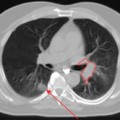
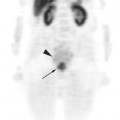
chapter 11 Genitourinary Cancer
INTRODUCTION
Positron emission tomography (PET) using 18F-fluoro-deoxy-D-glucose (FDG) is currently being used in clinical oncology for the diagnosis, staging, and treatment monitoring of various tumor types. However, the use of PET or PET-CT is still not standard of care in genitourinary oncology, despite the importance and frequency of this group of malignancies; in 2007, prostate cancer is predicted to be the most common noncutaneous cancer in men.1 The scope of this chapter is limited to kidney, prostate, bladder, and testicular cancers.
Genitourinary oncology has also witnessed a slow development in the use of the most commonly employed radiopharmaceutical probe, 18F-fluoro-deoxy-D-glucose (FDG). For kidney cancer, FDG-PET imaging has not been shown to improve the management of renal cell carcinoma (RCC) because the sensitivity and specificity of PET seem to be less than CT scan.2 Furthermore, the expression of glucose transporters such as GLUT-1, responsible for the accumulation of FDG, is low in kidney cancer. Prostate cancer is usually slow growing and exhibits low metabolic (glucose utilization) activity. It is also very difficult for PET to distinguish benign prostatic hypertrophy (BPH) or other inflammatory processes from prostate cancer.3 The excretion of FDG through the urinary tract has limited the use of FDG-PET for evaluation of primary bladder carcinoma as the visualization of bladder is difficult. However, PET-CT may be useful to detect distant metastases because metastatic lesions from bladder cancer are usually FDG-avid. FDG-PET has been shown to be most useful in testicular cancer, especially in the setting of residual or recurrent disease.4 Currently, there is a very limited role of radiotherapy for non-seminoma, but for early stage seminoma radiotherapy is still the standard of care. FDG-PET has been shown to be of value to assess residual masses that may contain fibrosis, necrosis, or harbor residual cancer. For residual tumor after treatment for seminoma, De Santis and colleagues found that the sensitivity, specificity, positive predictive value, and negative predictive value of FDG-PET were 100%, 80%, 100%, and 96%, respectively, while for computed tomography (CT) they were 74%, 70%, 37%, and 90%, respectively.5
Rationale for Use of PET and PET-CT in Genitourinary Malignancies
Staging and Re-staging
Despite the difficulty of using FDG-PET and FDG-PET-CT in the diagnosis and staging of primary cancer due to renal excretion of FDG, the use in detecting metastatic lesions of genitourinary primary is of value. Recently, Dilhuydy and colleagues demonstrated that a positive FDG-PET may alter the decision making in the management of metastatic RCC.6 With the advances made in using new PET tracers, including radiolabeled acetate and choline, the role of PET may expand in uro-oncology.7
Radiotherapy Treatment Planning
Currently, there are no published reports on integrating either PET or PET-CT in radiotherapy treatment planning for genitourinary malignancies. In our experience, we have incorporated the use of FDG-PET-CT in radiotherapy treatment planning for metastatic genitourinary cancers. The field of genitourinary oncology is currently seeing new advances. For example, two new molecular targeted therapies, namely sorafenib (Nexavar) and sunitinib (Sutent), have been recently approved for the treatment of metastatic RCC.8 Patients with metastatic RCC are going to live longer, thus the role of local treatment such as radiation therapy will become more important, especially for palliating and providing local control to symptomatic metastatic lesions. At the same time, the field of radiotherapy is also witnessing advances in technology including intensity-modulated radiotherapy (IMRT), image-guided radiotherapy (IGRT), and stereotactic body radiation therapy (SBRT).
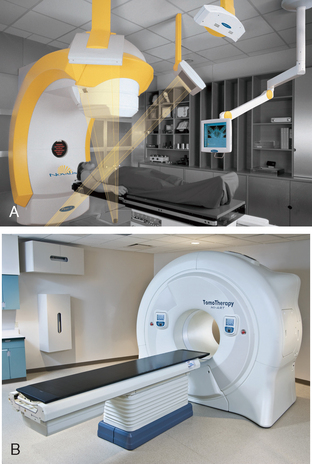
Imaging plays a very important role in the new era of radiotherapy whereby radiation oncologists can now deliver more precise radiation, allowing dose escalation to improve treatment outcome and conformal avoidance of surrounding normal tissues to decrease treatment-related side effects. The capability of combining anatomical (CT or magnetic resonance imaging [MRI]) with functional (PET) imaging has opened up new opportunities in the field of oncology. SBRT is an emerging technology in radiotherapy and is defined by American Society for Therapeutic Radiology and Oncology (ASTRO) and American College of Radiology guidelines as a “treatment method to deliver a high dose of radiation to the target, utilizing either a single dose or a small number of fractions with a high degree of precision within the body.”9 The emergence of SBRT is due to the advances in radiotherapy technology in IGRT. In our department of radiation oncology at the Methodist Hospital in Houston there are two IGRT machines: the BrainLab Novalis (BrainLab, Feldkirchen, Germany) stereotactic system (which uses kV X-ray for image guidance) (Figure 11-1A) and the Helical TomoTherapy (TomoTherapy, Inc, Madison, WI) (which uses megavoltage CT [MVCT] for image guidance) (Figure 11-1B). We have been delivering SBRT to both primary and metastatic genitourinary malignancies, especially in more radioresistant tumors such as RCC. The theoretical advantage is that higher fraction size will be beneficial in overcoming radioresistance. Recently, there is some evidence demonstrating the efficacy of SBRT in the radiotherapeutic management of RCC.10–12 In addition, there are also data to support the use of SBRT in oligometastases in a number of body sites, including lung, liver, bone, and spine from various primary tumors.13 Again it is believed that metastatic lesions (usually also of higher grade) are more radioresistant than their primary counterparts and thus the positive implication for higher fractional dose.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree