Fig. 5.1
Spermatic cord lipoma. Grayscale ultrasound demonstrates a well-defined mass with interspersed hyperechoic region lipoma (between calipers) within the spermatic cord
Computed Tomography (CT)
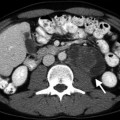
Lipoma can be demonstrated as a low attenuated mass on CT.
Magnetic Resonance Imaging (MRI)
Lipomas have high signal intensity, similar to that of subcutaneous fat, on both T1- and T2-weighted images.
Hemorrhagic lesions may also have high signal intensity with these pulse sequences, so a fat-suppressed sequence must be performed to confirm the diagnosis.
Lipomas do not enhance on contrast enhanced MRI.
Liposarcomas frequently contain soft tissue septa and calcification. On CT and MRI fat can be detected in approximately 80 % of cases.
Pathology
It is unclear whether or not most cord lipomas are truly neoplastic or whether they represent in situ adipose hyperplasia or invaginations of preperitoneal fat into the cord.
Most surgically examined specimens are removed during hernia repairs, but some accompany radical orchiectomies (Fig. 5.2).
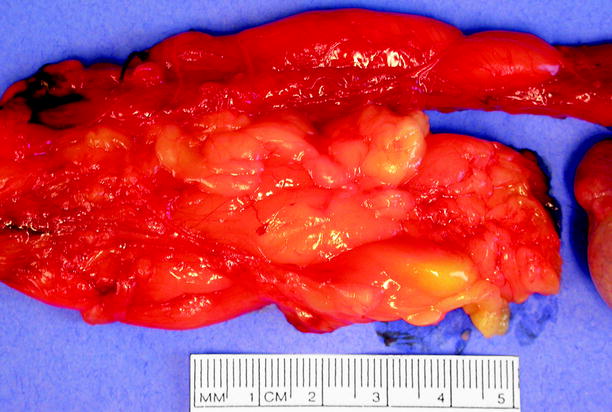
Fig. 5.2
Lipoma. The lipoma, at bottom, has been partially separated from the spermatic cord. In this case, part of the lipoma was indurated due to fat necrosis and was palpable, raising concern for malignancy and prompting radical orchiectomy
They are composed of mature adipose tissue, sometimes complicated by fat necrosis, which can form an indurated palpable nodule indistinguishable clinically from malignancy (Fig. 5.3).
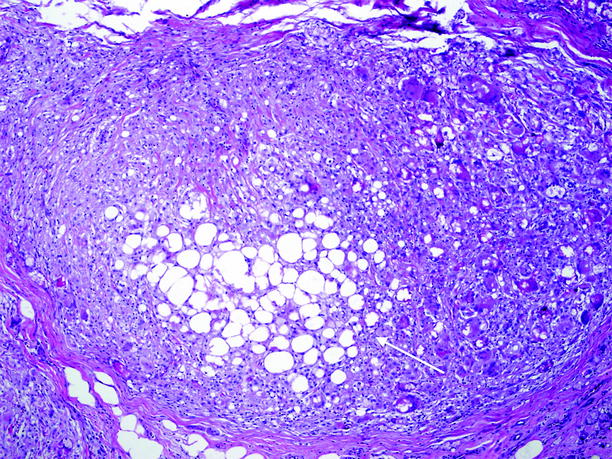
Fig. 5.3
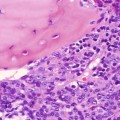
Lipoma, with fat necrosis. Necrotic adipose tissue, in center (arrow), is being engulfed by macrophages and multinucleated giant cells
Rhabdomyosarcoma
General Information
Rhabdomyosarcoma is the commonest primary paratesticular malignant neoplasm in patients between 7 and 36 years of age, with a peak incidence between 2 and 5 years; 60 % occur before the age of 20 years.
Imaging
Ultrasound
At conventional ultrasound, rhabdomyosarcoma has variable echogenicity with a heterogeneous hypoechoic appearance due to hemorrhage and necrosis. Rhabdomyosarcomas vary in appearance from mostly solid to primarily cystic with solid nodules.
Color flow Doppler ultrasound reveals increased blood flow with low resistance in rhabdomyosarcoma.
Magnetic Resonance Imaging
Rhabdomyosarcomas appear as heterogeneous solid extratesticular masses, which generally enhance avidly but heterogeneously after administration of intravenous gadolinium-based contrast material.
Pathology
Rhabdomyosarcoma arises in embryonal mesenchyma in any body site, including tunicae of the testis, epididymis, and spermatic cord. In the groin region, it presents as a painless mass that displaces the testis rather than replacing it. It is typically lobulated, poorly circumscribed, gray-white or tan-pink, and soft (Fig. 5.4).
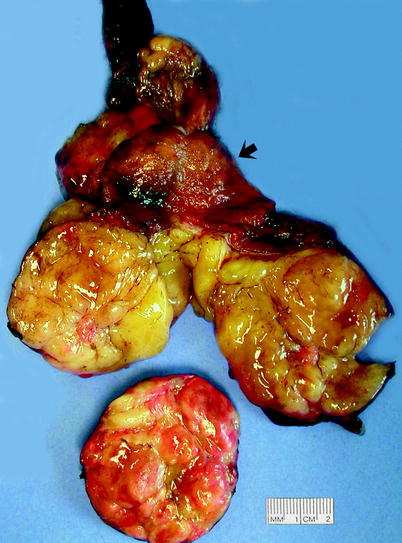
Fig. 5.4
Rhabdomyosarcoma. Embryonal rhabdomyosarcoma excised from a 15-year-old male. Arrow indicates the bisected normal testis
The majority are microscopically of embryonal type, composed of round to oval cells with small dark nuclei and minimal cytoplasm, and admixed with round, oval, or spindled differentiating rhabdomyoblasts with more abundant eosinophilic cytoplasm (Fig. 5.5).
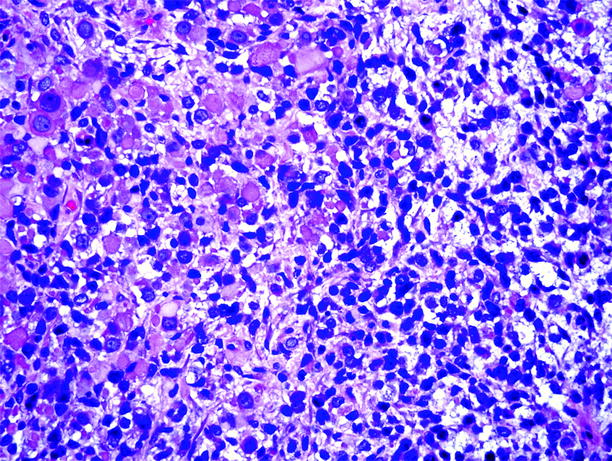
Fig. 5.5
Rhabdomyosarcoma. Embryonal rhabdomyosarcoma, composed of primitive dark round cells, short spindled cells, and large rhabdomyoblasts with copious eosinophilic cytoplasm
Leiomyosarcoma
General Information
Rare neoplasm originates from smooth muscle of spermatic cord.
Usually located in the scrotal part of the spermatic cord opposite to leiomyoma which is frequently located in the inguinal part.
Imaging
Ultrasound
Heterogeneous solid mass with internal vascular flow at color flow Doppler ultrasound
Magnetic Resonance Imaging
Heterogeneous mass which is hypointense on T2-weighted images with contrast enhancement.
MRI findings are similar to other extratesticular sarcomas.
Pathology
Microscopic examination revealed cells arranged in well-defined fascicles with central hyperchromatic nuclei and a moderate amount of eosinophilic cytoplasm.
The high cellularity, pleomorphism, necrosis, and mitotic activity distinguish this tumor from leiomyoma.
Neoplasms of Testicular Adnexae
Introduction
These neoplasms are rarely seen; however, they are clinically significant and affect patients of all ages. Most patients are asymptomatic, presenting with slow-growing nontender mass. Adenomatoid tumors are the most common tumors of the epididymis, followed by leiomyomas. Other benign tumors include fibroma, hemangioma, neurofibroma, and papillary cystadenoma. Malignant tumors are mostly sarcomas and include liposarcoma, rhabdomyosarcoma, lymphoma, fibrosarcoma, metastases, and rarer tumors such as pleomorphic hyalinizing angiectatic tumor, malignant schwannoma, and malignant fibrous histiocytoma. Metastases most frequently arise from prostate, malignant melanoma, renal, and gastrointestinal primary malignancies. Rare mimics of neoplasms include polyorchidism and splenogonadal fusion.
Imaging of Spermatic Cord Neoplasms and Pathologic Features
Adenomatoid Tumor
General Information
Among paratesticular neoplasms, adenomatoid tumor is second in frequency (by far) to lipoma, but accounts for about one-third of non-lipomas in this category. It is a lesion of mesothelial origin and is the commonest benign epididymal neoplasm. It is most often found incidentally in patients aged 30–40 years old, although the reported age range is 18–79 years.
Imaging
Ultrasound
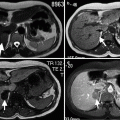
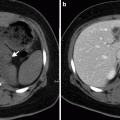
The adenomatoid tumor of the epididymis is usually a hypoechoic, spherical lesion with smooth, well-defined borders (Fig. 5.6a).
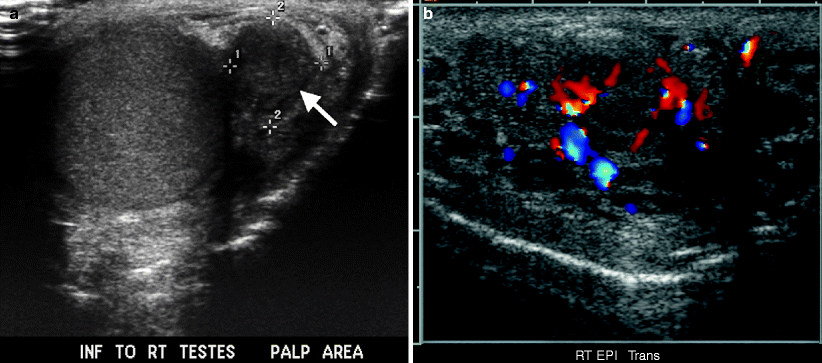
Fig. 5.6
(a) Adenomatoid tumor. Grayscale ultrasound demonstrates a well-circumscribed hypoechoic solid mass (arrow) located in the epididymal tail. (b) Color flow Doppler ultrasound reveals vascular flow in the lesion
These tumors are generally uniformly isoechoic, hypoechoic, or hyperechoic in relation to the adjacent testis.
Color flow Doppler may show vascularity within an adenomatoid tumor (Fig. 5.6b).
Magnetic Resonance Imaging
MR imaging may be helpful to distinguish adenomatoid tumor from intratesticular masses.
Adenomatoid tumor is hypointense mass relative to testicular parenchyma on T2-weighted images and usually does not enhance more intensely than the testis on contrast-enhanced images.
Pathology
The tail of the epididymis is the commonest site but adenomatoid tumor also arises in mesothelial-lined areas of the testis or spermatic cord, distant from the epididymis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree