chapter 6 Esophageal Cancer
INTRODUCTION
The optimal management of patients with localized esophageal cancer remains controversial. Treatment options include surgery alone, chemoradiation alone, preoperative chemoradiation, and preoperative chemotherapy. Radiotherapy plays a major role in potentially curative treatment, either as preoperative therapy or as definitive treatment. The majority of patients with locally advanced esophageal cancer are managed primarily with chemoradiotherapy. However, the prognosis for such patients is poor, with published 5-year survival rates of around 20%.1–3 Locoregional relapse remains a significant component of failure, with approximately 50% of patients failing locoregionally.2,4,5 Efforts to improve the outcome for patients treated with chemoradiation have included radiation dose escalation,5 combining external beam radiotherapy with brachytherapy,6 and employing neoadjuvant chemotherapy prior to chemoradiation.4 However, none of these approaches has proven successful and there is a clear need to develop alternative strategies to improve local tumor control.
The design of appropriate radiation fields is based on an understanding of the pathology and natural history of the disease, and on accurate localization of the tumor. Accurate radiotherapy simulation for esophageal cancer is challenging. Prior to the advent of computed tomography (CT) planning, conventional radiotherapy planning employed the use of esophagography to determine tumor extent, particularly the proximal and distal extent of the primary tumor in the esophagus. Precise identification of the proximal and distal tumor margins is important because recurrence can appear at the tumor edge due to clinically inapparent submucosal spread of disease longitudinally beyond the grossly visible tumor. The radiation treatment fields were generated by adding 5 cm proximally and distally, and 2 to 3 cm radially, beyond the perceived margins of the tumor to account for submucosal spread, as well as daily setup variations and patient movement. However, there are limitations to the use of indirect methods such as esophagography for tumor localization. Some early-stage tumors produce only minor mucosal abnormalities that may not be evident on barium esophagography. The quality of radiotherapy planning esophagograms is inferior to those obtained in a diagnostic radiology department where strict protocols are followed such as the administration of anticholinergic agents to prevent peristalsis of the esophageal wall. As such, the true extent of the tumor is not always apparent with planning esophagograms. In addition, esophagography provides no information regarding the radial extent of disease, and it has no role in defining mediastinal lymph nodes.
In most modern radiotherapy departments, CT planning represents the standard method for tumor volume delineation. Despite its widespread use, there are few studies addressing its impact on radiotherapy planning for esophageal cancer. Leung and colleagues have reported the results of a study comparing CT planning to traditional esophagogram-based planning.7 In this study, the authors projected the treatment fields determined from a conventional esophagogram-based planning protocol onto the CT scans of 75 consecutive patients with squamous cell carcinoma of the esophagus. They then assessed the adequacy of coverage of the primary tumor and metastatic nodes with respect to data from the CT scans. Adequate coverage for nodal disease was defined as complete inclusion within the field of all malignant nodes. Coverage of the primary tumor was defined as complete inclusion of imaging-evident tumor together with 5-cm proximal and distal margins. Their results showed that 38% of T2-3 tumors and 30% of short-length tumors (≤ 5 cm) had metastatic nodes outside the esophagogram-based treatment field. Sixty-three percent of all T2-3 tumors would not have been adequately covered with 5-cm proximal and distal margins in the esophagogram-based treatment field. In total, 79% of cases had inadequate coverage of the primary tumor and/or metastatic nodes.
Although CT allows localization of involved lymph nodes and definition of the radial extent of the primary tumor, it is even less accurate for defining the proximal and distal extents than properly performed esophagography.8 It is well recognized that the longitudinal extent of esophageal tumors is difficult to accurately define on CT imaging and this problem is compounded by the rising incidence of adenocarcinoma of the distal esophagus and gastroesophageal junction where the distal extent of the tumor (often in the cardia of the stomach) can at times be impossible to visualize on CT or esophagography. For diagnostic CT scans, dedicated methods have evolved to help delineate the distal extent of gastroesophageal junction tumors. These include administration of water or other oral contrast, intravenous anticholinergic agents to reduce peristalsis, and volumetric scanning in the supine and prone positions. Delineation of the proximal tumor extent is more problematic but again may involve the administration of anticholinergic agents and on-table esophageal contrast. Although such measures are routinely employed for diagnostic CT scans, they are not practical for radiotherapy planning CT scans which are usually performed without the use of contrast agents.
Several methods have been proposed to increase the accuracy of tumor volume delineation for esophageal cancer, particularly with respect to the proximal and distal margins of the primary esophageal tumor. Pfau and colleagues have evaluated the use of metal endoclips for tumor localization during radiotherapy planning.9 In this study, seven patients underwent endoscopic ultrasound for staging and to identify the longitudinal margins of the esophageal tumor. Under direct endoscopic visualization, a mucosal endoscopic metal clip measuring 1.2 mm × 6 mm was placed at the proximal and distal tumor margin. The patient then underwent standard simulation by a radiation oncologist based on barium esophagogram, CT, and endoscopy report. The process was then repeated by a second radiation oncologist using information based on endoclip location. Variations in field isocenter and radiation field length or width between the two simulation procedures was recorded. The results of this study demonstrated that simulation with the use of endoclips caused a mean shift in the field isocenter of 1 cm (range: 0–2.5). The mean longitudinal shift (y-axis) was 3.5 cm (range: 1–6), with five patients having an increase in field length and one patient having a decrease in field length. The mean lateral shift (x-axis) was 0.33 cm (range: 0–1), with two patients having an increase in field width of 1 cm and four having no change. In all patients, the field of radiation was changed based on endoscopic clip location. In some cases, the longitudinal dimension of the tumor was increased by as much as 5 to 6 cm when the clips were used to calculate radiation field size. The difficulties associated with this method of tumor localization include the requirement for an invasive procedure and the ability of clips to migrate or become dislodged.
Burmeister and colleagues have described a similar marker approach for localization of small esophageal tumors for radiation planning.10 In this study of 8 patients, the margins of the tumor were delineated at endoscopy by injecting contrast solution into the mucosa and submucosa with a sclerotherapy needle. The major difficulty with this method is that contrast solution dissipates into the tissues within minutes, making it impractical for routine clinical practice.
Endoscopic ultrasound (EUS) is widely used for local staging of esophageal cancer. Thomas and coworkers have described a method for incorporating the information obtained from EUS into the CT planning process.11 In this study, 11 patients underwent EUS in the left lateral position with the neck in a neutral position during quiet respiration. The position of the superior extent of the aortic arch was noted and used as a reference point. For each patient, the distance from this reference point to the proximal and distal tumor extent was recorded. Patients then underwent CT planning in the supine treatment position. The longitudinal position and length of the gross tumor volume (GTV) as defined by EUS was then transposed onto the planning CT by noting the table position of the superior aspect of the aortic arch and calculating the table positions of the proximal and distal extent of the tumor as determined by EUS. Two separate GTVs were then defined for each patient, one based on CT data alone (GTV-CT) and another based on EUS information (GTV-EUS). The location, length, and volume of GTV-CT and GTV-EUS were compared. The results showed a difference between the lengths of GTV-CT and GTV-EUS (mean = 5.95 and 6.91 cm, respectively; median = 6.0 and 6.0), while the volumes were similar (mean = 39.3 cm3 and 44 cm3, respectively; median = 38 and 40). There was considerable variation in the position of the GTV depending on the information used for planning. Assuming EUS to represent the true extent of disease, CT overestimated the proximal extent of the tumor in five patients by a mean value of 24 mm (range: 5–75), and underestimated the proximal extent of the tumor in five patients by a mean value of 14 mm (range: 5–25). CT overestimated the distal extent of the tumor in three patients by a mean value of 10 mm (range: 5–15), and underestimated the distal extent of the tumor in seven patients by a mean value of 26 mm (range: 10–55). The major problem with this method of tumor localization is that patients are not in the radiotherapy treatment position, thereby potentially affecting the position of mediastinal structures. Patients are also required to undergo EUS, which is not always done for patients being treated with definitive chemoradiation.
RATIONALE FOR THE USE OF PET AND PET-CT FOR ESOPHAGEAL CANCER
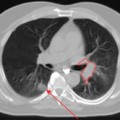
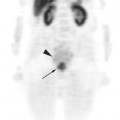
Positron emission tomography (PET) using 18F-fluoro-deoxy-D-glucose radiotracer (FDG) is a functional imaging modality that provides physiologic information based on altered tissue metabolism. The role and potential value of PET scanning in certain tumors, including esophageal cancer, has been widely investigated in recent years. When used for initial staging of esophageal cancer, PET is more accurate than computed tomography (CT) for detecting lymph node and distant metastases, thereby allowing more accurate selection of the most appropriate treatment.12–15 Flamen and colleagues have reported the results of a prospective study comparing the accuracy of FDG-PET with CT and EUS in staging 74 patients with potentially operable esophageal cancer.12 The sensitivity of PET for demonstrating the primary tumor in the esophagus was 95% (70 of 74 patients), with false-negative images found in four patients with small T1 lesions. FDG-PET demonstrated a higher accuracy for detection of metastatic disease compared with the combination of CT and EUS (82% vs. 64%), mainly due to superior sensitivity (74% vs. 47%) which resulted in the upstaging of 15% of the patients from M0 to M1 disease. In addition, the PET findings upstaged an additional 15% of patients and downstaged an additional 7% of patients. In the assessment of regional and distant lymph node involvement, FGD-PET had a higher specificity than the combined use of CT and EUS (98% vs. 90%) and a similar sensitivity (43% vs. 46%). Choi and colleagues investigated the accuracy of FDG-PET for evaluating individual lymph node groups in 48 consecutive patients with esophageal cancer.15 All patients underwent FDG-PET, CT, and EUS (45 patients) prior to esophagectomy and lymph node dissection. The imaging findings were then correlated to histopathologic findings. In the assessment of metastasis to individual lymph node groups, the sensitivity, specificity, and accuracy of FDG-PET was 57%, 97%, and 86%, respectively compared to 18%, 99%, and 78% for CT. For nodal (N) staging, FDG-PET was correct in 83% of patients compared to 60% for CT and 58% for EUS.
Recent studies have reported the use of FDG-PET to assess the early and late metabolic response of esophageal cancer to chemoradiation, which may be a valuable tool in predicting clinical and histopathologic response and patient long-term survival.16–23 Wieder and coworkers have evaluated the time course of tumor metabolic activity during chemoradiotherapy and correlated the reduction of metabolic activity with histopathologic tumor response and patient survival.16 In this study, 38 patients with squamous cell carcinoma of the esophagus underwent treatment with a 4-week course of preoperative chemoradiotherapy followed by esophagectomy. FDG-PET was performed before therapy, after 2 weeks of chemoradiotherapy, and prior to surgery (3 to 4 weeks after chemoradiotherapy). The authors found that changes in tumor metabolic activity after 14 days of chemoradiation were significantly correlated with tumor response and patient survival. Based on these early changes at 14 days, the subsequent histopathologic tumor response could be predicted with a sensitivity and specificity of 93% and 88%, respectively. Patients with a decrease of FDG uptake of < 30% had a median survival of 18 months compared to 38 months for patients with a decrease in FDG uptake of more than 30% (p = 0.03). This and other published studies suggest that FDG-PET could potentially be used to identify non-responders early during preoperative chemoradiotherapy so that therapy can be modified if necessary. Similarly, Downey and colleagues have reported the results of a prospective study correlating PET response to preoperative chemoradiotherapy with disease-free and overall survival rates after esophagectomy.22 In this study of 39 patients, the use of PET imaging after standard staging studies and prior to treatment identified undetected metastatic disease in 15% of patients. The 2-year disease-free survival after esophagectomy was 38% for patients with a < 60% decrease in standardized uptake value (SUV) (between PET images before and after preoperative chemoradiotherapy) and 67% for patients with a > 60% decrease in SUV (p = 0.055). The 2-year overall survival was 63% for patients with a < 60% decrease in SUV and 89% for patients with a > 60% decrease in SUV (p = 0.088).
Our group has also reported the impact of PET response to chemoradiotherapy on patient management and survival.23a Fifty-three consecutive patients with esophageal cancer underwent FDG-PET to evaluate their tumor response to chemoradiotherapy (50.4 Gy in 28 fractions) prior to possible surgery. Integrated into the routine PET request form, clinicians were required to prospectively indicate their treatment plan following chemoradiotherapy if PET was not available (e.g., based on restaging CT and endoscopy). This pre-PET treatment plan was recorded for each patient and then compared with the actual treatment that was delivered based on incorporation of the PET findings. The impact of incremental PET findings on management was considered high when the treatment intent or modality was changed (e.g., from curative to palliative treatment, or from chemoradiotherapy to surgery), and low when the PET results did not indicate the need for change. PET was considered to have had no impact when the management plan was not changed and the PET findings did not influence the decision-making process. After completion of chemoradiotherapy, 23 patients (43%) achieved a complete metabolic response with no evidence of residual disease on PET criteria. In 19 patients (36%) the treatment plan following chemoradiotherapy was significantly altered by the restaging PET findings that were different to those of restaging CT. There was a change in treatment intent in five patients (from curative to palliative in three patients and from palliative to curative in two patients). The treatment modality was changed in 14 patients. Nine patients who achieved a complete metabolic response on PET were closely observed instead of undergoing major surgery or additional chemotherapy. Two patients achieved a complete response on CT criteria but were found to have residual disease on PET and proceeded to surgical resection. The remaining three patients had a minimal response to chemoradiotherapy as determined by PET and received further courses of chemotherapy. Patients with a complete metabolic response on PET had statistically superior survival compared to those with an incomplete metabolic response (78% vs. 33% 2-year survival). The relative risk of death for patients with an incomplete metabolic response was increased 5.75-fold compared to patients with a complete metabolic response (95% CI, 1.94–17.05, p < 0.001), and this remained significant after multivariate analysis adjusting for tumor stage, histological subtype, and whether or not surgical resection had been performed.
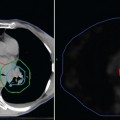
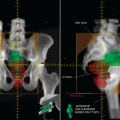
Although the precise role of PET in the management of esophageal cancer continues to evolve, it is apparent that this imaging modality is being used with increasing frequency. For radiation oncology, there is accumulating evidence to support the need for this type of functional information on esophageal tumors for the purpose of radiotherapy treatment planning. With the move to three-dimensional conformal radiation therapy (3D-CRT) techniques and dose escalation, it is even more important that the GTV be accurately and precisely defined. Without this accuracy, it is possible that “geographic misses” may occur due to the tight conformity of dose used in conformal radiotherapy. In recent years, several reported studies incorporating FDG-PET into conventional CT-based radiotherapy treatment planning for patients with lung cancer have shown that the use of PET simulation enhances the accuracy of target volume coverage.24–27 In these studies patients usually underwent sequential simulation with separate CT and PET units followed by co-registration of the CT and PET images. In recent years, specially designed integrated PET-CT scanners in which both imaging modules are mounted in line in one apparatus have become available and are reported to provide additional diagnostic information by comparison with conventional visual correlation of PET and CT.28 For radiotherapy purposes, the use of an integrated PET-CT scanner allows treatment planning to be performed without the need to image the patient separately and then co-register CT and PET images. This has the potential to eliminate the inaccuracies related to image co-registration such as patient setup in simulation CT and PET, acquiring data in different times, and breathing pattern and internal organ motion between the scans.
As demonstrated for lung cancer, the addition of PET to radiotherapy planning of esophageal cancer may likewise allow radiation oncologists to define the GTV with greater precision. PET has high sensitivity and specificity for disease within the esophagus with a sensitivity of 90% to 100%.12,29 It also demonstrates higher sensitivity and specificity for diagnosis of pathologic lymph nodes than does CT and EUS.15,30–32 The addition of PET information therefore has the potential to improve target volume delineation of both the primary tumor in the esophagus as well as involved lymph nodes. Whether or not the combination of PET and CT is able to truly delineate the extent of disease in the esophagus more accurately than CT alone is unknown because there have been no studies reporting pathologic correlation. However, it is probable that the use of combined PET-CT for radiotherapy planning is likely to be superior to our current standard of CT alone. It is also likely that the increasing availability of this technology will lead to more widespread use of PET-CT units for radiotherapy treatment planning. Over the past several years, over 200 combined PET-CT units have been installed in the United States,24 and dedicated PET-CT scanners designed specifically for radiotherapy treatment planning are now commercially available.