Basic Principles and Objectives
Primary breast carcinoma is a systemic, heterogeneous disease. It is considered a systemic disease because it is now known that even the smallest carcinoma can trigger metastases. Systemic medical treatment should therefore come after localized treatment of the breast carcinoma. Classes of substances available for systemic medical treatment are:
Cytostatics.
Hormones.
Antibodies.
New anticancer agents, such as angiogenesis inhibitors and receptor tyrosine kinase inhibitors.
Breast carcinoma is a heterogeneous disease in terms of its emergence, its individual characteristics, its treatment, and its course. The development of a breast carcinoma involves numerous cellular changes, on which the cancer cell depends completely for its survival and growth. Using molecular genetic identification of these changes and understanding how they interact, it is possible to define the breast carcinoma’s particular characteristics and from this to develop targeted points of attack for the therapy. The daily clinical routine includes, in particular, defining the cells’ immunohistological properties (hormone receptor status, HER2 status) and typing the carcinoma at the molecular biological level, which is increasingly important. Some of these points of attack will be dealt with in greater detail later in this chapter.
Cytostatic therapy follows a completely different approach. Empirically researched combinations of cytostatic substances damage the carcinoma tissue by interfering with various metabolic pathways that influence cell growth and cell division at different stages. In essence, the relatively selective effect of the cytostatics is achieved through the increased rate of cell division of the carcinoma cells.
In the drug-based treatment of breast cancer we can essentially differentiate four distinct disease categories with different therapeutic approaches and goals:
Adjuvant therapy of breast cancer: In adjuvant disease treatment, the primary breast carcinoma is first operated on and then treated by radiation therapy. The aim is cure. Both localized irradiation and the subsequent systemic therapy are preventive measures designed to reduce the risk of recurrence in a clinically tumor-free patient.
Neoadjuvant therapy of breast cancer: In neoadjuvant therapy, the systemic treatment precedes localized treatment. This form of therapy is also known as “primary systemic” or “preoperative” therapy. Here too, the therapeutic goal is cure of the patient. A certain histopathological diagnosis is always a prerequisite. Here too, the therapeutic goal is cure of the patient. Inflammatory and inoperable, locally far-advanced cases of carcinoma of the breast have always been indications for neoadjuvant chemotherapy. A further goal is to increase the rate of breast-conserving surgery 9: studies by the NSABP (National Surgical Adjuvant Breast and Bowel Project) group using primary systemic therapy have demonstrated efficacy equal to that of adjuvant therapy. 21, 22 The better the tumor’s response to the primary systemic therapy, the better is the patient’s prognosis.
Therapy of locoregional and thoracic wall relapses
Locoregional relapse is defined as the recurrence of the breast carcinoma in the remaining breast or axillary tissue on the same side. The therapeutic goal here is surgical cure. The 10-year survival rate is 40 to 58%. 66 With an isolated ipsilateral diagnosis the medical prognosis is considerably better. It is therefore vital to establish the absence of local and axillary tumors during aftercare examinations.
Thoracic wall relapse refers to recurrence of the breast carcinoma at the surgical site or in the irradiated area after a mastectomy. With isolated occurrence and successful removal of the recurrent lesion, there is a good chance of cure and a 5-year survival rate is about 65%. 52 The critical prognostic indicator here is the occurrence of distant metastases—approximately one-third of patients already have distant metastases when diagnosed.
Treatment of distant metastases: Cure is not possible in this advanced stage of the disease with the current state of knowledge. The aim is to maintain the best possible quality of life for the patient for as long as possible. The decision on the most suitable form of therapy is made for each individual situation. The average life expectancy, depending on the affected organ, is about 2 years.
Take Home Point
General Recommendations in the Medical Treatment of Primary Disease
Pharmacologic treatment of the primary disease is by chemotherapy, endocrine therapy, and/or anti-HER2 antibody therapy, given in various sequences and combinations, either before or after surgery.
12.2 Adjuvant Drug Therapy
12.2.1 Adjuvant Chemotherapy
Adjuvant chemotherapy is the standard treatment for the following indications:
Hormone receptor-negative tumors (endocrine nonsensitive: ER- and PgR-negative).
Equivocally hormone receptor-negative tumors (equivocally endocrine-sensitive).
HER-2-positive tumors treated with simultaneous antibody therapy using trastuzumab, over a period of 1 year, combined with a guideline-appropriate polychemotherapy.
Nodal-positive tumors.
Grade: G3.
Young age of onset (below 35 years).
Luminal B tumors with a “high proliferation rate.”
Triple-negative tumors (ER-/PgR-negative, HER2-negative = basal type).
The current evaluations by the EBCTCG (Early Breast Cancer Trialists’ Collaborative Group) under the direction of the statistician Sir Richard Peto showed that women below the age of 50 years, in particular, benefit from adjuvant chemotherapy in terms of the risk of death and recurrence. However, even in postmenopausal and older patients, the benefits of standard chemotherapy outweigh the higher rate of side effects when the corresponding risk constellation is taken into account.
Based on current understanding, chemotherapy should be started within the first 4 to 6 weeks after surgery, applied over 6 to 8 cycles, and repeated every 3 weeks. Standard drugs are anthracycline and taxane in combination or in sequence. The EBCTCG 2011 meta-analysis 16 evaluated the effectiveness of taxane for treating nodal-positive and nodal-negative breast carcinoma with high-risk constellations (e.g., G2/3; ER-/PgR-negative; pT stage above 2; age less than 35 years). 41 Taxane- and anthracycline-based regimes show a significant advantage in their effect on survival free from relapse and distant metastases and on overall survival compared with purely anthracycline-based regimes. Current studies corroborate these results.
More recent studies establish that dose-dense chemotherapy is beneficial in nodal-positive high-risk patients (four or more affected lymph nodes). 10, 43 Additional substances such as capecitabine and gemcitabine, and high-dose chemotherapy currently play no role in the adjuvant therapy of early breast cancer.
The conditions of therapy are being continuously improved through analysis of patients in clinical studies. As well as ensuring optimized care for the individual patient through participation in the study protocols per se, it also often provides the only means by which further data can be collected, new substances introduced, biomarkers determined, and unanswered questions resolved.
The meta-analyses from the EBCTCG have repeatedly shown that, for primary breast carcinoma, treatment with both modern polychemotherapy and endocrine therapy can significantly reduce the cumulative 15-year mortality risk by about one-third. 18, 53
Take Home Point
Systemic Adjuvant Therapy—What We Know For Sure
The use of systemic therapy reduces the relapse and mortality rates. This applies to polychemotherapy, in particular with anthracycline and taxane, the use of drugs to eliminate ovarian function, tamoxifen, aromatase inhibitors, and trastuzumab.
The indication for adjuvant systematic therapy is determined by considering the risk profile of the individual breast cancer patient and the likely benefits of this therapy. Even with current established methods it is not really possible to determine exactly which patient groups would not benefit from this therapy (due to overtreatment or lack of response). 46 The following classical clinical pathological parameters are considered in assessing the patient’s individual risk rating:
Tumor size.
Lymph node status.
Tumor grade.
Hormone receptor status.
HER2-status.
Menopausal status.
Age.
These are used to decide whether medication is necessary and the type of drug to be used. In every case, the need for the adjuvant therapy must be described to the patient in a detailed explanatory consultation; the side effects and the possibility of long-term health issues must be explained as well as the benefits.
Following the recommendations of the 2011 St. Gallen Consensus Conference, 28 it is for the first time possible to accommodate the types of breast carcinoma as defined by immunohistochemical properties in the decision making process. The immunohistological markers used for this are:
ER.
PgR.
HER2.
Ki-67 (as a proliferation index).
Along with the classic categorization, certain constellations of markers are increasingly being integrated into the nomenclature. These new classification subtypes are also referred to as “intrinsic types” of breast carcinoma (▶ Table 12.1).
Breast carcinoma type | ER | PgR | HER2 | Grade | Ki-67 (%) |
Luminal A | + | + / − | − | 1–2 | <14 |
Luminal B–HER2-negative | + | + / − | − | 1–3 | ≥14 |
Luminal B–HER2-positive | + | + / − | − | 1–3 | Any value |
HER2-positive | − | − | + | 1–3 | Any value |
Triple negative or basal type | − | − | − | 1–3 | Any value |
Abbreviations: ER, estrogen receptor; PgR, progesterone receptor; HER2, human epidermal growth factor receptor 2; Ki-67 (%), proliferation index (a nuclear antigen). | |||||
It is important to realize, however, that there is currently no validated threshold level for the Ki-67 labeling index, and that indices between 10 and 25% have no clear predictive value as to the proliferation rate.
12.2.2 Neoadjuvant Therapy
Systemic neoadjuvant therapy includes all the types of medical treatment that are administered after the confirmed histological diagnosis of a breast carcinoma but before any surgical measures. Here too, the therapeutic goal is cure.
Systemic neoadjuvant therapy has for decades been the standard treatment in patients with a locally advanced, primary inoperable or inflamed breast carcinoma. In these patients, the therapeutic goal was, through pharmacologic tumor reduction, to achieve an operable state that enables the complete excision of the carcinoma. Numerous studies on this concept have shown not only that operability can be improved but that neoadjuvant therapy offers additional advantages:
Improved options for breast-conserving surgery.
Individualization of the therapy through regular in vivo testing and monitoring of response.
Faster validation of new pharmaceuticals in the course of clinical trials.
In terms of the long-term survival rate, there are no differences between the adjuvant and neoadjuvant therapy experiences. Accordingly, neoadjuvant chemotherapy is now regarded as being equivalent in standing to adjuvant therapy.
Take Home Points
General Recommendations and Findings on Systemic Neoadjuvant Therapy
Neoadjuvant (primary, preoperative) systemic therapy is considered the standard treatment for patients with locally advanced, inoperable primary, or inflammatory breast carcinoma in the course of a multimodal therapy plan.
If chemotherapy is the indicated treatment, it can be given either before (neoadjuvant) or after surgery (adjuvant). The two methods are of equal value in terms of overall survival rates, but neoadjuvant therapy can result in a higher rate of breast-conserving treatment.
The effect is greatest with hormone receptor-negative carcinoma.
A resection within the new tumor boundaries is possible if a R0 resection can be achieved with a sufficient safety margin.
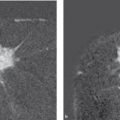
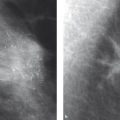
According to the Breast Committee of the AGO (Arbeitsgemeinschaft für gynäkologische Onkologie [Working Group for Gynecological Oncology]), according to international guidelines, suitable chemotherapeutic regimens are combinations containing anthracycline and taxane. Participation in clinical studies is strongly recommended. The neoadjuvant therapy should take place for a minimum of six cycles prior to surgery. Patients with a hormone receptor-positive carcinoma who respond well after two cycles of TAC (a combination of docetaxel 75 mg/m2 BSA + doxorubicin 50 mg/m2 BSA + cyclophosphamide 500 mg/m2 BSA every 3 weeks; BSA, body surface area) will benefit even more from eight cycles, according to data from the neoadjuvant GePAR-TRIO Study. 64 The therapeutic effect (therapeutic response) should be checked after two cycles of therapy and regularly thereafter. Clinical examination (including palpation), breast ultrasound, and mammography are well suited for this. If there is no response, action must be swift, with a corresponding adjustment to the neoadjuvant therapy or, depending on clinical findings, cessation of the treatment. This is where the advantages of neoadjuvant therapy come to the fore: the sensitivity of the tumor to the chemotherapy can be assessed and corresponding action can be taken. During a course of adjuvant therapy, a lack of response cannot be detected. However, the response of the tumor stem cells, which are responsible for the metastatic spread, is not evaluated in primary systemic therapy.
When there is lack of clinical response following two cycles of neoadjuvant chemotherapy, one can consider switching to a non–cross-resistant scheme with reduced side effects. In the GePAR-TRIO Study, 32, 62 it was shown that switching the chemotherapy from two cycles of TAC to four cycles of vinorelbine/capecitabine (days 1 and 8: 25 mg/m2 BSA; days 1 to 14: 2000 mg/m2 BSA, every 3 weeks) resulted in significantly prolonged relapse-free and overall survival rates. Patients with hormone receptor-positive breast carcinoma benefited particularly well from the changeover. However, the prognosis was not improved by the change in treatment for patients with triple-negative breast carcinoma, who showed no corresponding effects after two cycles of TAC therapy. A large proportion of patients suffered recurrence and metastases.
Pathological complete response (pCR) is defined as the absence of invasive tumor cell in the breast and the axilla following the completion of a course of neoadjuvant chemotherapy. 65 The pCR rate is a validated surrogate marker for the response to neoadjuvant chemotherapy and the long-term survival rate. Patients who respond after only two to four cycles of neoadjuvant therapy, have an approximately 4 times higher chance of a pCR and can be expected to have a very favorable long-term prognosis. This is especially true for patients with a HER2-positive/hormone receptor-negative or triple-negative breast carcinoma (basal type) and, with newly available data, also for those with Luminal B-/HER2-negative tumors. 65
Note
All patients whose breast carcinoma shows no change after the first cycles of neoadjuvant therapy have a distinctly worse prognosis.
The pCR rate serves as a surrogate marker in patients with the following tumor constellations:
Hormone receptor negative/HER2-positive.
Triple negative.
Luminal B/HER2-negative.
The pCR rate is not useful as a surrogate marker in Luminal A- and Luminal B/HER2-positive tumors.
Other predictive factors for a high likelihood of pCR have been collected by the Breast Committee of the AGO: in addition to younger age, smaller tumor size, higher grading, nonlobular histology, triple negativity, and positive HER2-status, the most important predictive factor for the effectiveness of neoadjuvant chemotherapy containing taxane–anthracycline is a negative hormone receptor status. A pCR rate of up to 40% can be achieved in patients who have a negative hormone receptor status.
If there is progression, or after completion of the neoadjuvant therapy, plans should be made for surgery. Before surgery, tumor localization should be precisely documented, for example, using pretherapeutic clip marking. The extent of the surgery should be determined by the effectiveness of the neoadjuvant therapy and should be carried out within the new tumor boundaries. The indications for postoperative systemic and local therapeutic measures are the same as for adjuvant therapy. 37
Neoadjuvant Therapy for HER2-Positive Breast Carcinoma
Standard treatment for patients with breast carcinoma overexpressing HER2 includes additional therapy with the antibody trastuzumab. This should be given preoperatively, simultaneously with chemotherapy; it significantly improves the pCR and survival rates. 65 These conclusions are from a meta-analysis data of 6,377 patients from seven German breast cancer studies involving neoadjuvant therapy. As with the adjuvant therapy, the antibody therapy should be continued for a full year. 1
Neoadjuvant Endocrine Therapy of Hormone Receptor-positive Breast Carcinoma
Endocrine therapy is a further neoadjuvant therapeutic option. Postmenopausal, highly hormone receptor-positive patients with locally advanced, primary inoperable or inflammatory breast carcinoma, where surgery or chemotherapy are contraindicated or have been declined, will benefit from endocrine therapy. With this indication, therapy with third-generation aromatase inhibitors for longer than 3 months is recommended. 55
12.2.3 Adjuvant Endocrine Therapy
According to a meta-analysis of the EBCTCG, adjuvant hormonal therapy with tamoxifen can significantly reduce the cumulative risk of death over a 15-year period in patients with a hormone receptor-positive breast carcinoma by about one-third or by an absolute 9.2%. This effect is independent of the age of the patient, the nodal status, the menopausal status, the size of the tumor, and the administration of adjuvant chemotherapy.
Take Home Point
General Recommendation for Endocrine Treatment of Receptor-Positive Tumors
Endocrine treatment is indicated in patients with estrogen receptor-and/or progestogen receptor-positive tumors.
As part of the primary diagnosis process, the hormone receptor status is determined by way of a quality-controlled evaluation of the paraffin section or of tissue previously obtained from a core needle biopsy. The content of ER(α) and PgR in the cells is determined immunohistochemically. If no receptor staining is observed (ER content 0%), then this form of therapy is ineffective.
The progesterone receptor, PgR, has particular prognostic significance in breast carcinoma patients—a less favorable prognosis may be assumed in the absence of PgR expression.
Note
A response to endocrine therapy is assumed if the cell ER content is >1%, with endocrine sensitivity correlating with the strength of the receptor expression. 60
At the time of diagnosis, approximately one-third of all breast cancer patients are pre- or perimenopausal. About 60% of the tumors in these patients overexpress the ER. The goal of systemic endocrine therapy is blockade of the intracellular ER of the tumor cells and/or reduction of the circulating estrogens to a postmenopausal level of 20 pg/mL or less. Before endocrine therapy with aromatase inhibitors is proposed to a patient, her menopausal status should be clarified and an exact menstrual history should be documented. It should be noted that, in women younger than 50 years, chemotherapy can induce amenorrhea with high levels of follicle-stimulating hormone (FSH) and luteinizing hormone (LH). In these women, even years later, follicles can continue to ripen, producing estrogen. This process is supported by the administration of aromatase inhibitors. Thus, high estrogen levels in association with aromatase inhibitor therapy can be fatal because they stimulate tumor growth. 56 Only from the age of 55 years can a patient reliably be considered to be postmenopausal.
Endocrine Therapeutic Agents
Tamoxifen
The standard therapeutic option for patients who are premenopausal is selective ER-blockade with tamoxifen, using a dosage of 20 mg/day over a period of 5 years.
Take Home Point
General Recommendations for Endocrine Therapy in Premenopausal Patients
In premenopausal patients, tamoxifen is the endocrine therapy of choice. Antiestrogen therapy with tamoxifen at 20 mg/day should be administered over a period of 5 years or until a relapse.
As a SERM (selective ER modulator), tamoxifen competitively inhibits the binding of estrogens to hormone receptors, so with its use there is a decrease in cell division in estrogen-dependent tissues, leading to apoptosis of the corresponding tumor cells. Tamoxifen is a prodrug; in an initial reaction, it is converted by the cytochrome P450 enzyme CYP2D6 into its active metabolites, endoxifen and 4-hydroxytamoxifen. In addition to its antiestrogenic effect, tamoxifen also exhibits a slight organ-specific agonistic, estrogenic effect. Because of this, on the one hand tamoxifen exerts osteoprotective properties, while on the other it leads to an increase in endometrial changes and thromboembolic events (in ca. 1 to 3% of cases). The relative risk for the occurrence of endometrial carcinoma after a 5-year course of tamoxifen therapy is 2.2 compared with the average for the population. This risk is low in contrast with its benefits (reducing the risk of recurrence of breast carcinoma), and is only relevant from the age of 55 years and above. Furthermore, the mortality rate is not increased by these side effects. 16
The conclusions of the Consensus Conferences in St. Gallen in 2005 and 2007 are helpful in developing treatment recommendations (▶ Table 12.2). The current AGO recommendations 1 extend the indication spectrum for the sole use of an endocrine therapy with tamoxifen to patients with a moderate risk of recurrence:
Patients with high risk: According to the current AGO treatment recommendations 1, the standard treatment for premenopausal patients with an endocrine-sensitive breast carcinoma and a high-risk constellation is adjuvant sequential chemoendocrine therapy with tamoxifen over a period of 5 years. However, the current EBCTCG meta-analysis 16 does not demonstrate any disadvantage in using different therapy types simultaneously.
Patients with moderate risk: These patients can be treated either with a sequential chemoendocrine therapy or with endocrine therapy alone, depending on the individual risk assessment, the level of endocrine sensitivity, and the safety considerations. 1
Patients with low risk: Endocrine therapy with tamoxifen alone or combined with GnRH analogs may be recommended to these patients (see the following section). In this group all of the following conditions must be met:
Age at least 35 years.
Tumor size no greater than 2 cm (T1).
A good degree of differentiation (G1).
ER and/or PgR positive.
HER2/neu status negative.
No positive axillary lymph nodes (N0).
Practical Tip
Patients who have a moderate risk profile, for whom there are contraindications to chemotherapy, who decline chemotherapy, or who belong to a lower risk group can be offered tamoxifen monotherapy as an alternative.
Table 12.2 Risk categories (consensus of 2007 St. Gallen Conference 26)
Status
Low risk
Moderate risk
High risk
Nodal status
0 and all other criteria
1–3 and HER2-negative or 0 with at least one other criterion
≥4, or 1–3 and HER2-positive
Tumor size (cm)
1 (maximum 2)
2–4
Degree of differentiation
1
2 and 3
Vascular collapse
0
1
Hormone status (ER/PgR)
+
HER2/neu status
−
+
+
Age (years)
≥35
>35
<35
Gonadotropin-Releasing Hormone (GnRH) Analogs
Ovarian ablation is another proven therapeutic principle for hormone suppression, but it has not yet been definitively established when GnRH analogs should be used and for which indication.
GnRH analogs are synthetically produced derivatives of the neurohormone GnRH, which suppress the gonadotropic function of the pituitary gland and thereby ovarian function. These analogs bind to the pituitary receptor with a 100 times greater affinity than natural GnRH and exhibit a longer half-life. After the initial binding there is a temporary increase in release of FSH and luteinizing hormone (“flare-up effect”), but in the course of therapy there is a reduction in the number of receptors. The secretion of the pituitary hormones, LH and FSH, is thereby permanently inhibited, preventing the maturation of ovarian follicles and the associated estrogen production. A hypogonadotropic, hypoestrogenic state is achieved within 2 weeks that is similar to that of a postmenopausal woman. The effects of a GnRH analog therapy are reversible and there are relatively few side effects; radiotherapeutic ovarian ablation or oophorectomy provides irreversible suppression.
A course of treatment with a GnRH analog over a 2- to 5-year period in addition to tamoxifen therapy to eliminate ovarian function would appear to benefit only premenopausal, hormone receptor-positive, and nodal-positive breast cancer patients who are younger than 40 years of age. 8, 14, 15