Key Points
- •
Measurement of the optic nerve sheath diameter (ONSD) by ocular ultrasound provides a rapid, noninvasive quantitative method to assess for elevated intracranial pressure at the bedside, with a measurement of >5 mm considered elevated.
- •
Ocular ultrasound is a valuable tool in the evaluation of ocular trauma and can be used to assess for foreign body, globe rupture, traumatic detachments, lens dislocation, and vitreous hemorrhage.
- •
In patients with painless vision loss, ocular ultrasound can be used to evaluate for posterior vitreous detachment or retinal detachment and for the presence of blood flow in the central retinal artery and vein.
Background
The eye’s superficial location and fluid-filled constitution are ideal for bedside diagnostic ultrasound evaluation. The well-defined borders of the globe and deeper ocular structures, including the optic nerve, retinal artery, and retinal vein, can be easily imaged with ultrasound. Although the use of ocular ultrasound has appeared in ophthalmology literature since the late 1950s, we have only begun to appreciate the diagnostic potential of point-of-care ocular ultrasound over the past decade. Minimal training is required to acquire the skills to perform precise and accurate point-of-care ultrasound examinations of the eye for specific findings.
Bedside ultrasound of the eye can provide valuable diagnostic information, especially when physical examination is limited by bright lighting, facial swelling, or pain due to trauma. Ocular ultrasound allows rapid assessment for potentially vision- threatening conditions when evaluation by an ophthalmologist or with computed tomography (CT) or magnetic resonance imaging (MRI) may be unavailable or delayed. Furthermore, ocular ultrasound enables providers to evaluate the posterior chamber of the eye when direct visualization through the lens is limited by hyphema, hypopyon, or cataracts. Noninvasive assessment for elevated intracranial pressure can also be performed using ocular ultrasound. The five primary indications to perform an ocular ultrasound exam are:
- ▪
Loss of vision (partial or complete)
- ▪
Ocular trauma
- ▪
Atraumatic eye pain
- ▪
Intraocular foreign body (IOFB)
- ▪
Elevated intracranial pressure
Normal Anatomy
The eye normally appears as a circular, well-circumscribed, hypoechoic structure on ultrasound ( Figures 31.1 and 31.2 ). The human eye is 24–25 mm in anteroposterior diameter, with minimal variation from person to person. The cornea appears as a thin, arch-shaped, hyperechoic layer parallel to the overlying eyelid and is contiguous with the sclera. The anterior chamber lies beneath the cornea and is filled with aqueous humor, thus appearing anechoic by ultrasound. The iris and anterior reflection of the lens constitute the posterior wall of the anterior chamber and appear as a hyperechoic line that abuts the pupillary aperture. The lens appears as a biconvex structure with distinct anterior and posterior borders and an anechoic center. Posterior to the lens is a large, anechoic space that is the vitreous body. In younger patients, the vitreous body appears black (anechoic), but in older patients, small, low-intensity echoes are scattered in the vitreous body (“floaters”) due to vitreous syneresis, or liquefaction of vitreous gel. The retina, choroid, and sclera form the posterior border of the globe, and these layers cannot be normally differentiated by ultrasound, except when pathologic findings such as retinal detachment are present. Posterior to the globe, the optic nerve and surrounding echogenic retro-orbital fat can be visualized. The optic nerve extends posteriorly and appears as an echogenic, linear structure perpendicular to the retina. A hyperechoic sheath surrounds the optic nerve. The optic nerve enters the eye slightly inferior and medial to the posterior pole of the globe. With subtle adjustment of the transducer angle, the optic nerve can be visualized longitudinally in the center of the screen. The central retinal artery and vein are within the center of the optic nerve and can be identified using color flow Doppler over the distal optic nerve. The central artery and vein can be distinguished from one another by evaluating the waveforms using pulsed-wave Doppler. In both transverse and sagittal imaging planes, dense orbital bones create acoustic shadows that form the lateral borders of the globe.
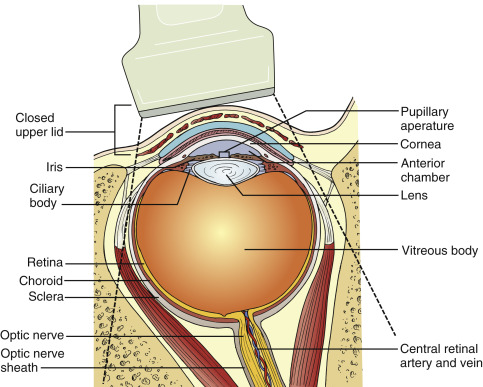

Image Acquisition
Because the eye is superficial and requires high-resolution images to be evaluated, a high-frequency linear array transducer (7.5 MHz or greater) is used. Ophthalmologists often use transducers with higher-than-average frequencies (20–50 MHz) to maximize resolution, but these specialized transducers are not available with most portable ultrasound machines. A two-dimensional (B-mode) ultrasound mode should be selected, and most ultrasound machines include an “ocular” preset. Optimizing near-field gain settings should be performed.
The patient should be positioned lying supine. The head of the bed may be elevated if the patient cannot tolerate lying completely flat. The exam is performed with the patient’s eyelids closed. As with any ultrasound application, an acoustic coupling medium, usually water-soluble ultrasound gel, is used to eliminate the air interface between the transducer and skin. A copious amount of ultrasound gel should cover the entire eyelid to allow adequate imaging, with the transducer placed on the gel without applying any pressure to the eye. Chilling the ultrasound gel results in increased viscosity and allows the gel to stack easily. Ultrasound gel is safe if it comes into contact with the eye, but we recommend use of sterile gel or placing a transparent film dressing, such as a Tegaderm, over the eyelid to prevent contamination of the conjunctiva. Also, a transparent film may improve patient comfort and should be carefully removed without pulling eyebrow hair or eyelashes.
While standing to the patient’s side, begin scanning in a transverse plane by placing the transducer across the eyelid with the transducer marker pointing to the patient’s right ( Figure 31.3 ). The provider’s wrist can be stabilized against the patient’s zygomatic arch or nose bridge to steady the image. Instruct the patient to look straight ahead, and identify the cornea, iris, lens, vitreous body, retina, and optic nerve. The transducer may need to be positioned slightly laterally and angled inferomedially to capture a true longitudinal view of the eye, including the optic nerve. Adjust the depth of the image to visualize 1–2 cm beyond the entrance of the optic nerve into the globe. Tilt or fan the transducer systematically to thoroughly visualize the entire eye throughout the globe. Return to the initial mid-eye image and, while holding the transducer steady, ask the patient to slowly look in all four directions and evaluate the eye for any abnormalities. Objects that disappear as the eye is moved are most likely artifacts, rather than true pathology. After imaging in a transverse plane, turn the transducer 90 degrees to a sagittal plane with the transducer marker pointing toward the patient’s head ( Figure 31.4 ). Obtain a mid-eye view including the cornea, iris, lens, vitreous body, retina, and optic nerve. Repeat the process of tilting or fanning the transducer while having the patient maintain a static gaze. Scan the entire orbit from its medial to lateral edges, noting any abnormalities and correlating findings with those seen on transverse images. After returning to the initial mid-eye view, hold the transducer steady and have the patient slowly look in all four directions. After imaging the entire globe, the ONSD should be measured from a mid-eye transverse view, and the central retinal artery and vein can be evaluated with color flow and pulsed-wave Doppler.
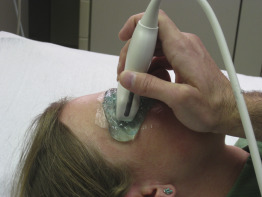
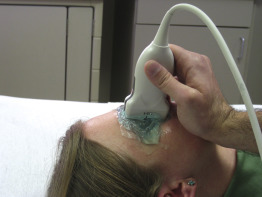
Repeat the same procedure to evaluate the contralateral eye. It may be helpful to scan the unaffected eye first and compare the appearance of the normal eye to any abnormal findings in the affected eye.
Pathologic Findings
Optic Nerve Sheath Diameter
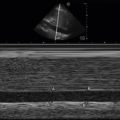
The optic nerve is considered part of the central nervous system, as opposed to the peripheral nervous system, because it is an extension of the brain. The optic nerve sheath encircles the optic nerve and is composed of three layers of meninges. The subarachnoid space surrounding the optic nerve sheath is contiguous with that of the brain and spinal cord, and the same cerebrospinal fluid (CSF) circulates in the subarachnoid space around the brain, spinal cord, and optic nerve. Increases in intracranial pressure (ICP) are transmitted to the CSF in the optic nerve sheath and result in dilation of the optic nerve sheath. The optic nerve sheath diameter (ONSD) can be measured with ocular ultrasound, and a growing body of literature has demonstrated a correlation between increased ONSD and elevated ICP when compared to intraventricular pressure monitoring or evidence of increased ICP on CT scan.
To measure the ONSD accurately, an on-axis, longitudinal cross-sectional view of the optic nerve sheath must be obtained. The borders of the sheath appear sharply demarcated and are parallel to one another. Because the optic nerve enters the globe slightly inferior and medial to the posterior pole, the ideal view of the optic nerve will show an off-axis view of the anterior chamber, iris, and lens. It is imperative to acquire a true on-axis, longitudinal cross section of the optic nerve sheath because off-axis imaging results in erroneous measurement of the ONSD. The ONSD should be measured 3 mm posterior to where the optic nerve sheath engages the retina. The subarachnoid space in the optic nerve sheath does not dilate uniformly, and the greatest variability and most pronounced response to increased fluid in the subarachnoid space occurs 3 mm posterior to the optic nerve sheath–retina junction. Starting in a transverse plane, measure the width of the optic nerve sheath 3 mm posterior to the retina and then rotate the transducer clockwise to measure the ONSD in a sagittal plane, perpendicular to your first measurement ( Figure 31.5 ). Repeat these steps in the contralateral eye to obtain transverse and sagittal measurements of the ONSD. The ONSD is reported as the average of these four values. Any intracranial process causing elevated ICP should affect ONSD of both eyes equally. The presence of unilateral increased ONSD suggests a lateralizing process, such as optic neuritis or compressive optic neuropathy. Papilledema may also be noted as optic disc bulging into the retina and protruding into the vitreous body.
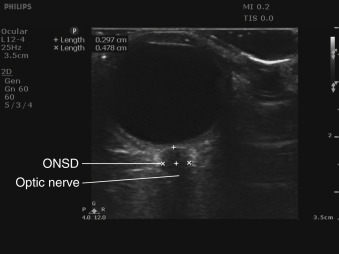
The cutoff value for increased ONSD correlating with increased ICP has been debatable. Based on the initial study of ultrasound measurement of ONSD, many authors cite a diameter >5 mm as elevated in patients older than age 4. Two recent meta-analyses of six studies evaluated the correlation between ONSD and ICP >20 cm H 2 O and calculated a pooled sensitivity and specificity of 87–90% and 79–85%, respectively; however, the cutoff for abnormal ONSD varied from 5.0 to 5.9 mm in these studies, with half of the studies utilizing a cutoff ≥5.7 mm. We recommend using an ONSD >5 mm as abnormal in patients with clinical concern for elevated ICP based on the current body of literature.
Retinal Detachment and Posterior Vitreous Detachment
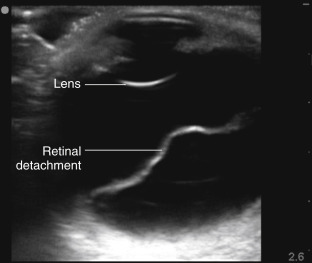
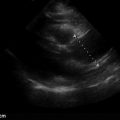
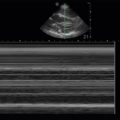
Retinal detachment (RD) and posterior vitreous detachment (PVD) have characteristic findings that can be difficult to detect with bedside ophthalmoscopy, but both of these conditions can be readily diagnosed by ocular ultrasound. Posterior vitreous detachments occur with increased frequency in older patients, with a higher prevalence in myopic patients. PVD is due to separation of the vitreous body from the posterior portion of the retina, and patients complain of seeing “floaters” or brief flashes (photopsia). Retinal detachment is due to separation of the sensory retina from the retinal pigment epithelium. There are several causes of retinal detachments, but acute posterior vitreous detachment with retinal tear is the most common cause. Retinal detachments may be preceded clinically by increased floaters or flashers followed by perception of a curtain coming down and visual field loss. If RD involves the macula, there may be a loss of central vision and impaired acuity. Retinal detachments appear by ultrasound as thick, hyperechoic membrane-like structures with multiple folds that appear to be lifted off the posterior surface of the globe and move in conjunction with ocular movements ( Figure 31.6 ). Vitreous detachments appear similarly as linear structures lifted off the posterior surface of the globe ( Figure 31.7 ), but they are thinner and smoother than retinal detachments and have a more mobile, undulating appearance. The retina is firmly tethered to the choroid posteriorly at the optic nerve and anterolaterally at the ora serrata in the ciliary body. Therefore, retinal detachments should not cross over the optic nerve or extend to the ciliary body, but a posterior vitreous detachment may do so. The macula can be seen just lateral (temporal) to the optic nerve on transverse views, and it may be possible to distinguish macular involvement by ultrasound. A choroid detachment, where the choroid separates from the underlying sclera, may also appear similar to a PVD or RD, but a choroid detachment can be distinguished by its smooth, bulging convex shape that does not move with eye movements and possible extension anteriorly to include the ciliary body.