images with optimal renal parenchymal enhancement as well as excreted contrast material in the urinary tract. The recent development of iterative reconstruction techniques has the potential to allow for reduced dose CT imaging of the kidneys and urinary tracts with preserved image quality.6
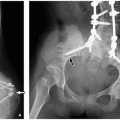
assess the male urethra in the setting of trauma (e.g., pelvic or perineal injury with hematuria) or suspected stricture.
TABLE 17.1 Routine Pediatric Voiding Cystourethrogram Images Acquired in Children with Typical Renal and Urinary Tract Anatomya | ||
|---|---|---|
|
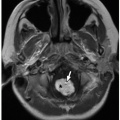
on longitudinal ultrasound images early in life (Fig. 17.2).15 Contralateral renal parenchymal hypertrophy is often present, even early in life. CT and MRI demonstrate similar findings and can be used to search for residual renal tissue, if clinically necessary. There is no specific medical or surgical therapy for renal agenesis, and prognosis is generally good assuming the contralateral kidney is normal. Care should be taken to preserve contralateral kidney function.
 FIGURE 17.2 1-day-old girl with right renal agenesis. No kidney is identified in the right renal fossa, and the right adrenal gland (arrows) appears abnormally elongated. |
ureteric insertion, ureterocele, or severe upper urinary tract narrowing) with resultant disordered assembly of renal tissue elements. While sometimes isolated, renal dysplasia can be associated with a variety of predisposing conditions, including upper urinary tract duplication (upper moiety parenchyma is most often dysplastic), posterior urethral valves, prune-belly syndrome, and mullerian anomalies.16,17 Microscopically, immature renal tubules embedded in primitive stroma, islands of cartilage, epithelium-lined cysts, and large aberrant blood vessels may be seen (Fig. 17.4).
of malignancy.20,21 In the setting of unilateral renal dysplasia, care should be taken to preserve contralateral kidney function.
identified by CT and MRI. Suspicion should be high when imaging reveals an empty renal fossa. Ectopic kidneys are commonly nonrotated or malrotated, and they may have mild pelvicaliectasis, even in the absence of urinary tract obstruction or VUR (Fig. 17.8). The arterial supply and venous drainage of ectopic kidneys are usually anomalous, with renal arteries arising from nearby major arterial structures and renal veins draining to nearby major systemic venous structures. Ectopic kidneys are often asymptomatic but may be complicated by ipsilateral VUR, UPJ obstruction, and urolithiasis.25 Small, poorly functioning malpositioned kidneys rarely can present with urinary dribbling and/or incontinence due to an associated ectopic ureter.26
kidneys may be associated with VUR, UPJ obstruction, and urolithiasis. Cross-fused renal ectopia also generally requires no specific treatment.
often associated with infection by Proteus mirabilis, a ureasplitting bacterium that forms struvite calculi. At histology, XGP is characterized by the presence of chronic inflammation, including lipid-laden macrophages. At imaging, a large obstructing calculus, sometimes staghorn in appearance, classically is present in the renal pelvis. The affected kidney usually appears diffusely enlarged and contains numerous round areas of decreased echogenicity at ultrasound or decreased attenuation at CT that are due to severe hydronephrosis (“bear paw” sign) and parenchymal necrosis (Fig. 17.15). Extensive perinephric inflammatory changes are typically present, and abscess formation may occur in the perinephric space, adjacent psoas muscle, and even body wall. Delayed contrast-enhanced CT and renal scintigraphy reveal minimal or no renal function, and radical nephrectomy is generally indicated for definitive treatment. A minority of XGP cases are focal, involving only a portion of the kidney, sometimes appearing mass-like.36
Ascending fungal infection within the renal collecting system is best appreciated by ultrasound, sometimes presenting as nonspecific echogenic debris in the urine or as an echogenic, circumscribed, sometimes mobile fungus ball (mycetoma) (Fig. 17.16).38 On occasion, ascending infection can infiltrate the renal medulla. Renal or perinephric abscesses due to fungal infection can rarely occur. Treatment is typically antifungal medical therapy, although, in certain children, asymptomatic funguria is due to colonization and does not require medical therapy. Rarely, such fungal infections can result in urinary tract obstruction or nonresolving abscesses that require percutaneous drainage or surgical management.39
hemihypertrophy, macrosomia, midline abdominal wall defects, ear anomalies, and/or neonatal hypoglycemia), sporadic aniridia, WAGR syndrome (Wilms tumor, Aniridia, Genitourinary anomalies, and mental Retardation), and Denys-Drash syndrome (gonadal dysgenesis, mesangial renal sclerosis leading to nephrotic syndrome and chronic kidney disease). All of these predisposing conditions relate to abnormalities of chromosome 11 and the WT1 and WT2 genes.54,55
and moderate specificity for identifying preoperative Wilms tumor rupture, with extension of ascites beyond the pelvic cul-de-sac being the best indicator.58
rind-like soft tissue abnormality replacing the renal cortex (Fig. 17.28).61,62
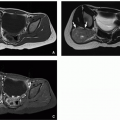
lymphatic invasion. Renal parenchymal satellite lesions are also common.73 This neoplasm has an extremely poor prognosis with survival typically <6 months, and it is frequently metastatic at the time of diagnosis.74
include a solitary renal mass or large retroperitoneal mass engulfing the kidney.80 Nearby retroperitoneal lymph node enlargement is common. Renal lymphomatous deposits typically regress with chemotherapy.
TABLE 17.2 American Association for the Surgery of Trauma Kidney Injury Grading Scale | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||
vary in size from very small to quite large.85,86 Extravasation of intravascular contrast material on contrast-enhanced CT suggests active bleeding that may require treatment (e.g., arterial embolization). On occasion, hemorrhage may be confined between the renal capsule and underlying parenchyma. Subcapsular hematomas are generally crescentic or elliptical in shape and exert mass effect upon underlying renal parenchyma (Fig. 17.38). Acute and subacute subcapsular hematomas demonstrate higher attenuation than water at CT. On occasion, subcapsular hematomas and associated renal compression may cause renal failure and renin-mediated hypertension (so-called Page kidney).87
blood flow as well as “arterialization” of venous blood flow with increased turbulence (Fig. 17.39).85 Many of these lesions are small and involute without intervention, although large lesions may persist and require arterial embolization.86
often diagnosed during adulthood, it can present during childhood. ADPKD is classified as a “ciliopathy” and can be due to a mutation in either the PKD1 (85%) or PKD2 (15%) gene.91
 FIGURE 17.42 9-year-old girl with enuresis. Longitudinal gray-scale ultrasound image through the left kidney lower pole shows a complicated cyst (arrows) containing multiple septations. |
have less severe renal involvement. Such abnormalities include congenital hepatic fibrosis and bile duct ectasia (Caroli syndrome), with associated splenomegaly, portosystemic varices, and ascites due to portal hypertension.96 Almost all individuals affected by ARPKD require dialysis or renal transplantation by adulthood, sometimes very early in life.
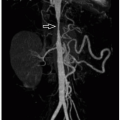
measure pressure gradients, as well as evaluate the aorta (Figs. 17.48 and 17.49). CBDSA has superior spatial resolution compared to other imaging modalities, detects accessory renal artery stenoses, and allows for simultaneous endovascular therapy of certain lesions.102 Renal artery stenoses are frequently intrarenal in children,102 and bilateral lesions occur in about 40%.103 Aneurysm formation is also common, often poststenotic in location (Fig. 17.49).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree