after SAH. We have an incomplete understanding of the pathophysiology of vasospasm, and it is difficult to predict which patients will develop vasospasm after SAH. The Hunt and Hess grade, Fisher score, hypertension, smoking, cocaine use, age range of 40 to 59 years, and early rise in the middle cerebral artery blood flow on transcranial Doppler have been shown to be independent risk factors for vasospasm. Cerebral salt wasting has also been associated with vasospasm. This article discusses the multiple medical and endovascular therapies that are used to prevent or treat vasospasm.
Introduction
Cerebral vasospasm following aneurysmal subarachnoid hemorrhage (SAH) is a delayed, reversible narrowing of the intracranial vasculature that occurs most commonly 4 to 14 days after aneurysmal SAH and can lead to permanent ischemic injury ( Fig. 1 ). Although it has been the focus of much research and clinical effort, vasospasm remains difficult to treat and is responsible for significant morbidity and mortality in patients with ruptured cerebral aneurysms. Angiographic spasm occurs in up to 70% of patients following SAH, and approximately half become symptomatic. In the past, mortality rates from vasospasm have been reported to range from 30% to 70%, with 10% to 20% of patients experiencing severe neurologic deficits.
With advancements in diagnostic and interventional technology, estimates of patients who are disabled by vasospasm, or die because of it, range from 5% to 9%, with vasospasm accounting for 12% to 17% of all fatalities or cases of disability after SAH. We have an incomplete understanding of the pathophysiology of vasospasm, and it is difficult to predict which patients will develop vasospasm after SAH. The Hunt and Hess grade, Fisher score, hypertension, smoking, cocaine use, age range of 40 to 59 years, and early rise in the middle cerebral artery blood flow on transcranial Doppler have been shown to be independent risk factors for vasospasm. Cerebral salt wasting has also been associated with vasospasm. This article discusses the multiple medical and endovascular therapies that are used to prevent or treat vasospasm.
Vasospasm prophylaxis and medical treatment
Prophylactic treatment for cerebral vasospasm following aneurysmal SAH is controversial and varies among institutions. Calcium channel blockers, magnesium, 3-hydroxy-3-methylglutaryl coenzyme A (HMG CoA) reductase inhibitors (statins), fasudil, thrombolytics, endothelial receptor antagonists, and tirilazad have all been used in attempts to prevent and treat vasospasm. In addition, triple-H (hypervolemia, hypertension, and hemodilution) therapy has been a mainstay of vasospasm treatment for years. At our institution, we start all patients with SAH with triple H as soon as the aneurysm has been secured, as long as there are no medical contraindications, such as congestive heart failure or severe pulmonary disease. We use prophylactic nimodipine and magnesium sulfate and are considering using statins as new data emerge.
Calcium Channel Antagonists
Calcium channel antagonists have been shown to decrease the overall incidence of cerebral infarction after SAH by 34% and the incidence of poor outcomes by 40%. The physiologic reasoning behind the use of calcium channel blockers is that the central event in vascular smooth muscle contraction is the influx of calcium into cells, which has been shown to occur after SAH. This, in turn, leads to several downstream events, including free radical formation, production of vasoconstricting prostaglandins, and activation of the myosin light chain kinase that causes smooth muscle contraction. The most common calcium channel blocker used after SAH is nimodipine, which has a certain degree of specificity for the cerebral vessels. Multiple trials have shown its efficacy in improving outcomes, although it may not improve angiographic outcome.
Two randomized controlled trials (RCTS) using nicardipine for treatment of and prevention of cerebral vasospasm have been performed. The largest RCT treated 449 patients with intravenous (IV) nicardipine (0.15 mg/kg/h) and 457 patient with placebo. Patients treated with nicardipine had significantly reduced vasospasm compared with placebo ( P <.001). Clinical outcomes were similar between the 2 groups at 3 months. Another small study that randomized 16 patients to receive nicardipine prolonged-release implants placed into the basal cisterns at the time of surgical clip placement demonstrated significantly reduced angiographic vasospasm incidence ( P <.05) and improved modified Rankin Scale scores at 1 year.
Magnesium
Obstetricians have used magnesium to treat eclampsia, as magnesium is thought to alter calcium physiology and thus alter vascular tone in the uterine circulation. Magnesium competes with calcium-binding sites, thus preventing muscular contraction and allowing vascular muscle relaxation. Level 1 evidence now exists for the usage of magnesium sulfate in the prevention of cerebral vasospasm. Westermaier and colleagues have shown that magnesium sulfate significantly reduces cerebral ischemic events after SAH.
