Abstract
This chapter describes diagnostic imaging for myocarditis. Various imaging modalities are discussed, comparing their specific advantages and disadvantages. For imaging acute myocarditis, cardiac magnetic resonance offers the greatest potential—compared to other cardiac imaging modalities. Current clinical status, persisting challenges, and future perspectives of imaging myocardial inflammation are reviewed.
Keywords
Myocarditis, Magnetic resonance imaging, Oedema, Echocardiography, Myocardial, Contrast enhancement.
14.1
Clinical background
14.1.1
Etiology and presentation
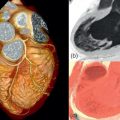
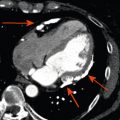
Acute myocardial inflammation can induce a broad clinical spectrum, from subclinical disease to fulminant acute heart failure [ ]. Myocarditis more often affects young male than female or elderly patients [ ]. The clinical presentation differs depending on age and gender [ ]. Young men often present with acute chest pain similar to myocardial infarction after a few days of airway or intestinal infection ( Figure 14.1a and b ) [ ]. In female patients, the clinical presentation is frequently less obvious and imaging findings are less impressive than in young males. On the other hand, some patients can present with fulminant myocarditis, requiring circulatory support in intensive care [ ]. In most cases, acute viral infection is the underlying mechanism, but drug-induced toxicity [ ] and autoimmune diseases have also a role and their prevalence as a cause of myocarditis is probably underestimated.

Myocarditis is an important differential diagnosis in acute coronary syndrome or acute heart failure. Non-invasive imaging plays an increasing role in ACS for the differentiation of the underlying injury. Once significant coronary artery disease (CAD) has been excluded, cardiac imaging is often requested to elucidate the reason for elevated troponin [ ]. The main objective of further testing is the safe and accurate identification of the cause of raised cardiac biomarkers, especially if invasive procedures are to be avoided. The order of the procedures (invasive vs. non-invasive) depends on several conditions, including clinical features and pre-test probability of disease. An intelligent and innovative setting is helpful for speeding up the diagnostic procedure and avoiding unneeded invasive procedures, especially in young patients.
14.1.2
Course of disease
The time course of myocarditis depends on the initiating cause and the patient constitution. The so-called “natural course” of disease typically lasts 5 weeks [ ]. In the majority of cases, patients do recover after 3–6 weeks. Prognosis also depends on the type of initial clinical presentation and LV function at baseline [ ]. In particular, young male patients with a presentation similar to a myocardial infarction most often recover despite troponin elevations [ ]. However, some patients may develop dilated cardiomyopathy. In some patients with fulminant myocarditis (mainly young females), ventricular dilatation and hypocontractility persist for several weeks or months but it often eventually recover to a normal or almost normal state [ ].
14.2
Biopsy as “the invasive standard”
Endomyocardial biopsy (EMB) is the only method that can identify the underlying cause of myocarditis and differentiate the causative organism [ ]. In experienced hands, this invasive procedure has a low risk [ , ]. The indication for EMB in clinical routine is limited due to sample error and the lack of therapeutic consequences. The currently accepted indications are published in a consensus statement of AHA, ACC, and ESC based on the description of 14 clinical scenarios ( Table 14.1 ) [ ]. According to guidelines, EMB is indicated especially if it may change management in unexplained heart failure, e.g., in suspected giant cell myocarditis. More data are required to develop new therapeutic strategies based on EMB and non-invasive imaging [ ]. It had been suggested that cardiovascular magnetic resonance (CMR) might guide the location of biopsies to increase sensitivity. However, inflammatory lesions often have an intramural or epicardial location less accessible to biopsy. In addition, the biopsy forceps are a rigid tool that cannot be directed to any location indicated by CMR. CMR might indicate whether left ventricular (LV) or right ventricular (RV) myocardial biopsy appears more promising in an individual case, but Yilmaz et al. demonstrated that biopsies did not yield higher sensitivity when taken from regions (septal or lateral wall) with positive late gadolinium enhancement (LGE) by CMR [ ]. In addition, they found that combined RV and LV biopsies were more sensitive than samples from one ventricle alone. In acute coronary syndrome without obvious etiology, biopsy and CMR may offer synergistic diagnostic power superior to either approach alone [ ]. Myocardial involvement in systemic disorders is more difficult to assess, especially in focal pronounced disease, such as sarcoidosis (see Chapter 15 ). Therefore, current recommendations are restrictive. EMB in a clinical setting is only indicated if a specific therapeutic consequence is expected mostly depending on patient hemodynamic stability, as indicated in Table 14.1 . Research settings will have other degrees of freedom. EMB allows the specific evaluation of the kind of virus and could stratify a specific therapy. But inflammation may be caused by viral and non-viral agents and the diagnostic performance is also related to the scatter of evaluated causes. There is no doubt that EMB as well as all imaging techniques also depends on the experience of the centre.
| Clinical scenario | Class of recommendation | Level of evidence | |
|---|---|---|---|
| 1 | New-onset heart failure of 2 weeks’ duration associated with a normal-sized or dilated left ventricle and hemodynamic compromise | I | B |
| 2 | New-onset heart failure of 2 weeks’ to 3 months’ duration associated with a dilated left ventricle and new ventricular arrhythmias, second- or third-degree heart block, or failure to respond to usual care within 1 to 2 weeks | I | B |
| 3 | Heart failure of 3 months’ duration associated with a dilated left ventricle and new ventricular arrhythmias, second- or third-degree heart block, or failure to respond to usual care within 1 to 2 weeks | IIa | C |
| 4 | Heart failure associated with a DCM of any duration associated with suspected allergic reaction and/or eosinophilia | IIa | C |
| 5 | Heart failure associated with suspected anthracycline cardiomyopathy | IIa | C |
| 6 | Heart failure associated with unexplained restrictive cardiomyopathy | IIa | C |
| 7 | Suspected cardiac tumors | IIa | C |
| 8 | Unexplained cardiomyopathy in children | IIa | C |
| 9 | New-onset heart failure of 2 weeks’ to 3 months’ duration associated with a dilated left ventricle, without new ventricular arrhythmias or second- or third-degree heart block, that responds to usual care within 1 to 2 weeks | IIb | B |
| 10 | Heart failure of 3 months’ duration associated with a dilated left ventricle, without new ventricular arrhythmias or second- or third-degree heart block, that responds to usual care within 1 to 2 weeks | IIb | C |
| 11 | Heart failure associated with unexplained HCM | IIb | C |
| 12 | Suspected ARVD/C | IIb | C |
| 13 | Unexplained ventricular arrhythmias | IIb | C |
| 14 | Unexplained atrial fibrillation | III | C |
14.3
Therapeutic options
The therapy depends on the clinical situation of an individual patient. The clinical picture ranges from cardiogenic shock over mild symptoms to asymptomatic disease. A fulminant myocarditis needs full intensive care support, potentially including assist devices. According to EMB guidelines, a specific diagnosis is warranted in these patients. For example, giant cell myocarditis is a rare, but life-threatening condition requiring application of steroids. In nearly all patients, including those with mild symptoms, heart failure medication is administered. The duration and extent of therapy depends on individual circumstances [ ]. It is common sense that physical activities should be avoided in the acute stage. Defining the appropriate time of restarting exercise is often difficult [ ]. Data regarding this problem are scarce. The only trial is based on mice experiments which indicate that physical activity in myocarditis increased mortality [ ].
In patients with myocardial involvement in systemic disorders, an intensification of the applied disease specific medication is usually recommended. A detailed discussion based on clinical stages and/or on the likelihood of the disease was published by Sagar et al. ( Table 14.2 ) [ ].
| Criteria | Histological confirmation | Biomarker, ECG or Imaging abnormalities needed | Treatment | |
|---|---|---|---|---|
| Possible subclinical acute myocarditis | In the clinical context of possible myocardial injury without cardiovascular symptoms but with at least one of the following 1 Biomarkers of myocardial injury raised 2 ECG findings suggestive of cardiac injury 3 Abnormal cardiac function on echocardiogram or cardiac MRI | Absent | Needed | Not known |
| Possible acute myocardits | In the clinical context of possible myocardial injury with cardiovascular symptoms and at least one of the following 1 Biomarkers of myocardial injury raised 2 ECG findings suggestive of cardiac injury 3 Abnormal cardiac function on echocardiogram or cardiac MRI | Absent | Needed | Per clinical syndrome |
| Definite myocarditis | Histological or immunohistological evidence of myocarditis | Needed | Not needed | Tailored to specific cause |
14.4
Imaging
14.4.1
Echocardiography
Echocardiography is an important first-line tool readily available at the bedside, including intensive care units, and is generally the first non-invasive imaging test in suspected myocarditis.
14.4.1.1
Wall motion
Echocardiography can readily assess global and regional wall motion abnormalities. Generally, echocardiographic findings in myocarditis are rather unspecific as described in small patient series and single case reports [ , ]. Echocardiography may also be less accurate than, for example, CMR in detecting subtle contractile impairment as are typical for myocarditis ( Figure 14.2 ).

14.4.1.2
Oedema
Acute inflammatory or ischemic myocardial injury results in myocardial oedema detectable as transient regional wall thickening. Even if wall thickening might be a subtle phenomenon, it can be detected by echocardiography [ , ]. Grossly affected myocardial segments may occasionally appear brighter than remote myocardium. So far, however, there is no reliable link between echo signal and myocardial texture. If the pericardium is involved in the inflammatory process, a small pericardial effusion may be present and detected by echocardiography.
14.4.1.3
Advanced techniques: strain imaging and tissue Doppler imaging
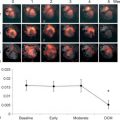
Ejection fraction, as the most frequently reported parameter for systolic function, overemphasizes radial contraction while early impairment of longitudinal contraction is neglected. Global longitudinal strain has proven to be a stable parameter that detects early impairment in contractility when EF is still normal, e.g., in patients after chemotherapy or with aortic stenosis. In acute myocarditis, initial experience suggests that longitudinal strain can detect myocardial impairment even if ejection fraction is close to normal ( Figure 14.3 ) [ ]. In addition, markers for diastolic dysfunction like low mitral annular plane systolic excursion (MAPSE), mitral e ′ or elevated mitral E / e ′ were found in patients with acute myocarditis in the presence of normal left ventricular ejection fraction (LVEF) [ ]. Even if a single abnormal parameter may have limited sensitivity for myocardial damage in acute myocarditis, the combination of various advanced echocardiographic markers may be sufficient to reveal subtle myocardial impairment [ ].

14.4.2
Nuclear imaging
Different nuclear tracers have been applied to diagnose myocarditis, especially gallium-67 and indium-111-labelled antimyosin antibodies, which have been under evaluation for detecting myocarditis in conjunction with single-photon emission computed tomography (SPECT) [ ]. The latter binds specifically to exposed myosin molecules when cellular integrity is lost due to the inflammatory process (see Chapter 10 ). Sensitivity of antimyosin antibody scanning is reported to be very high, however, with lower specificity, as other cell injury (e.g., myocardial infarction) results in uptake of the tracer. Perfusion SPECT using standard tracers (e.g., 99mTc-tetrofosmin, 99mTc-sestamibi, or 201-Thallium) may reveal defects in the more severe cases of myocarditis (e.g., Chagas disease). Whereas most of the approaches no longer have a place in the clinical routine, PET technology is opening new insights and has the potential to add information. For example, PET can particularly contribute to the diagnosis of myocardial sarcoidosis after applying commercially available tracers [ ].
14.4.3
Coronary angiography
The clinical picture of acute ST elevation infarction (STEMI) and acute myocarditis can be similar, and where STEMI is suspected, instant coronary angiography is warranted. Where the clinical picture is strongly suggestive of myocarditis, especially in young males with a history of infection and ECG changes in more than a single coronary territory, non-invasive imaging may be considered as a first line test. If a well-trained CMR unit is in the same hospital and the investigation can be applied immediately, a CMR can be considered. CMR is safe in acute coronary syndrome in experienced hands [ , ] and may provide the diagnosis without requiring invasive angiography [ , , ]. A typical pattern of epicardial and or patchy late enhancement in the absence of infarction scar may be the basis to defer coronary angiography in a young patient without risk factors for CAD. Alternatively, CAD may be excluded by coronary computed tomography (CT) angiography.
14.4.4
Cardiovascular magnetic resonance
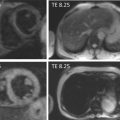
CMR plays an increasing role in the diagnosis and management of myocarditis, mainly due to its unique ability for tissue characterisation and differentiation between ischaemic and non-ischaemic myocardial injury. In addition to the highly accurate assessment of contractile function and morphology, CMR allows assessment of three tissue markers that are relevant in myocarditis: myocardial oedema imaging, hyperaemia and capillary leakage, and myocardial necrosis and fibrosis. Two CMR methods (T2- and early T1-weighted imaging) allow for the detection of oedema. The interpretation is based on a semi-quantitative analysis, and a “cut-off” is given. The use of this cut-off is only reasonable if the detailed technical parameters (sequence-details, coils, field-strengths, contrast-media- characteristics) are respected. Hyperaemia and capillary leakage are assessed with early enhancement (EE) images and irreversible injury can be identified on LGE-images.
14.4.4.1
Diagnostic criteria for myocarditis
Of the available CMR methods, LGE has been the most popular due to high availability, advanced experience due to application in infarction imaging, best comparability between platforms, and highest specificity in myocarditis. The disadvantages of LGE in myocarditis are limited sensitivity and its inability to differentiate between acute and chronic lesions. In 2009, an international expert panel suggested a clinical “routine” protocol for acute myocarditis, the so-called “Lake Louise criteria” [ ]. The Lake Louise criteria recommended the use of a multi-parametric CMR protocol to increase sensitivity and specificity compared to the analysis of a single CMR parameter. The authors propose that the CMR study is called positive if 2 or more of the 3 tissue-based criteria are positive ( Table 14.3 ). Furthermore, the combination of T2-weighted imaging, EE, and LGE may help to assess how acute the myocardial damage is [ ]. Importantly, CMR findings vary depending on the patients studied. The typical young male patient most often presents with focal oedema and LGE in corresponding locations. Fewer patients may present without LGE and still do have true myocarditis. A subgroup of patients presents with global oedema and increased EE.