Introduction
Gynecologic malignancies are a considerable source of morbidity and mortality, accounting for nearly 672,000 deaths globally in 2020 ( ). Imaging complements surgical and clinical gynecologic cancer staging. Pelvic contrast-enhanced magnetic resonance imaging (CE-MRI) is commonly used in clinical practice to evaluate gynecologic tumor size and local extent due to its exceptional soft tissue contrast ( ). Other advantages of MRI include the availability of diffusion weighted and dynamic-contrast enhanced sequences, which facilitate tumor visualization. 18-flourodeoxyglucose (FDG) positron emission tomography/computed tomography (PET/CT) is frequently utilized to identify lymph node and distant metastases from gynecologic malignancies ( ; ; ). A particular advantage of FDG-PET is its ability to detect metabolically active tumor cells in lymph nodes that otherwise appear morphologically normal on anatomical cross-sectional imaging ( ).
Positron emission tomography/magnetic resonance imaging (PET/MRI) combines the advantages of both FDG-PET and MRI, potentially providing a “one-stop shop” imaging modality for gynecologic cancer staging and treatment planning ( ; ). This may result in fewer patient visits and a more efficient and accurate diagnostic work-up. PET/MRI also has a lower radiation dose than PET/CT, which may be of particular importance in younger patients who undergo multiple scans ( ). Furthermore, PET/MRI may be used for other clinical indications such as treatment response evaluation, prognosis determination, and radiotherapy planning in the case of uterine cervical cancer.
This chapter discusses the role of PET/MRI in patients with gynecologic cancers. The utility of PET/MRI to characterize indeterminate ovarian lesions is also briefly discussed. The potential limitations of PET/MRI are addressed as well.
PET/MRI protocol
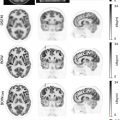
Several approaches are used to acquire PET/MRI in patients with gynecologic malignancies with scan acquisition times generally ranging from 60 to 90 min. Most integrate a dedicated pelvic protocol, heavily based on high resolution fast spin echo T2-weighted sequences without fat saturation, with a whole-body protocol that generally consists of T1-weighted Dixon images, T2-weighted half-Fourier acquired single-shot turbo spin echo (HASTE) images, fluid-sensitive inversion recovery images, and diffusion weighted images (DWI) ( ). In some centers, aqueous intravaginal gel is administered to distend the vagina, which provides better visualization of the vaginal walls and ectocervix. Differences arise in acquiring the pelvic protocol before or after the whole-body acquisition, and in focusing dynamic contrast-enhanced sequences over the pelvis or liver. Moreover, in some institutions, like ours, all MRI sequences are acquired simultaneously with the FDG-PET acquisition, which starts 60–90 min after FDG injection. Meanwhile, in some other institutions, a stand-alone pelvic MRI protocol is acquired immediately after FDG injection. This reduces the exam time for the patient but eliminates the value offered by simultaneously acquiring pelvic MRI and PET, potentially leading to an increased incidence of misregistration artifact Fig. 13.1 .

Uterine cervical cancer
Clinical overview
The incidence and mortality rates associated with cervical cancer are directly related to the availability and access to primary and secondary prevention. Specifically, they depend on the existence of human papillomavirus (HPV) vaccination and screening programs ( ; ). Globally, there are approximately 604,000 new cervical cancer cases and 342,000 deaths annually, making cervical cancer the fourth most common cancer in women ( ). An estimated 85% of these cases occur in resource-limited regions of the world with limited vaccination and screening programs. In these areas, cervical cancer ranks as the second most common type of cancer (15.7 per 100,000 women) and the third most common cause of cancer-related mortality (8.3 per 100,000) ( ). In the United States (U.S.), cervical cancer is both the third most common gynecologic cancer and cause of gynecologic cancer-related death, accounting for an estimated 14,500 new cases and 4300 deaths annually ( ).
Cervical cancer is staged clinically and the diagnosis is established by histologic evaluation of cervical tissue on biopsy. In 2018, the International Federation of Gynecology and Obstetrics (FIGO) revised the staging system for cervical cancer to include tests and procedures, including imaging, to facilitate appropriate staging assignment ( ; ). The revised staging system also includes changes in tumor size categories and the inclusion of lymph nodal metastatic status, with the goal of enhancing treatment approach and the prognostic predictive value of staging. Under the new FIGO system, cervical cancer staging remains clinical and allows for imaging use to vary depending on the practice setting ( ). In resource-limited settings, modalities such as chest radiographs and pelvic ultrasound may be used. In more affluent settings, staging may be assigned following findings from an expanded list of imaging examinations inclusive of CT, MRI, and FDG-PET ( ; ).
PET/MRI for initial staging
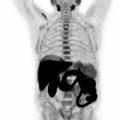
Pelvic CE-MRI is currently the first-line imaging modality used for simultaneously assessing cervical cancer size and extent of central pelvic tumor spread ( ; ; ). Several studies have shown that PET/MRI and pelvic CE-MRI evaluate local tumor extent with similar efficacy ( ; ; ). For example, in a study of 53 patients with newly diagnosed cervical cancer, PET/MRI and pelvic CE-MRI enabled an accurate tumor (T) stage designation in 85% and 87% of patients, respectively ( ). Furthermore, both imaging modalities shared comparable sensitivities and specificities for the detection of tumor extension into the parametrium, pelvic sidewall, and nearby pelvic organs. In another study, both PET/MRI and pelvic CE-MRI correctly identified the T-stage in 25 out of 30 patients with cervical cancer ( ). In contrast, Steiner et al. reported that PET/MRI was superior to pelvic CE-MRI in identifying parametrial, vaginal, and deep cervical stromal primary tumor invasion ( ). Fig. 13.2 demonstrates PET/MRI images of a cervical cancer confined to the cervix without local invasion. In contrast, Figs. 13.3 and 13.4 show PET/MRI images of locally invasive cervical cancers that extend into the parametrium and rectum, respectively.



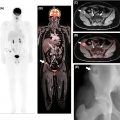
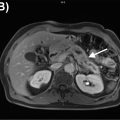
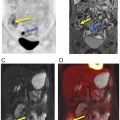
FDG PET/CT, when available, is used as a noninvasive alternative to surgical lymphadenectomy to diagnose cervical cancer lymph node metastases ( ; ). PET/CT is more sensitive than CT in detecting both lymph node and distant metastases ( ; ). Interestingly, several studies have demonstrated that PET/MRI is either comparable or superior to FDG PET/CT in identifying regional and distant lymph node metastases from cervical cancer. Kitajima et al. reported that PET/MRI and FDG PET/CT had an identical sensitivity of 92%, specificity of 88%, and accuracy of 90% for the detection of nodal metastases ( ). Another study of 79 patients with cervical cancer revealed that PET/MRI performed significantly better than FDG PET/CT in detecting lymph node metastases ( P = .03) ( ). In this same study, utilization of PET/MRI allowed radiologists to recognize six additional metastatic lymph node groups that were otherwise not seen with FDG PET/CT. Compared to FDG PET/CT, Anner et al. demonstrated that PET/MRI had a higher specificity (77% vs. 69%), positive predictive value (75% vs. 69%), and negative predictive value (67% vs. 64%) for metastatic lymph node detection in the pelvis ( ). However, both imaging modalities shared the same sensitivity of 64%. Fig. 13.5 demonstrates a cervical cancer loco-regional lymph node metastasis (i.e., infrarenal abdominal and pelvic retroperitoneum) that is better visualized with PET/MRI than FDG PET/CT.

While several studies have reported that PET/MRI performs well for the detection of distant metastases, most do not make a distinction between distant lymph node metastases and distant extranodal metastases such as lung, bone, liver, and peritoneal metastases. This is important to consider, particularly with lung metastases, as pulmonary nodules are more difficult to visualize on MRI than CT ( ). Additionally, anatomic localization of small peritoneal implants may be hindered by respiratory and bowel motion artifact, which is encountered more commonly with MRI due to its longer acquisition time than CT ( ). However, according to a recently published prospective study that enrolled 164 patients, PET/MRI detected peritoneal carcinomatosis with a higher sensitivity than current standard of care imaging modalities, including CT, FDG PET/CT, and MRI (97% vs. 54%) ( ). The protocol utilized in this study incorporated DWI, T2-weighted HASTE, and delayed contrast-enhanced T1-weighted MRI sequences, which were simultaneously acquired with PET. In terms of detecting and characterizing liver metastases, PET/MRI is unparalleled. In fact, the sensitivity for metastatic liver lesion detection was higher for PET/MRI (95%) than stand-alone PET (83%) and stand-alone MRI (88%) in a study of 79 patients with solid extrahepatic malignancies ( ).
PET/MRI for other clinical indications
The evaluation of cervical cancer treatment response is most commonly predicated on change in tumor size measured on pre- and posttreatment anatomical cross-sectional imaging studies ( ). However, this approach is limited as it does not take into account metabolic information added by FDG-PET as well as functional information added by MRI dynamic contrast-enhanced and DWI sequences. PET/MRI provides morphologic, metabolic, and functional information, which may aid in distinguishing treatment responders from nonresponders. In fact, a study from 2018 demonstrated that several quantitative parameters derived from PET/MRI correlated well with Response Evaluation Criteria in Solid Tumors and PET Response Criteria in Solid Tumors for differentiating those who did and did not respond to chemotherapy and concomitant chemoradiation ( ). Tumors in treatment responders demonstrated a 60% reduction in size, 64% reduction in maximum standardized uptake value (SUVmax), and 38% increase in minimum apparent diffusion coefficient (ADCmin). Furthermore, several measures of tumor perfusion decreased, including a 39% reduction in the transfer constant of capillary permeability (Ktrans) and a 47% reduction in the efflux rate constant (Kep). In contrast, the patient who did not respond to treatment demonstrated only a 9% reduction in tumor size, 6% reduction in SUVmax, and 35% increase in ADCmin. An increase in tumor perfusion parameters, including a 15% increase in Ktrans and a 22% increase in Kep was also observed in the treatment nonresponder.
Both FDG-PET and MRI provide valuable prognostic information in cervical cancer patients. In one study, higher tumor volume (>60 cm 3 ) estimated by FDG-PET was associated with worse overall survival and progression free survival in cervical cancer patients who were treated with radiation therapy ( ). In another study of patients with newly diagnosed cervical cancer, parametrial involvement detected by MRI and para-aortic lymph node involvement detected by FDG PET/CT were better predictors of progression free survival than various clinical parameters alone, including age at cervical cancer diagnosis and tumor grade ( ). Furthermore, Surov et al. reported that tumors with higher SUVmax to ADCmin ratios on PET/MRI were more likely to demonstrate increased expression of nuclear protein Ki-67, a biomarker of tumor proliferation ( r = 0.71, P = .001) ( ).
Both FDG PET/CT and MRI are typically acquired for cervical cancer radiotherapy planning ( ). CT is used primarily for dose calculation and patient positioning, whereas FDG-PET and MRI are used primarily for contouring of the radiation field. Ahangari et al. demonstrated that PET/MRI with a synthetic CT derived from MRI images was effective for radiotherapy planning in cervical cancer patients ( ). This may obviate the need for cervical cancer patients to undergo multiple imaging examinations prior to the initiation of radiation therapy.
Uterine endometrial cancer
Clinical overview
Worldwide, uterine corpus cancer is the second most common gynecologic malignancy, followed by uterine cervical cancer ( ; ). It is estimated that over 90% of uterine corpus cancers arise from the endometrium. Uterine mesenchymal tumors make up the remaining 10% of uterine cancers. Approximately 1%–2% of women in the U.S. will develop endometrial cancer in their lifetimes, most commonly in the sixth and seventh decades of life ( ; ). The increased incidence of endometrial cancer may be secondary to the increased prevalence of obesity, diabetes mellitus, and nulliparity, as well as the decreased use of progesterone therapy in menopausal hormone replacement regimens ( ).
Approximately 80% of endometrial cancers are low-grade (FIGO grades 1 and 2) endometrioid adenocarcinomas and present as early stage tumors with a favorable prognosis ( ). In contrast, grade 3 endometrioid and nonendometrioid tumors are rarer and more aggressive ( ). Risk factors for developing low-grade tumors include long-term exposure to exogenous unopposed estrogen, elevated endogenous estrogen levels, tamoxifen therapy, obesity, nulliparity, diabetes mellitus, and hypertension, as well as several genetic conditions, including Lynch and Cowden syndromes ( ). Risk factors for the development of high-risk endometrial cancer are less well established but may include lower body mass index, non-White race, and older age ( ). Molecular profiling of endometrial cancers has demonstrated promising prognostic and therapeutic potential ( ). Genomic, transcriptomic, and proteomic analyses have resulted in the identification of four molecular subtypes of endometrial cancer with distinct clinicopathologic features and prognostic outcomes ( ).
Endometrial cancer is staged surgically. Given that most low-grade endometrial cancers are also early-stage tumors, the risk of extrauterine disease is low ( ). Imaging of patients with suspected endometrial cancer involves preoperative ultrasonography, usually performed prior to endometrial sampling and as part of the evaluation of symptoms such as postmenopausal or abnormal uterine bleeding. In contrast, high-grade tumors are associated with a risk of extrauterine disease ( ; ; ). In these cases, imaging of the chest, abdomen, and pelvis, usually with CT or FDG-PET, is routinely performed preoperatively to identify peritoneal, pulmonary, and lymph nodal metastases. This may influence the choice of surgical technique (i.e., laparotomy vs. a minimally invasive route of surgery), intent of surgery (i.e., cytoreductive surgery vs. surgery performed solely for staging disease), and approach to lymphadenectomy. Imaging may also guide the recommendation for primary treatment with neoadjuvant chemotherapy before interval debulking surgery. Although the role of secondary cytoreductive surgery in the management of recurrent oligometastatic endometrial cancer is largely informed from retrospective data, selection of appropriate patients for this treatment approach relies on pre-operative imaging to determine the location of recurrence and surgical resectability of the tumor ( ). Imaging is also used to confirm the presence of a suspected isolated recurrence ( ).
PET/MRI for initial staging
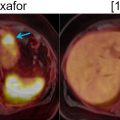
Pelvic CE-MRI can be particularly useful for the initial staging of high-grade endometrial cancers that have a predilection for extrauterine spread ( ). Several studies have demonstrated that PET/MRI and pelvic CE-MRI perform similarly for the assessment of local tumor extent. In one study, both PET/MRI and pelvic CE-MRI correctly determined the T-stage in 24 out of 30 patients with endometrial cancer ( ). In another study, both imaging modalities had an identical accuracy for the detection of uterine serosal, parametrial, vaginal, and adnexal invasion ( ). Fig. 13.6 illustrates how PET/MRI facilitates the assessment of myometrial invasion from endometrial cancer.