Statins
Five RCTS have used statins for the prevention and treatment of cerebral vasospam following SAH. Three of the trials have shown a benefit for statins in reducing vasospasm, delayed ischemic neurologic deficits (DINDs), and mortality. However, 2 of the more recent trials have shown no significant difference in reducing vasospasm and clinical outcome.
Fasudil
Fasudil acts as a calcium channel blocker via the Rho-kinase signaling pathway inhibition. It antagonizes the vasoconstrictive effects of endothelin by dilating cerebral arteries in an animal vasospasm model. Fasudil has been studied in 2 RCTs. Shibuya and colleagues showed significant reductions in angiographic and symptomatic vasospasm, hypodensities on computed tomography (CT) scan, and poor outcome with use of the drug. In another study, Zhao and colleagues compared IV fasudil to IV nimodipine and found no significant differences in vasospasm incidence or clinical outcome.
Thrombolytics
Two RCTs have been performed using thrombolytic therapy for the prevention or treatment of cerebral vasospasm. Findlay in an RCT of 100 patients who received either a 1-time intracisternal bolus of 10 mg of tissue plasminogen activator (t-PA) or placebo at the time of surgical clip placement found a significant reduction in severe vasospasm without improvement in outcome. Hamada and colleagues, in another randomized controlled trial (RCT) of 110 patients who received either intrathecal urokinase (60,000 International units (IU) over 20 minutes) or no thrombolytic therapy following aneurysm coiling, showed a significant reduction in symptomatic vasospasm with improved outcome.
Endothelin receptor antagonists
A subclass of drugs has been developed to inhibit the binding of endothelin I, a potent vasoconstrictive peptide, to its target receptors of vascular smooth muscle cells. Two drugs in this class, clazosentan and TAK-044, have been studied in RCTs for the treatment and prevention of cerebral vasospasm following aneurysmal SAH. Shaw and colleagues, in study of 402 patients who were randomized to IV TAK-044 or placebo, found a decreased incidence of delayed cerebral infarction without improvement in the Glasgow Outcome Scale. Furthermore, Vajkoczy and colleagues, in a small RCT comparing patients receiving IV clazosentan to placebo, found a significant reduction in cerebral vasospasm incidence and severity. Fifty percent of the crossover placebo patients had reversal of vasospasm following the initiation of clazosentan. The Clazosentan to Overcome Neurologic Ischemia and Infarction Occurring After Subarachnoid Hemorrhage (CONSCIOUS-I) trial randomized 413 patients to either dose-escalated clazosentan or placebo. Significant dose-dependent reductions in moderate and severe vasospasm were noted with increased pulmonary complications, hypotension, and anemia.
Triple-H therapy
Hypervolemia, hypertension, and hemodilution therapy (triple-H) has long been a mainstay of medical therapy in patients with aneurysmal SAH. The rationale behind triple-H therapy is that maintenance of high circulating blood volume, increased perfusion pressures, and decreased blood viscosity will enhance cerebral blood flow in the setting of vasoconstriction. Although in healthy adults, changes in cardiac output do not change the local cerebral flow, they do affect cerebral blood flow in patients suffering from cerebral vasospasm. Our goal is to maintain euvolemia as defined by the central venous/wedge pressure that allows for the highest cardiac output.
The vast majority of our patients with SAH receive central lines so that central venous pressures can be closely monitored to achieve optimized volume expansion without causing pulmonary edema. We prefer central venous pressures in the range of 8 to 12 mm Hg, but these are highly individualized and must be closely monitored in relation to the clinical findings in each patient. Patients with cardiopulmonary disease may need to be evaluated with a Swan Ganz catheter, as the ideal venous pressure in these patients needs to be related to the ideal cardiac output. One series of 184 patients reported a 13% risk of device-related sepsis in patients with pulmonary artery catheters, as well as a 2.0% risk of congestive heart failure, 1.3% risk of subclavian vein thrombosis, and 1.0% risk of pneumothorax. Most patients with SAH require invasive blood pressure monitoring. In the post-clipping or post-coiling vasospasm period, we often allow patients to autoregulate with systolic blood pressure in the range of 200 mm Hg. The role of red blood cell transfusion is not well studied, but transfusion could certainly increase the oxygen carrying capacity. Three RCTs have evaluated the efficacy of triple-H therapy.
Endovascular therapy
Endovascular treatment with intra-arterial (IA) infusion of a vasodilator or balloon angioplasty is indicated in patients with symptomatic vasospasm refractory to medical therapy to prevent neurologic deficits referable to the vascular territory of the angiographic vasospasm. Any patient who is a candidate for cerebral angioplasty in the context of cerebral vasospasm must be evaluated with a CT scan of the head to rule out hemorrhage and the presence of hypodensity in the vascular territory to undergo angioplasty. A vessel diameter reduction between 25% and 50% from the initial angiographic diameter is usually treated with IA infusion of vasodilators. Vessel diameter reductions greater than 50% from the initial angiographic diameter are treated with a combination of mechanical and chemical angioplasty. The timing of endovascular intervention for vasospasm is critical, and a 2-hour window from the time of symptoms may exist for restoration of blood flow to ultimately improve the patient’s outcome.
Intra-arterial vasodilators
Papaverine
The most studied IA pharmacologic agent to date is papaverine. It is an opium alkaloid that is thought to alter adenosine 3′,5′–cyclic monophosphate levels in smooth muscles ( Table 1 ). The half-life is approximately 2 hours. In a review of IA agents for treatment of cerebral vasospasm, Hoh and Ogilvy reported that papaverine produced clinical improvement in only 43% of the treated patients. The effectiveness of treatment was short given its half-life; therefore, multiple treatments were required, which led to a variable and increased risk of complications. Platz and colleagues more recently reported a case of spontaneous hemorrhage following IA use of papaverine. They hypothesized that local increased levels of infused papaverine possibly led to a blood–brain barrier (BBB) breakdown with subsequent intracranial hemorrhage. Furthermore, there has been a recent report from Pennings and colleagues. They observed an abnormal response to topical application of papaverine on the cerebral cortical microvasculature during aneurysm surgery. They noted rebound vasoconstriction in 2 of 14 cases. In the cases in which there was some increase in vessel diameter compared with baseline, it did not reach statistical significance. There is wide variation in the IA use of papaverine in terms of dosage and duration of infusion. Firlik and colleagues demonstrated no correlation with the clinical response and angiographic picture in cases of vasospasm from aneurysmal SAH treated with IA papaverine. In their cohort of 15 patients, they had 23 IA treatments with papaverine, leading to partial angiographic reversal in 18 of the 23 IA treatments. However, major clinical improvement was seen in only 6 treatments and minor improvement or none in 17.
| Agent | Typical Intra-arterial Dose | Half-Life, Hours | Side Effects |
|---|---|---|---|
| Papaverine | 300 mg per vascular territory at 0.3% concentration over 20 min. | 2 | Cortical necrosis, permanent neurologic deficits, raised intracranial pressure, systemic hypotension |
| Verapamil | 1–2-mg bolus over 2 min with maximum of 10 mg per vascular territory. | 7 | Increased intracranial pressure |
| Nimodipine | 1–3 mg at 25% dilution over 10–30 min in each vascular territory; maximum dose 5 mg. | 9 | Increased intracranial pressure |
| Nicardipine | 0.2–0.5 mg/mL in ml aliquots; maximum dose of 20 mg per vascular territory. | 16 | Increased intracranial pressure |
Papaverine hydrochloride is supplied as a 3% concentration, 30 mg/mL, in an acidic mixture of pH 3.3. Papaverine may form crystal precipitates, with crystal size up to 100 μm, when mixed with human serum at 3.0% or 0.3% concentrations. A precipitate has also been seen when 3% papaverine solution was mixed with heparinized saline in concentrations of 2000 to 10,000 units of heparin per liter. The typical papaverine concentration infused is 0.3%, produced by diluting 300 mg of papaverine in 100 mL of normal saline. The entire dose (300 mg) is given in a vascular territory over 20 to 30 minutes. If more than 1 vascular territory is involved, additional infusions of 300 mg can be given. The catheter for infusion in the anterior circulation should be placed past the ophthalmic artery to prevent possible precipitate being introduced into the retina. In the posterior circulation, the catheter should be positioned past the origin of the anterior inferior cerebellar artery to prevent respiratory arrest and the potential of cardiac dysfunction because of transient depression of the medullary respiratory and cardiovascular nuclei. Papaverine can cause systemic hypotension and elevation of intracranial pressure during infusion; therefore, these parameters should be monitored closely.
Side effects from IA infusion of papaverine also included transient neurologic deficits, such as mydriasis, transient hemiparesis, and respiratory depression. Given the short half-life, the transient effect on the local cerebral vasculature, and significant side effects, we no longer use papaverine for chemical angioplasty at our institution.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree