3D angiography
2D fluoroscopy
Real-time interventional CT
Flexible geometry with appropriate and safe patient access (i.e., for anesthesia)
OR environment: surgical lights spots and sterility facility
Surgical microscope (with ICG and intrinsic optical imaging and 3D output)
Coupling of FP to neuronavigation
Multimodality image co-registration
Neuromonitoring
3D monitor
Monitor display with vital signs and ICPs
Adjusted angio carbone table
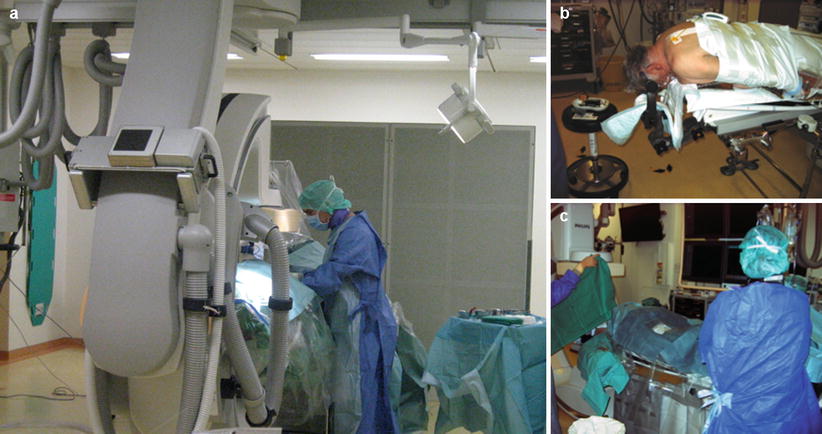
Fig. 40.1
Positioning and preparation. (a) Classic femoral approach. (b) Ventral decubitus positioning. (c) A radial arterial approach prepared. Note that the surgical microscope and neuronavigation are coupled with the FP system. A 3D screen and neuromonitoring system are located beside the table and on the field of view of the interventional neuroradiologist
4.
Intraoperative angiogram
(a)
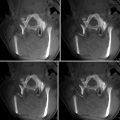
Vascular access: The femoral approach is the usual vascular access for intraoperative angiographic procedures. It permits a straight pathway to approach supra-aortic trunks and cervical vessels. A 5 or 6 F sheath is usually used for procedures where the patient is placed in supine position. This sheath is placed at the same time of patient’s preparation for surgery and kept with a continuous flushing system of saline to avoid thrombotic complications. For lateral, prone, or sitting positions, we use a long protected sheath (e.g., Arrow super flex, 5 or 6 F, 45 cm). This type of sheath is designed for difficult vascular access and to avoid kinking during different positions of the patient. The technique of the angiography does not differ from conventional diagnostic procedure where 5 F diagnostic catheters are used with different curves depending on the individual vascular anatomy. Alternatively, we had the possibility of using a radial approach. It is used when the femoral is not available or the catheterization is difficult. The puncture site is prepared at the beginning of the surgical procedure, but the puncture is performed just before the angiography. All intracranial vessels can be accessed with a practical reminder of using diagnostic catheters with Simmons curves (e.g., type 2 and 3) (Figs. 40.2 and 40.3).
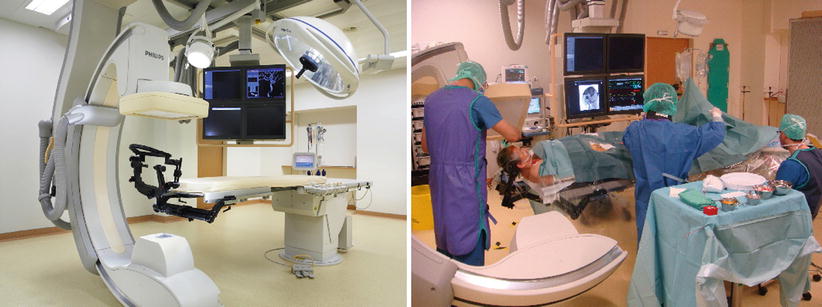
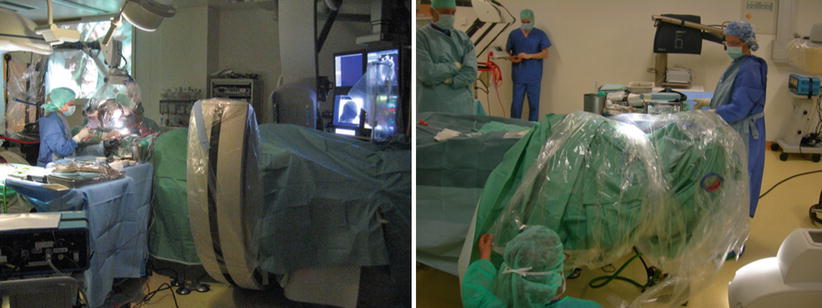

Fig. 40.2
Geneva facility: Illustration of basic setup in the hybrid room. The Flat Panel system, including table with radiolucent head holder and overhead monitors. Left: Hybrid system from Philips Allura FD20 with a radiolucent head holder and a radiolucent angiography/operating table (Philips). Right: Simultaneous preparation of the patient. Placement of the femoral sheath by the neuroradiologist and head position by the neurosurgeon

Fig. 40.3
Geneva facility: Left: Intraoperative setup with C-arm positioned caudally to the surgical field. Right: Preparation of patients’ head for intraoperative control
Clinical Application
The purpose of surgery for neurovascular disorders is the complete exclusion or excision of the vascular lesion. However, the surgeon may be incapable to verify these results with direct visual control of the surgical site alone. The final result always needs to be confirmed with a diagnostic angiography. When it is done during the postoperative time, a second procedure is imposed if there is a residual of a vascular lesion. So, the first aim of introducing the angiogram inside the neurosurgical room is to avoid an eventual second procedure.
The first report on intraoperative angiography has been published in the 1970s, and they reported 39 patients harboring intracranial aneurysms or AVMs treated with intraoperative angiographic control [7]. At that time, a dynamic image was obtained with series of multiple plain radiographies after contrast injection. Adaptation of head holders and tables started to be adapted for intraoperative angiograms [8]. In the 1980s, with the development of more powerful computers and suitable hardware, the images could be acquired digitally. Thus, the setting of digital angiography was too much complex in the beginning; they had to adapt in dedicated rooms.
Digital X-ray came to operative rooms with the development of mobile C-arms. Dynamic intraoperative angiography became then possible. More and more centers performing intracranial vascular procedures started to use intraoperative angiography and report their surgical results with great improvement [7–12].
Intracranial Aneurysms
At the University Hospital of Geneva, a consecutive series of n = 65 patients, n = 20 of which having presented with SAH, underwent microsurgical treatment of a total of n = 88 intracranial aneurysms. After 3D RA angiogram control post clipping, neck remnants were visualized in n = 11 aneurysms and parent and/or branching arterial compromise in n = 3 (Fig. 40.4). In a total of 9.1 % (n = 8) of aneurysms, where it deemed possible and useful, intraoperative 3D RA has led to repositioning of the clip(s). In one patient intraoperative superselective thrombolysis had to be performed after thrombus formation post clipping visualized after intraoperative angiography. Six patients underwent combined endovascular and open surgical treatment of their aneurysms, i.e., in the case of multiple aneurysms, of which one had to be clipped and the other to be coiled, or be it in the case of change of the intraoperative strategy (e.g., after repeated rupture of a MCA aneurysm during dural opening, where it was decided to continue with endovascular therapy) (see Figs. 40.5 and 40.6) [1].
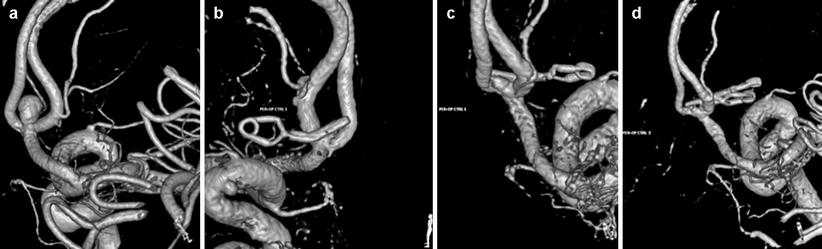
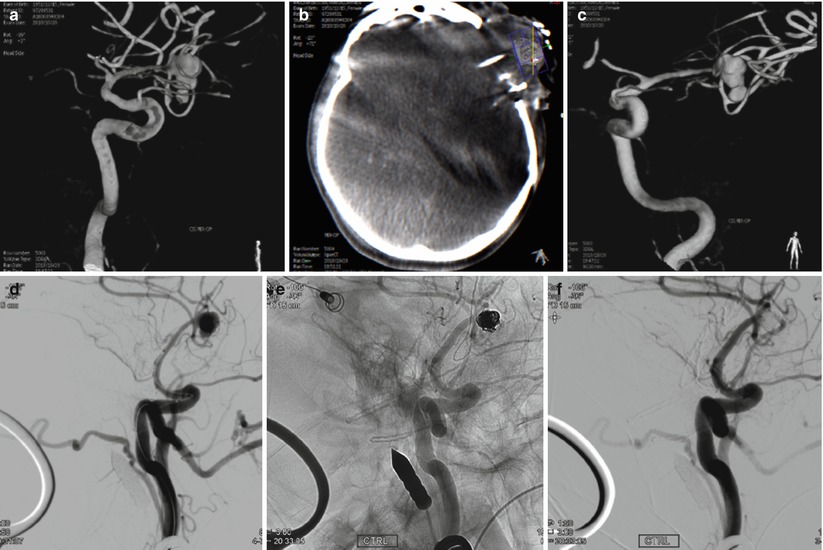
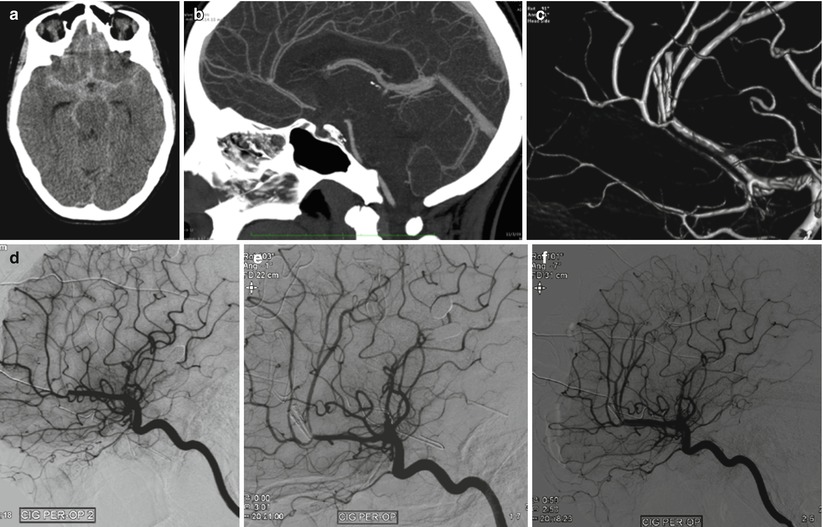

Fig. 40.4
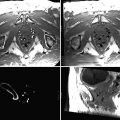
Example of clipping for ruptured AcoA aneurysm: 65-year-old patient presented with a SAH Fisher grade 4, WFNS grade 4. (a) Preoperative 3D angiogram. Anterior communicating artery aneurysm. Dimensions: 4.7 × 3.6 mm with a neck of 3.2 mm. (b) Intraoperative 3D RA with reconstruction. Anterior view. Image showing the clipped aneurysm with apparently no residual neck. (c) Intraoperative 3D angiogram with reconstruction. Posterior view, revealing a residual posterior neck (“dog ear”). (d) Second intraoperative 3D angiogram with reconstruction. Posterior view. Complete occlusion of the aneurysm

Fig. 40.5
Example for change of intraoperative strategy from open surgical clipping to endovascular rescue coiling of a ruptured left-sided MCA aneurysm, where the hybrid room proved essential: 54-year-old patient, SAH, Fisher grade 4, WFNS grade 4. In our institution a surgical indication. However, the patient presented 2 rebleedings during craniotomy and prior to dural opening. Intraoperative CT and angiogram was performed after dural opening, when extensive brain swelling and protrusion had occurred. Rescue coiling was then decided for the aneurysm in conjunction with decompressive hemicraniectomy. The patient survived with permanent hemiparesis. (a) Preoperative 3D angiogram. Middle cerebral artery aneurysm. Dimensions 8.7 × 6.3 mm with a neck of 4.3 mm. (b) Intraoperative XperCT showing severe brain edema. (c) Intraoperative 3D RA showing a metallic marker positioned at large distance from the MCA bifurcation (d) Intraoperative angiogram. Working oblique projection. (e, f) Intraoperative angiogram after rescue coiling. Working oblique projection. Complete aneurysmal occlusion

Fig. 40.6
Female patient, 22 years, SAH Fisher grade 4 (a), WFNS grade 2. (b) Angioscan demonstrating a small pericallosal aneurysm with a large neck with surgical indication. (c) 3D angiographic reconstruction after clipping, demonstrating a stenosis of the pericallosal. (d) Intraoperative angiographic control 15 min after the previous one. Lateral view. Occlusion of the pericallosal artery. (e) Intraoperative angiographic control. Lateral view. Intra-arterial injection of 2 mg of r-tPA with recanalization of the pericallosal artery. (f) Intraoperative angiographic control. Lateral view. Final control with proof of arterial patency
One surgical series reported a modification of the surgical procedure by clip repositioning or placement of additional clips in 24 cases (26 %) out of 91 operations [4]. They reported the results of successive angiograms when need and a second intraoperative angiographic procedure were conducted in 19 cases (21 %), and the surgical procedure was once again modified in eight cases (8 %). Tang et al. reported a consecutive series of 517 patients with intracranial aneurysms clipped with intraoperative angiographic control where it had led to modifications with clip replacement in 64 (12.4 %) patients. Proximal CA location demonstrated the highest rates of revision accordingly to intraoperative angiography with revision rates around 40 %. The revision rates in the remainder of the anterior circulation were lower, ranging from 4.3 % in PCoA aneurysms to 16.7 % at the ICA terminus. Concerning the size, giant and large aneurysms were frequently associated with clip revision. Different series reported 24.4 % of global revisions after multimodal intraoperative monitoring (intraoperative angiography and ICG) [5].
The accuracy of the intraoperative angiography was evaluated in a series where repeated angiograms were obtained in 95 patients postoperatively and they corresponded in 95 % [4]. This small difference can be explained by the absence of 3D imaging in that series, which is a valuable tool to evaluate the vessels and aneurysm residuals without interference of incidences, superposition, or 2D artifacts. Figure 40.4 shows an example of a posterior residual in aneurysm of the anterior communicating artery visible only due to the 3D rotational image and otherwise undetectable on 2D. Unexpected findings in postoperative consecutive angiograms have been reported to be around 12 % [2, 9]. Macdonald reported on 66 patients harboring 78 cerebral aneurysms who underwent postoperative angiography [13]. The benefit of intraoperative angiography is well demonstrated, and we advocate for all surgical procedures for treatment of intracranial aneurysms [1].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree