INTRODUCTION
 Imaging Agents
Imaging Agents
Technetium-99m MDP/HDP
Bone scintigraphy is most often performed utilizing technetium-99m (Tc-99m) methylene diphosphonate (MDP), or Tc-99m hydroxymethylene diphosphonate (HDP). Both compounds are diphosphonates (bisphosphonates) that show chemisorption to hydroxyapatite and chelate the radionuclide Tc-99m for imaging purposes.1 Tc-99m has ideal imaging properties secondary to its relatively low energy and abundant gamma ray at 140 kilo electron volts (keV), its short 6 hour half-life, and its ready availability in generator form. The resulting chelates show an in vivo distribution of radioactivity that reflects the osteoblastic activity of bone with relatively high sensitivity but low specificity. This low specificity often requires correlation with other imaging studies and patient history to better define lesion etiology.2 The usual administered adult dose is approximately 740-1100 megabecquerels (MBq), [20–30 millicuries (mCi)], resulting in an effective total dose of approximately 5.9-8.8 millisieverts (mSv).3
Indium-111-Labeled White Blood Cells
An extremely useful method for imaging inflammation/infection is the utilization of radiolabeled white blood cells (WBC). The vast majority of white cells extracted from peripheral blood are polymorphonuclear leukocytes (PMNL) that are chemotactically attracted to foci of most acute inflammatory/infectious processes. Both Tc-99m hexamethylpropyleneamine oxime (HMPAO) and indium-111 (In-111) oxyquinoline are lipophilic compounds that may be used to label isolated peripheral WBCs. For imaging musculoskeletal infections, In-111 is felt to be the optimal radionuclide because of its longer half-life of 67 hours and its medium energy gamma rays at 174 and 247 keV.1 The longer half-life allows more time for labeled leukocytes to localize in regions of osteomyelitis that may have overall lower numbers of associated WBCs compared with acute pyogenic abscesses. The medium energy photopeaks allow for simultaneous image acquisition with Tc-99m-labeled bone marrow for improving the specificity of osteomyelitis detection. This technique poses moderate risk for the patient. Proper labeling of peripheral WBCs is technically complex and is usually performed at experienced tertiary commercial radiopharmacies. This requires collection and centralization of blood samples from multiple, usually highly infectious, patients daily with the associated risk of cross-contamination. Radiation-sensitive white cells, including a small number of lymphocytes, are internally exposed to ionizing radiation resulting in an unknown but assumable significant risk. The usual dose of In-111 for this procedure is 10-20 MBq (0.3–0.5 mCi), resulting in an effective total dose of approximately 6-12 mSv.4
Gallium-67 Citrate
Gallium-67 (Ga-67) citrate is a radionuclide that has been used for over 30 years for tumor and inflammatory imaging. Its localization in infectious or inflammatory lesions is felt to be secondary to multiple factors including binding to transferrin and lactoferrin, both found in high concentration in inflammatory foci. Direct uptake by iron binding bacteria as well as by activated phagocytic cells has also been seen. The imaging properties of Ga-67 are somewhat less than ideal with multiple low-to-medium energy gamma rays at 93 keV, 184 keV, 296 keV, and 388 keV, and a relatively long half-life at 78 hours. It continues to be useful in conjunction with the bone scan for detection of osteomyelitis in the spine and chronic osteomyelitis where the localization of PMNL may be low. It may also be used in immunocompromised patients with few circulating WBCs.1 The usual administered adult dose is 150–220 MBq (4–6 mCi), resulting in an effective total dose of approximately 18–26.4 mSv, similar to that of a diagnostic chest/abdomen/pelvis computed tomography (CT) study.5
Technetium-99m Sulfur Colloid
Tc-99m-labeled sulfur colloid is a radiopharmaceutical composed of small particles injected intravenously that are then phagocytized by reticuloendothelial cells resulting in a normal distribution to the liver, spleen, and bone marrow. Both In-111-labeled WBCs and Tc-99m sulfur colloid localize normally to a small degree in bone marrow. While labeled WBCs may accumulate in regions of osteomyelitis, Tc-99m-labeled sulfur colloid does not. The technique of simultaneous image acquisition of images of both radiopharmaceutical distributions has been shown to significantly improve the specificity of detection of osteomyelitis with In-111-labeled WBCs.6 The usual injected dose of Tc-99m sulfur colloid for this application is 300–370 MBq (8–10 mCi), resulting in an effective total dose of approximately 4.2–5.2 mSv.4
Fluorine-18 Sodium Fluoride
Fluorine-18 (F-18) is a positron-emitting radionuclide, which is administered intravenously in its ionic form as a sodium salt, followed by rapid and nonspecific binding to bone hydroxyapatite. This stable binding that forms fluoroapatite, results in an almost twofold greater detectable activity in bone compared with the Tc-99m-labeled agents. The uptake of F-18 is approximately three times higher in most bony metastatic lesions when compared with the immediately surrounding bone. The pharmacokinetics of F-18 are superior to those of the Tc-99m-labeled bisphosphonates with nearly 100% first-pass extraction from bone capillaries and a more rapid renal clearance of unbound tracer. These characteristics of F-18 result in a higher lesion to background ratio and a significantly shorter injection to imaging time.7 All of the kinetic energy of the positron and some energy from interactions of the resulting annihilation photons contribute to the total patient dose when using F-18 for bone scanning. If a concurrent CT study is performed, this also adds to the total radiation dose the patient receives for the study. The usual injected dose for an F-18 bone scan is 185–370 MBq (5–10 mCi), resulting in an effective total dose of approximately 4.4–8.9 mSv.8
Fluorine-18 Fluorodeoxyglucose
Fluorine 18-labeled fluorodeoxyglucose (FDG) was first introduced in 1976 as an experimental tracer and after the introduction of commercially available positron emission tomography (PET) scanners in the 1990s gained widespread use in tumor imaging. FDG is transported into cells by the glucose transporter protein one (GLUT1) and is then phosphorylated by hexokinase two (HKII) to FDG-6-phosphate, which is retained within the cell. Most tumor cells show high glucose metabolism with resulting up-regulation of GLUT1 and HKII activity, thus increased uptake and retention of FDG. Activated PMNL and macrophages also show up-regulation of these enzymes and therefore increased tracer accumulation.9 The usual injected dose for an FDG scan is 370–740 MBq (10–20 mCi), resulting in an effective total dose of approximately 7.0–14 mSv.10
 Imaging Techniques
Imaging Techniques
Planar Imaging
Planar or conventional bone nuclear medical imaging is widely available in nearly all medical facilities today. It is performed utilizing a single or multiple headed gamma camera acquiring individual static or multiple dynamic images of specific body regions. The technology of the gamma camera is largely unchanged since its introduction approximately 50 years ago with incremental increases in field of view and resolution over the years. Images are normally obtained approximately 3 hours after injection of the Tc-99-labeled bone scanning agents. Often bilateral images are obtained to allow direct comparison of the degree of radiopharmaceutical uptake in areas of interest.
Three-Phase Bone Scan
In three-phase bone scintigraphy, an initial dynamic set of images is obtained representing blood flow or hyperemia to the region of interest. Immediately following flow imaging, static images of the same area are acquired representing initial extracellular fluid pool extraction of the radiopharmaceutical. Together these imaging phases give an indication of the degree of inflammation in the region of interest. When combined with delayed phase imaging of underlying osteoblastic activity, the three-phase bone scan is highly sensitive for detection of osteomyelitis. The specificity of three-phase bone scans decreases when there are underlying perturbations of bone such as fractures, prostheses, or implants.1
SPECT and SPECT-CT Imaging
Single-photon emission computed tomography (SPECT) is an evolutionary technology utilizing widely available modified standard gamma camera equipment. Multiple planar images are acquired circumferentially around a subject, which are then computer reconstructed into a three-dimensional dataset. The data can be tomographically displayed in any orientation to aid in lesion detection and localization. This technology allows increased sensitivity for detection of small and/or low activity lesions, especially in deeper structures such as the vertebral column or pelvis. The hybridization of simultaneously acquired and displayed CT images aids in localization and lesion characterization. Newer dedicated hybrid SPECT-CT scanners including CT components capable of acquiring high-resolution diagnostic images are now becoming available.
PET-CT Imaging
PET is now a widely commercially available imaging technology with most large institutions capable of performing hybrid PET and computed tomography (PET-CT) studies with newer dedicated scanners. Hybrid imaging with a PET-CT scanner allows direct comparison of tracer uptake with co-registered detailed anatomic density images. Because of the very high bone to soft tissue ratio of F-18 bone scanning, a hybrid CT scan for attenuation correction is felt unnecessary. This technique results in a total radiation dose to the patient comparable to a standard Tc-99m bone scan. A hybrid CT study may be obtained with low dose technique to allow localization of lesions in whole body scans for metastatic disease. Most modern scanners may perform this CT study at total body radiation doses as low as 3 mSv or less. However, a diagnostic dose technique may be used at doses in the range of 5–20 mSv allowing full radiographic characterization of possible bony lesions if clinically indicated. A PET only F-18 bone scan may be immediately reviewed by the attending imaging physician, while the patient is still positioned in the scanner with focal repeat hybrid PET and full diagnostic CT images obtained of chosen regions for improved lesion characterization. This method would allow the greatest diagnostic sensitivity and specificity with the lowest achievable radiation dose possible.8
TUMORS
 Malignant Bone Tumors
Malignant Bone Tumors
Primary Bone Tumors
Sarcomas
The most common primary bone tumor in children is osteosarcoma, while in young adults Ewing sarcoma is more frequent. In adults, multiple myeloma is the most common primary bone tumor followed by osteosarcoma. Bone sarcomas are usually diagnosed and characterized utilizing conventional radiography, magnetic resonance imaging (MRI), and biopsy. Scintigraphy plays its largest role in staging primary bone tumors utilizing FDG PET-CT (Figure 20-1). Osteosarcoma most often metastasizes to the lung and here CT is the most sensitive imaging modality for detection. The second most common metastatic site is bone, where FDG PET-CT and/or F-18 PET-CT bone scanning are most sensitive for detection. Uptake of FDG in lung lesions detected at CT may also help to distinguish benign from metastatic lesions. Modern therapy for primary bone sarcomas often involves use of neoadjuvant chemotherapy. This treatment is often quite toxic and early detection of success indicated by a significant decrease in FDG uptake after several cycles compared with a baseline study has been shown to be a useful indicator (Figure 20-2).11
Figure 20-1. Ewing sarcoma of the right knee. (A) Axial-fused FDG PET-CT of Ewing sarcoma of the right lateral distal femur and knee joint capsule in a 21-year-old man showing moderate FDG uptake in the primary tumor measuring 2.9 SUV (see color insert). (B) CT soft tissue window axial section at the same level as that in A, showing stippled internal calcifications within the primary tumor mass (arrow).
Figure 20-2. Ewing sarcoma lung metastases. Summed coronal FDG PET maximum intensity projection (MIP) images from the same patient in Figure 20-1. (A) Subsequent development of multiple, primarily basilar, bilateral lung metastases with marked FDG uptake measuring up to 12 SUV. (B) Decreased uptake in the metastatic lung lesions after three cycles of neoadjuvant chemotherapy prior to surgical resection.
PEARLS
Multiple Myeloma
The most common primary bone tumor in adults is very often occult on conventional planar and SPECT bone scintigraphy. The primarily lytic marrow-based lesions of this tumor rarely show significant uptake on Tc-99m or F-18 bone scans (Figure 20-3). Until recently the relatively insensitive imaging modalities of whole body bone survey radiography and CT have been used to stage and assess response to therapy for multiple myeloma. Most multiple myelomas demonstrate avid uptake of FDG and this agent as well as MRI are showing promise as new gold standards for staging and evaluation of this tumor.11
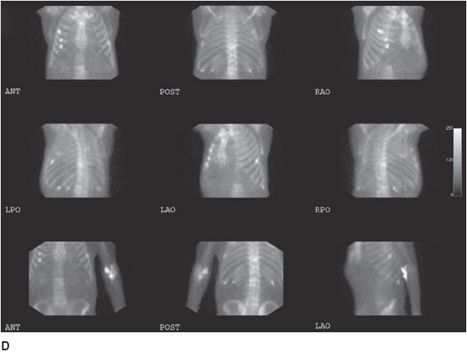
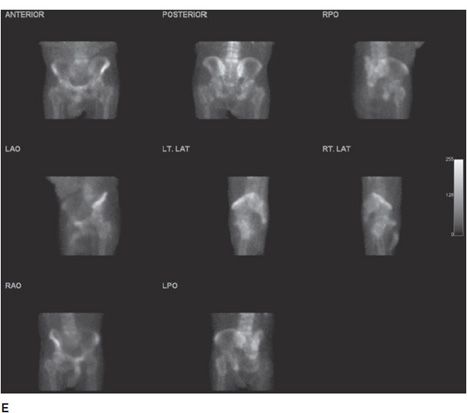
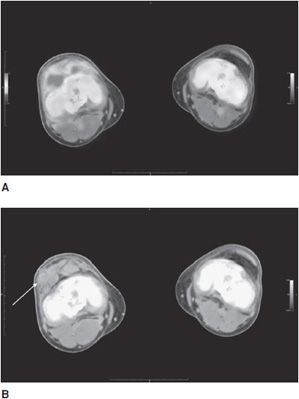
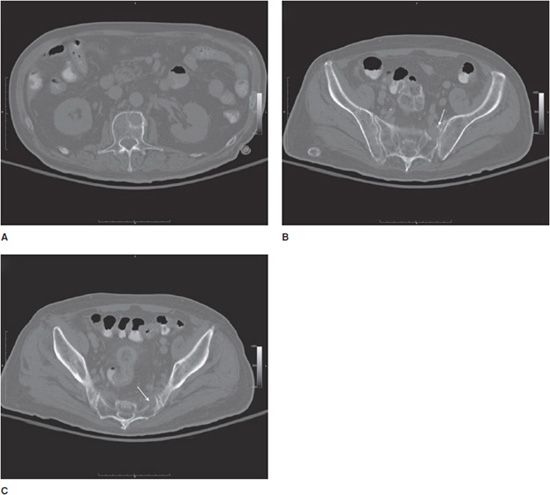
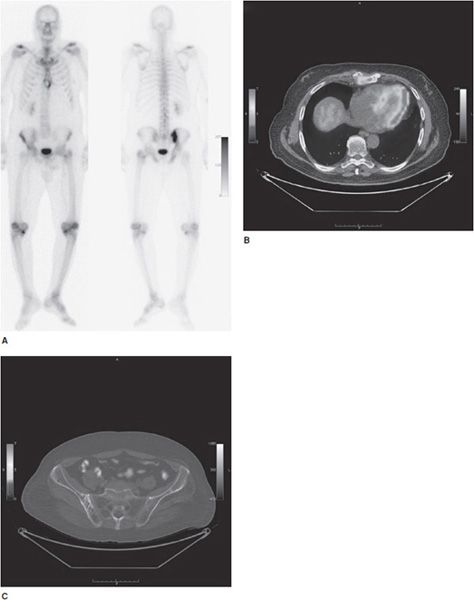
Figure 20-3. Multiple myeloma. Multiple axial images from a diagnostic CT in a 83-year-old patient with histologically proven multiple myeloma showing discrete lytic lesions. (A) Lytic lesion in the left anterior L1 vertebral body. (B) Lytic lesion in the left superior anterior sacral ala (arrow). (C) Lytic lesion in the left inferior anterior sacral ala (arrow). (D) and (E) are multiple orthogonal views from a concurrent Tc-99m MDP conventional bone scan. No significant increased uptake is seen in the L1 vertebral body. (D) Mild increased uptakes are seen in multiple ribs secondary to subacute trauma. Increased activity is also seen in the left ante-brachium representing mild extravasation of radiopharmaceutical at the injection site. (E) No increased uptake is seen in the left sacral ala.
 Metastatic Bone Tumors
Metastatic Bone Tumors
Many malignancies show a propensity to metastasize to bone. The most commonly diagnosed cancer, nonsmall cell lung carcinoma, has metastasized to bone at the time of diagnosis in approximately 20–30% of patients. Other commonly diagnosed carcinomas including differentiated thyroid carcinoma and renal cell carcinoma have metastasized to bone 15–30% of the time. Conventional bone scintigraphy and SPECT imaging with the Tc-99m agents show a very high sensitivity for detection of most bony metastases. However, F-18 bone scanning has been shown to have even greater sensitivity for detection of these lesions due to its superior pharmacokinetics and the now widespread use of high-resolution hybrid PET-CT scanners.9 For these reasons, routine skeletal surveys for metastatic disease utilizing F-18 PET-CT are likely to become the standard of practice (Figure 20-4). Some metastatic lesions, for example renal cell carcinoma or de-differentiated thyroid cancers, are very aggressive and primarily lytic when metastatic to bone. These lesions may show little uptake of the bone imaging agents, but because of their more aggressive nature they will usually show high affinity for F-18 FDG (Figure 20-5).12 The use of conventional scintigraphy or SPECT imaging with the Tc-99m agents, or the even more sensitive PET-CT studies with FDG or F-18, leads to detection of many benign bony lesions as well as metastatic disease. However, the specificity of all these studies for differentiating benign from malignant disease is highly dependent on careful radiologic correlation with co-registered hybrid CT imaging, other imaging modalities such as radiograph or MRI, patient history, and clinical exam results.
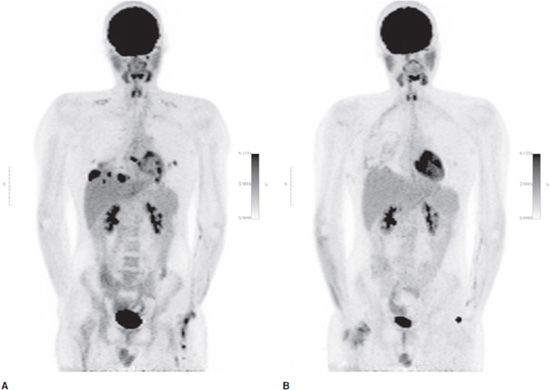
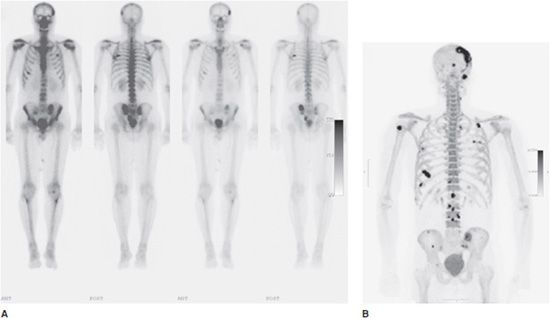
Figure 20-4. Metastatic prostate cancer. (A) Three-hour delayed whole body anterior and posterior images from a conventional bone scan in a 55-year-old patient with metastatic prostate cancer. The left set of images is reproduced at a higher intensity level to accentuate lower activity lesions. Moderate-to-marked activity is seen in metastatic lesions in the left skull, the bilateral posterior lateral sixth ribs, the anterior right sixth rib, the left medial iliac wing, and the left mid sacrum. (B) Summed coronal fluorine-18 PET maximum intensity projection (MIP) image from the same patient in (A) showing detection of multiple smaller skull, rib, spine, and pelvic lesions.
Figure 20-5. Bone metastasis from renal cell carcinoma. (A) Three-hour delayed whole body anterior and posterior images from a conventional bone scan in a 65-year-old patient with a history of renal cell carcinoma status post left nephroureterectomy now with sternal pain. The images show mild multifocal uptakes over the knees and shoulders, likely degenerative. There is mild uptake in a ring shape pattern over the xiphoid region and moderate-to-intense uptake in the region of the right ilium. These findings and the history warranted further investigation with FDG PET-CT imaging. (B) Axial-fused image showing an expansile lytic bony lesion of the xiphoid with moderate FDG accumulation measuring approximately 6.5 SUV, biopsy-proven metastatic renal cell carcinoma (see color insert). (C) Axial-fused image with bone CT window showing coarsened trabecular markings in the right ilium in the region of increased uptake on the bone scan without associated FDG accumulation. Findings more likely representing Paget disease than a metastatic lesion. In the case of both lesions, hybrid PET-CT imaging helped guide biopsy decisions and further diagnostic and prognostic information (see color insert).
 Benign Bone Tumors
Benign Bone Tumors
Osteoid Osteoma
Osteoid osteoma is a benign bone lesion with a vascular central nidus and surrounding reactive sclerosis. It is often associated with intense and focal pain that is relieved with nonsteroidal anti-inflammatory medications. Because of the vascular nature of the nidus, the lesion is often focally positive on the flow phase of the three-phase bone scan. Delayed phase imaging usually shows intense radiopharmaceutical localization due to the surrounding remodeling and sclerosis (Figure 20-6). For these reasons, three-phase bone scanning is highly sensitive for detection of this benign bone tumor.13
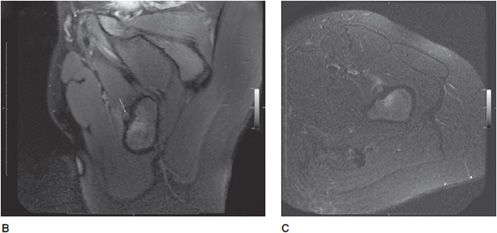
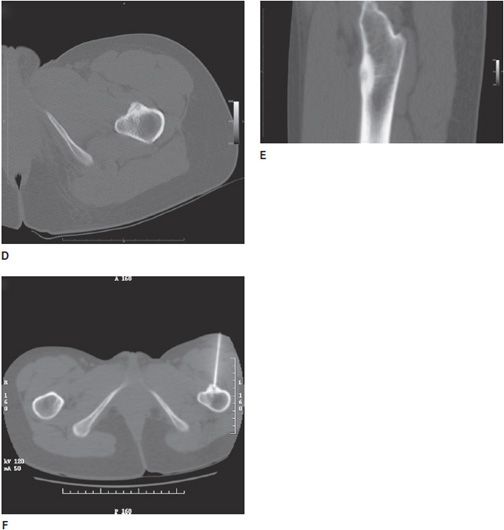
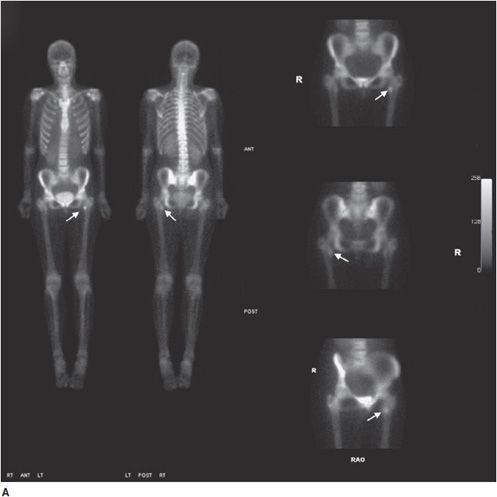
Figure 20-6. Osteoid osteoma. Multiple images obtained from a 25-year-old woman patient with a history of intermittent moderate-to-severe left hip pain, often more severe at night, and relieved after taking oral nonsteroidal anti-inflammatory medications. (A) Three-hour delayed phase conventional bone scan images showing somewhat atypically only moderate grade uptake over the anterior proximal metadiaphyseal cortex of the left femur (arrows). (B) Axial oblique fat-saturated proton density MR image through the lesion seen in the anterior proximal left femur in (A) showing bone surrounding a central nidus (arrow) in the anterior cortex with mild surrounding soft tissue and marrow changes. (C) Axial short-tau inversion-recovery (STIR) image through the lesion showing adjacent marrow and soft tissue edema. (D) Axial and (E) sagittal CT images through the lesion showing the dense nidus and surrounding dense reactive cortical bone. (F) Axial CT screen shot images showing interventional radiography-guided biopsy and radio-frequency ablation of the osteoid osteoma.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree