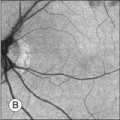
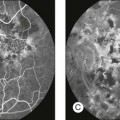
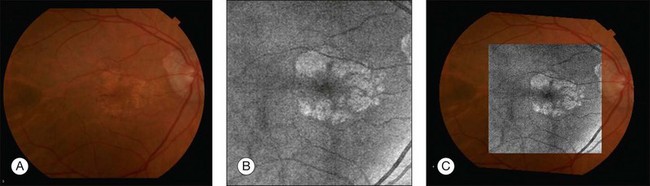
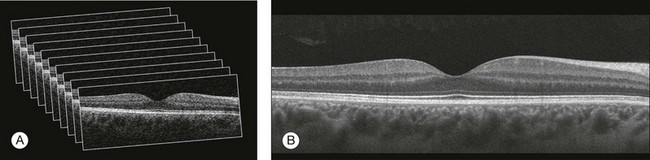
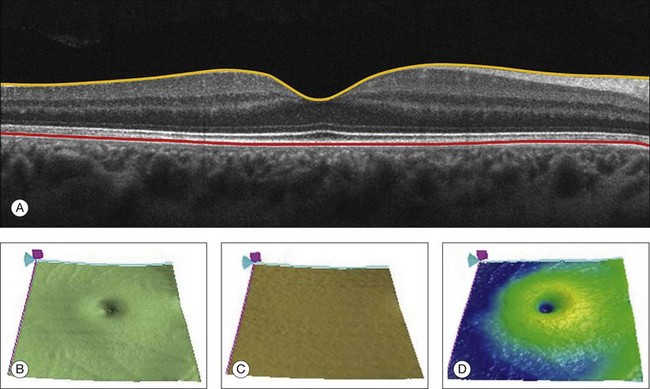
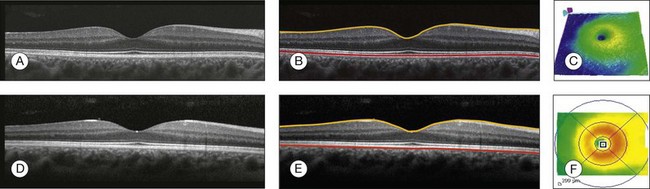
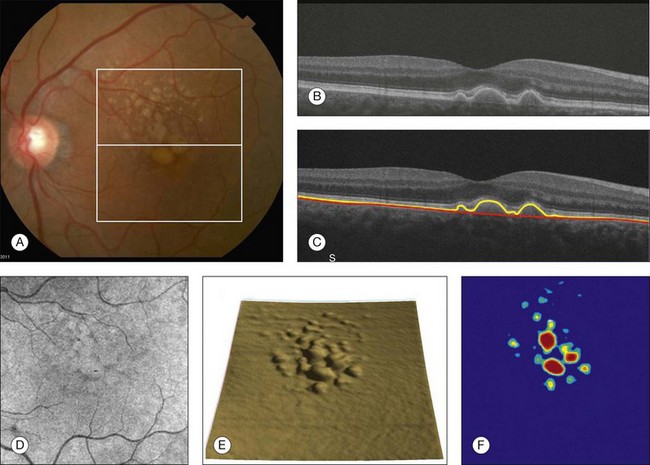
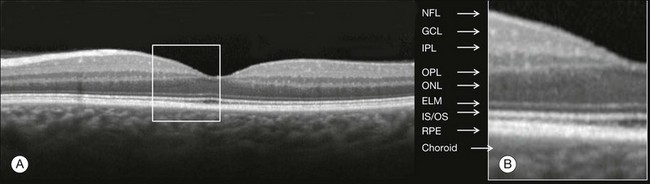
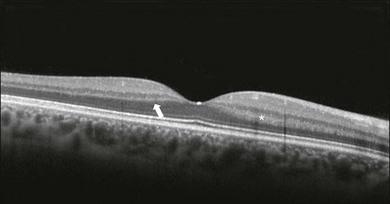
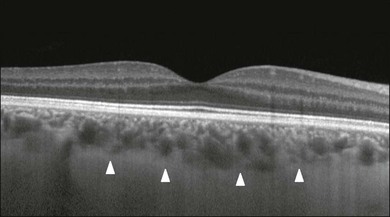
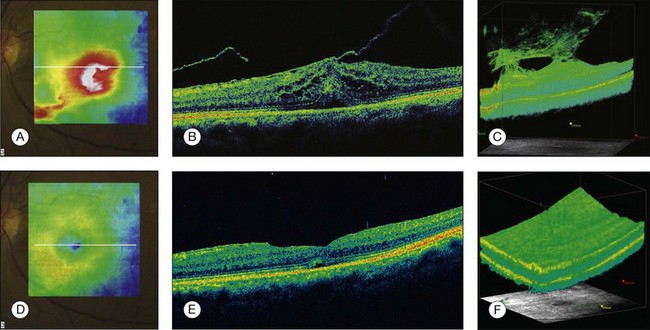
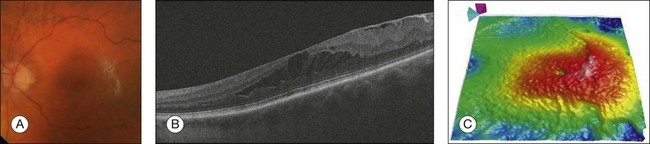
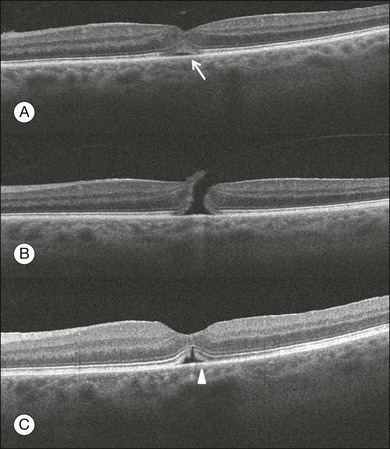
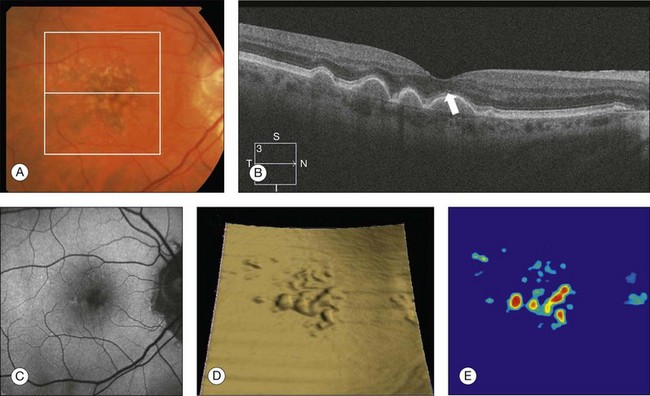
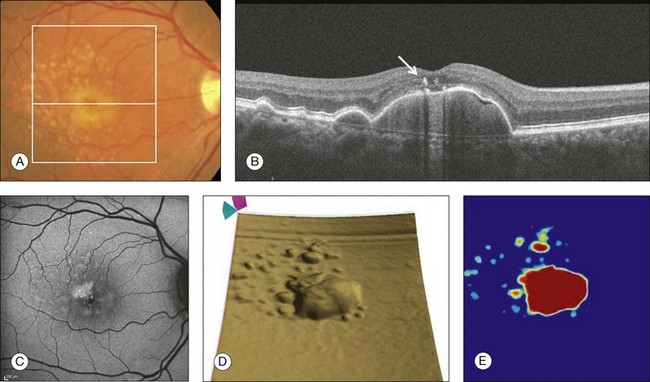
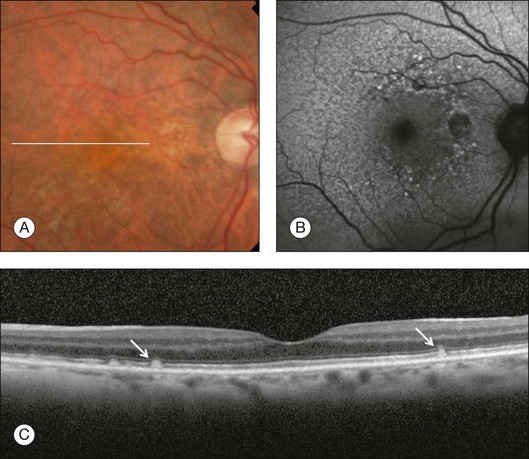
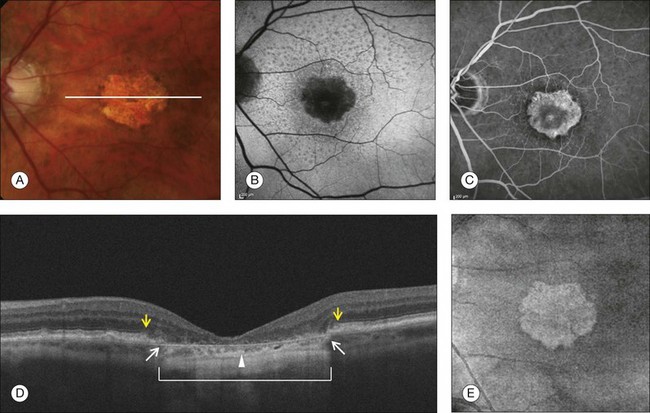
Chapter 3 Carlos Alexandre de Amorim Garcia Filho, Zohar Yehoshua, Giovanni Gregori, Carmen A. Puliafito and Philip J. Rosenfeld During the past two decades, optical coherence tomography (OCT) has become an essential tool in ophthalmology. Its ability to image detailed ocular structures noninvasively in vivo with high resolution has revolutionized patient care.1,2 OCT technology is based on the principle of low-coherence interferometry, where a low-coherence (high-bandwidth) light beam is directed on to the target tissue and the scattered back-reflected light is combined with a second beam (reference beam), which was split off from the original light beam. The resulting interference patterns are used to reconstruct an axial A-scan, which represents the scattering properties of the tissue along the beam path. Moving the beam of light along the tissue in a line results in a compilation of A-scans with each A-scan having a different incidence point. From all these A-scans, a two-dimensional cross-sectional image of the target tissue can be reconstructed and this is known as a B-scan. Typically OCT instruments use an infrared light source centered at a wavelength of about 840 nm. For a given wavelength, the axial resolution is dictated by the bandwidth of the light source. The latest commercial instruments typically have an axial resolution of approximately 5 µm, while research instruments have been built with a resolution as high as approximately 2 µm.1 The lateral resolution is limited by the diffraction caused by the pupil and it is normally about 20 µm. For clinical purposes, the image acquisition time is limited by the patient’s ability to avoid eye movements, i.e., less than 2 seconds in the typical patient. The instrument’s scanning speed (number of A-scans acquired per second) is then the crucial parameter determining the amount of data available for a single OCT dataset. The early OCT instruments, known as time domain OCT (TD-OCT), used a single photo detector, and an A-scan was created by moving a mirror to change the optical path of the reference beam in order to match different axial depths in the target tissue. This setup limited the scanning speed to a few thousand A-scans per second. A newer technique, known as spectral domain OCT (SD-OCT), Fourier domain OCT (FD-OCT), or high-definition OCT (HD-OCT), is able to acquire an entire A-scan by using an array of detectors instead of using multiple reference beams from a moving mirror. Scanning speeds with SD-OCT instruments can exceed 100 000 A-scans per second, about 200 times faster than TD-OCT. Currently available SD-OCT commercial systems operate at a scanning rate of approximately 27 000 A-scans per second.1 The scanning pattern with the commercial TD-OCT instrument (Stratus OCT, Carl Zeiss Meditec, Dublin, CA) incorporated six radial, concentric, 6-mm-long B-scans centered on the fovea. With the recent development of high-speed SD-OCT systems, several novel and important imaging strategies have been introduced based on acquiring three-dimensional datasets and B-scan averaging (Fig. 3.1). Three-dimensional datasets are obtained using a dense two-dimensional raster array over a relatively large retinal region. The resulting datasets can be rendered as a volume image in three dimensions and can be analyzed by showing two-dimensional slices (i.e., sequences of parallel B-scans). Three-dimensional datasets give detailed information about the retinal structure over large areas. In addition, it is possible to generate en face fundus-like images directly from the OCT datasets. These OCT fundus images (OFIs) provide an accurate spatial colocalization of retinal features observed on the en face and cross-sectional images. Therefore, exact correlations can be achieved between the retinal cross-sectional geometry seen on the OCT B-scans and the retinal landmarks seen on en face images, known as the OFI. The potential exists for registration between several SD-OCT datasets of the same eye and images obtained using other imaging modalities, such as color fundus photography, fluorescein angiography, and fundus autofluorescence imaging. This holds the promise for an unprecedented ability to describe and monitor changes in the local geometry of the retina.3 In addition to the OFI generated by a full OCT dataset, partial OFIs (or slabs) can be generated to produce en face renderings that correspond to particular retinal layers or features.4 These slabs can be very useful to visualize and quantify specific pathologies (Fig. 3.2). Fig. 3.2 (A) Color fundus image of a patient with geographic atrophy secondary to age-related macular degeneration. (B) Optical coherence tomography (OCT) fundus image, which is the en face image from the reflected light from each A-scan, of the same patient obtained with a scan pattern of 200 × 200 A-scans in the Cirrus high-definition OCT instrument. (C) Registration of the color fundus image with the OCT fundus image. Since the area of the OCT fundus image is known to be 6 × 6 mm, it is possible to quantify lesion area and calibrate the fundus camera to use this technique. The scanning speed of SD-OCT can also be used to produce very high-quality individual B-scan images through a combination of high sampling density and image averaging. One of the main factors affecting the perceived quality of OCT images is noise, in particular the speckle noise which is responsible for the characteristic “granular” appearance of OCT. Noise can be reduced through the acquisition, registration, and averaging of a number of B-scans at approximately the same retinal position (Fig. 3.3). Fig. 3.3 Averaging process. (A) Multiple B-scans acquired through the foveal center of a normal patient. (B) The registration and averaging of these B-scans can reduce the speckled noise and improve the image quality. In these examples the image was averaged 20 times using the Cirrus high-definition optical coherence tomograph. Recently, different companies have invested in research in the field of retinal imaging, especially in the development and improvement of SD-OCT. It is not an objective of this section to discuss the differences between each of the currently available instruments since these instruments are continuously evolving. Table 3.1 lists currently available instruments. Table 3.1 Commercially available spectral domain optical coherence tomography (OCT) instruments Several commercially available SD-OCT instruments offer some level of quantitative analysis using different, proprietary segmentation algorithms. The various segmentation algorithms make different design choices and have been shown to have very different performance profiles in terms of accuracy, reproducibility, and robustness.5–8 Care should be exercised when comparing measurements obtained from different OCT instruments. The most commonly used quantitative parameter derived from OCT datasets is retinal thickness, obtained by segmenting the internal limiting membrane (ILM) and a boundary representing the retinal pigment epithelium (RPE). This information can be used to generate surface maps of the ILM and the RPE as well as two-dimensional and three-dimensional retinal thickness maps. These maps can be very useful in identifying and describing deviations from the normal anatomy and changes over time. Registering OCT datasets acquired over time can give very precise information about the dynamics of disease progression and response to treatment based on changes in retinal anatomy (Fig. 3.4). Fig. 3.4 Segmentation process. (A) B-scan through the foveal center of a normal patient with a yellow line identifying the internal limiting membrane (ILM) and a red line corresponding to the retinal pigment epithelium (RPE). Three-dimensional map of the ILM (B), RPE (C), and the retinal thickness map (D) acquired with a 200 × 200 scan pattern with the Cirrus high-definition optical coherence tomography instrument. It is important to keep in mind that there is some confusion in the definition of the outer retinal boundary. In a normal eye, the bright reflective band at the external aspect of the retina, often referred to as the RPE complex, can be resolved in ultrahigh-resolution images, and occasionally in images acquired with a commercially available SD-OCT instrument, consisting of three individual layers.9 Different segmentation algorithms from different instruments tend to follow different edges and therefore result in different measurements. For example, the Spectralis SD-OCT instrument typically follows the posterior edge of the RPE complex, the Stratus TD-OCT instrument typically follows the inner segment–outer segment (IS/OS) junction, which is anterior to the RPE complex, and the Cirrus SD-OCT instrument typically follows the anterior edge of the RPE layer (Fig. 3.5). This situation becomes even more complicated and sometimes inconsistent when the normal retinal structure is deformed by the presence of pathology.10 Fig. 3.5 Differences in segmentation and retinal thickness maps between instruments. The B-scan, the B-scan identifying the internal limiting membrane (yellow line), and the retinal pigment epithelium (red line), and the retinal thickness map acquired with the Cirrus high-definition optical coherence tomograph (A–C) and the Spectralis (D–F). Note that, using the Cirrus instrument, the segmentation algorithm identifies the actual retinal pigment epithelium (B) and using the Spectralis the segmentation algorithm identifies Bruch’s membrane. This subtle difference in the segmentation algorithm between each instrument can be responsible for different retinal thickness measurements. In addition to total retinal thickness, a number of other quantitative parameters have been proposed. For example, it is possible to obtain measurements of particular retinal layers, such as the thickness of the ganglion cell layer or the thickness of the photoreceptors’ outer segments, as well as measurements of retinal lesions, like the area of geographic atrophy (GA).9,11,12 An area of particular promise is the measurement of RPE deformations associated with drusen.13,14 These measurements are obtained by comparing the actual RPE geometry with the geometry of a virtual RPE free of deformations. Parameters like drusen area and volume can be generated in a fully automated manner and have been shown to be quite robust and reproducible (Fig. 3.6). Fig. 3.6 Retinal pigment epithelium (RPE) deformation algorithm. (A) Color fundus image of a patient with drusen. A 6 × 6 mm white box was superimposed on the image to represent the scan area. (B) B-scan from the spectral domain optical coherence tomography dataset that corresponds to the central line on the color fundus image. (C) B-scan with a yellow line representing the RPE segmentation and a red line showing the RPE floor (virtual map of the RPE free of deformations). (D) En face image of the 6 × 6 mm scan pattern (optical coherence tomography fundus image). (E) Three-dimensional RPE map delineating the drusen conformation. (F) RPE elevation map with drusen area (1.41 mm2) and volume (0.08 mm3). The amount of information provided by each dataset, together with the possibility for image registration and longitudinal studies, makes SD-OCT a very promising new tool for the quantitative study of retinal pathologies. These capabilities solve the biggest problems associated with the Stratus TD-OCT retinal thickness maps: the lack of precise correspondence between the B-scans and the retinal topography, the difficulties associated with eye movements, and the need for significant interpolation of the data due to undersampling of the retina. Despite the advantages of SD-OCT, segmentation algorithms can produce artifacts, particularly in the presence of macular disorders with complex morphology like neovascular age-related macular degeneration (AMD).15–19 Therefore, it is important to be vigilant and monitor the quality of the segmentation in order to eliminate artifacts arising from flawed segmentation and associated measurements. The OCT image closely approximates the histological appearance of the macula and, for this reason, it has been referred to as an in vivo optical biopsy. With the increase in the axial resolution of the new SD-OCT instruments (5–8 µm) and the ultrahigh-resolution OCT (2 µm), it has become possible to correlate OCT images accurately with histological features of the retina.20 However, care must be taken when making assumptions about these correlations because histological sections require fixation and exogenous staining to produce contrast within tissue, and this can introduce artifacts, while OCT relies on intrinsic differences in tissue optical properties to produce image contrast.21 When light travels through the retinal tissue it can be reflected, scattered, or absorbed, and this creates the multilayered pattern of the retina. The angle of incidence of the light, motion artifacts, speckled noise, and image contrast can affect the axial resolution of retinal imaging. Therefore, one-to-one correspondence of histology with OCT images cannot be expected.2,21 Although the interpretation of features of the inner retina, which can be defined for our purpose to span from the ILM to the junction of the inner and outer segments of the photoreceptors, appears to correlate well with histology, the OCT features of the outer retina are less well understood and remain a topic of discussion (Fig. 3.7).9,22,23 Fig. 3.7 Spectral domain optical coherence tomography (Spectralis, Heidelberg) image of a normal individual. The multilayered retinal architecture can be observed and each retinal layer can be identified. NFL, nerve fiber layer; GCL, ganglion cell layer; IPL, inner plexiform layer; OPL, outer plexiform layer; ONL, outer nuclear layer; ELM, external limiting membrane; IS/OS, junction of the inner and outer segments of the photoreceptors; RPE, retinal pigment epithelium. The first detected layer in most OCT scans is the ILM that appears as a hyperreflective layer at the vitreoretinal interface. In some patients, the posterior hyaloid can be seen above the ILM as a hyperreflective layer. Within the retina, the retinal nerve fiber layer and the plexiform layers (both inner and outer) are seen as hyperreflective while the ganglion cell layer and the nuclear layers (both inner and outer) are hyporeflective. A recent study demonstrated that the incidence of the light beam could affect the appearance of Henle’s fiber layer by OCT, resulting in a thin hyperreflective layer corresponding to the photoreceptor synapses or a thicker hyperreflective layer corresponding to photoreceptor axonal extensions enveloped by the outer cytoplasm of Müller cells (Fig. 3.8).24 The retinal vessels may sometimes be seen on OCT images as circular hyperreflective structures located in the inner retina, with a vertical shadow or reduced reflectivity extending into deeper layers. Fig. 3.8 Spectral domain optical coherence tomography of the same patient using a different light incidence. This results in a thin hyperreflective layer that corresponds to the photoreceptor synapses (white arrow) or a thicker hyperreflective layer corresponding to photoreceptor axonal extensions enveloped by the outer cytoplasm of Müller cells (white asterisk). Outside the central fovea, commercially available SD-OCT instruments typically resolve four bands in the outer retina. There is discordance between different authors regarding which anatomical structure correlates with each band.20,21,25 The innermost band has been attributed to the external limiting membrane (ELM). This band is typically thinner and fainter than the others. The nomenclature for the middle two bands has much less supportive evidence. The second of the four bands has been commonly ascribed to the boundary between the IS/OS of the photoreceptors and the third band is referred to as either the OS tips or as Verhoeff’s membrane.9,26 A recent study suggested that the second band was the ellipsoid section of the photoreceptors (inner segment) instead of the IS/OS junction and that the third band appears to correspond to the contact cylinder between the RPE apical process and the external portion of the cone outer segment. This band typically merges with the fourth band in the central fovea and this is explained by a greater height of the contact cylinder of the cones and RPE outside the fovea.25 The fourth hyperreflective outer retinal band is attributed to the RPE, with potential contribution from Bruch’s membrane and choriocapillaris, with abundant experimental and clinical evidence supporting this designation.9,22,27 Although the current SD-OCT uses a short wavelength of approximately 840 nm, which results in light scattering at the level of the RPE and a lower signal from the deep choroidal tissue, it is also possible to image the choroid and extract quantitative information (Fig. 3.9).28–31 Choroidal thickness may be influenced by age, axial length, and perhaps refractive abnormalities. It also varies in different retinal regions within the same normal subject, being thickest beneath the fovea,31 or in the superior outer macula (Early Treatment Diabetic Retinopathy Study (ETDRS) subfield), with the thinnest choroid being located in the nasal outer ETDRS subfield.32 When centering the optic nerve head as a reference point, the choroid appears thin in the peripapillary region and increases in thickness with eccentricity in all directions, up to a certain point, except inferiorly.32 This is the embryonic location of the optic fissure closure and thus may be responsible for the localized thinning.32,33 High-penetration OCT uses a light source with a wavelength around 1050 nm that allows a better visualization of the posterior choroid and sclera than currently available SD-OCT instruments.30,33,34 Fig. 3.9 Enhanced-depth spectral domain optical coherence tomography image (Spectralis, Heidelberg) of a normal subject showing the boundaries of the choroid (arrowheads). The high axial resolution and the different scan patterns offered by SD-OCT provide comprehensive structural information that can be used to map retinal layer thicknesses and perform volumetric analyses. Using different SD-OCT instruments, several authors have reported an approximate central retinal thickness of 265 µm in normal subjects.35 However, caution is required, as errors in automated measurements may occur and are more often found in macular disorders with complex morphology like neovascular AMD, which alters the ability of segmentation algorithms to detect normal boundaries.15–19 Therefore, care must be taken that high-quality and artifact-free scans are obtained before running the retinal thickness algorithm. Vitreoretinal interface disorders VMT syndrome results from persistent vitreoretinal adhesions in the setting of a partial posterior vitreous detachment.36 In normal eyes, as the vitreous liquefies due to age, it detaches from the macula. This natural progression has been demonstrated using OCT.37 In some people, an unusually strong adhesion is present between the vitreous and macula, and as the vitreous detaches peripherally, it continues to pull on areas of the macula. The vitreoretinal adhesions transmit tractional forces to the retina from the vitreous body, having the potential to cause tensile deformation, foveal cavitations, cystoid macular edema (CME), limited macular detachment, or a macular hole.38,39 Patients can present with visual loss and metamorphopsia. Diagnosis of VMT by biomicroscopy may be challenging, particularly when the area of vitreoretinal attachment is broad. OCT better defines the vitreoretinal relationships in eyes with VMT and also documents concomitant ERM and macular edema.40–44 With OCT imaging, the abnormal VMT bands from the prominent posterior hyaloid are well delineated as reflective lines from the perifoveal area into the vitreous cavity, distorting the macular contour with or without accumulation of intraretinal or subretinal fluid (Fig. 3.10). Fig. 3.10 Vitreomacular traction syndrome: color fundus image of the left eye of a 71-year-old woman superimposed with the retinal thickness map (A) showing an increase in the retinal thickness (red areas). The B-scan of the macular region shows an increase in the retinal thickness and the presence of subretinal fluid and intraretinal cysts due to vitreomacular traction and an epiretinal membrane (B). A three-dimensional spectral domain optical coherence tomography (courtesy of Cirrus, Carl Zeiss Meditec) is presented (C). The patient underwent surgery and, 2 months after pars plana vitrectomy, the retinal thickness decreased, with resolution of the intraretinal cysts (D–F). In recent years OCT has been most beneficial in diagnosing VMT and subsequently directing treatment of this condition. In some cases, spontaneous resolution can occur with separation of the vitreous from the macula, leading to subsequent resolution of the intraretinal and subretinal fluid and restoration of normal vision.45,46 However, in most eyes, VMT persists and vitrectomy may be an effective treatment option for patients with symptomatic VMT.40,47,48 Consequently, OCT is useful in monitoring subtle changes in vitreoretinal adhesions and retinal architecture and assisting the treatment decision-making process. ERM occurs in approximately 6% of patients over the age of 60, with incidence increasing with age.49,50 ERMs can be classified as idiopathic or secondary to an initiating event. Most idiopathic ERMs are thought to result from fibroglial proliferation on the inner surface of the retina secondary to a break in ILM occurring during posterior vitreous detachment.51,52 Secondary ERMs result from an already-existing ocular pathology such as central or branch retinal vein occlusion, diabetic retinopathy, uveitis, and retinal breaks with or without detachment.53 Glial cells, RPE cells, and myofibroblasts are shown to be mostly involved in ERM formation.51,52 ERM may lead to loss of normal retinal anatomy, with the patient experiencing metamorphopsia, micropsia, monocular diplopia, and decreased visual acuity. These symptoms vary in severity depending on the location, density, and contraction of the membrane. On slit-lamp biomicroscopy, a mild ERM appears as a glistening layer on the retinal surface. Denser membranes may be seen as a gray sheet overlying the retina and causing distortion in the macular vascular architecture. Occasionally, ERMs can evolve into macular pseudoholes and ERMs are often seen in conjunction with idiopathic full-thickness macular holes.38 Fluorescein angiography may demonstrate macular leakage, which can be variable from case to case. OCT provides qualitative and quantitative information about the retinal anatomy, which can identify factors contributing to vision loss in patients with ERM. On OCT, ERMs are seen as a highly reflective layer on the inner retinal surface (Fig. 3.11). In most eyes, the membrane is globally adherent to the retina but, in some cases, it can be separated from the inner aspect of the retina, which enhances its visibility by OCT. In this situation, it is usually distinguishable from a detached posterior hyaloid. Secondary effects of the membrane include loss of the normal foveal contour, increased retinal thickness, and the presence of cystoid changes, and these features may be observed in more advanced membranes. OCT is useful for monitoring changes in cases that are being observed and for documenting the response to treatment in patients undergoing pars plana vitrectomy with membrane peeling. Fig. 3.11 Epiretinal membrane – color fundus image of the left eye of a 65-year-old man with grayish tissue over the retina (A). Cross-sectional optical coherence tomography image showing a hyperreflective tissue overlying the retina, resulting in increased retinal thickness and cysts in the retina (B and C). Idiopathic macular holes typically occur in the sixth to seventh decade of life with a 2 : 1 female preponderance. Symptoms include decreased visual acuity, metamorphopsia, and central scotoma. Bilateral involvement occurs in 15–20% of patients.21 A full-thickness defect in the neural retina as seen with OCT can differentiate a true macular hole from a pseudohole seen clinically. Pseudoholes are seen in the presence of a dense sheet of ERM with a central defect that overlies the foveal center, giving the ophthalmoscopic appearance of a true macular hole.21,54 Gass described the stages of macular hole formation based on biomicroscopic findings.55 A stage 1 impending hole is characterized by a foveal detachment seen as a yellow spot (1A) or ring (1B) in the fovea (Fig. 3.12A). Spontaneous resolution will occur in approximately 50% of these cases. In stages 2–4, there is a full-thickness retinal defect, with a complete absence of neural retinal tissue overlying the foveal center. What differentiates these stages is the size of the retinal defect (<400 µm in stage 2 and >400 µm in stage 3) or the presence of a complete posterior vitreous detachment regardless of the hole size (stage 4) (Fig. 3.12B). Fig. 3.12 Macular hole. (A) Stage 1 macular hole in a 63-year-old woman with a 3-month history of decreased visual acuity (20/60). An outer retinal defect can be observed in the B-scan (arrow). (B) A full-thickness retinal defect developed after 2 months of follow-up with worsening in the visual acuity (20/80). The posterior vitreous remains adhered to the edge of the macular hole. (C) One month after surgery, the macular hole was closed and the visual acuity improved to 20/50, but a persistent foveal outer defect could be observed (arrowhead). OCT has enhanced our understanding of the pathogenesis of macular holes, the healing process after surgical repair, and helped in identifying pre- and postoperative features that are related to visual outcome. The anatomic changes identified on OCT have been correlated with the various stages of macular hole. In stage 1A, patients usually present with a localized foveolar detachment, which can resolve spontaneously after the posterior vitreous detachment with resolution of the yellow foveal spot, or it can progress to stage 1B with a development of a pseudocyst with loss of the outer retinal layers, and later develop into a full-thickness macular hole.56,57 Generally, the retinal defect is accompanied by a variable amount of intraretinal fluid appearing as cysts and a variable amount of subretinal fluid at the edge of the hole. The edge of the hole can appear elevated, as a result of the significant intraretinal fluid accumulation or due to persistent vitreofoveal traction. In a stage 4 macular hole, OCT can demonstrate the complete hyaloid separation and occasionally a retinal operculum can be seen floating above the foveal center. Vitrectomy has become the standard treatment for macular hole with anatomical success rates of 85–100%.58,59 OCT can be used to confirm complete macular hole closure and restoration of the normal foveal contour.60–63 In cases with suboptimal postoperative visual outcomes, OCT can visualize persistent retinal abnormalities despite anatomically successful macular hole surgery (Fig. 3.12C). Restoration of the ELM and the so-called junction of the inner and outer segment of photoreceptors may reflect the morphologic and functional recovery of the photoreceptors in surgically closed macular holes.62–65 A residual small defect in the ELM is often still evident in closed holes, particularly in those that are spontaneously healed. The ability to perform OCT imaging in eyes filled with gas or silicone oil has also been useful as an adjunct to determine the extension of the face-down position in patients following vitrectomy for macular hole.66–68 AMD is a common cause of irreversible vision loss among the elderly worldwide. It is estimated that approximately 30% of adults older than 75 years have some sign of AMD and that approximately 10% of these patients have advanced stages of the disease.69–72 AMD can be classified in two forms: non-neovascular (dry) and neovascular (wet or exudative). The non-neovascular form accounts for 80–90% of cases while the neovascular form accounts for 10–20% of cases, but was responsible for the majority of severe vision loss (80–90%) prior to the widespread use of vascular endothelial growth factor (VEGF) inhibitors.71,73 Non-neovascular (dry) AMD is characterized by abnormalities of the RPE, Bruch’s membrane, and choriocapillaris. These abnormalities may be asymptomatic or accompanied by compromised vision, and are considered to be the precursors of GA and choroidal neovascularization (CNV).74,75 Early non-neovascular AMD: drusen and pigmentary changes: Drusen appear clinically as focal white–yellow excrescences deep to the retina. They vary in number, size, shape, and distribution. Several grading strategies have been developed to image drusen using color fundus imaging.76,77 Although color fundus imaging is useful for assessing the appearance of drusen, these images only provide two-dimensional area information on the geometry of the drusen and cannot be used to measure quantitative properties such as drusen volume. Until the advent of high-speed spectral domain technology, evaluation of drusen with OCT was often difficult as motion artifacts commonly resulted in apparent undulation of the RPE, mimicking the appearance of drusen.78,79 SD-OCT can provide a three-dimensional, geometric assessment of drusen. The high-definition B-scans obtained with SD-OCT are useful to assess the ultrastructure of drusen and to evaluate for evidence of disruption of adjacent retinal layers. Drusen are seen as discrete areas of RPE elevation with variable reflectivity, which is consistent with the variable composition of the underlying material (Fig. 3.13).80,81 In larger drusen or drusenoid retinal pigment epithelial detachments (PEDs), the RPE has a greater elevation with a dome-shaped configuration.82 Larger drusen may often become confluent and can sometimes be accompanied by fluid accumulation under the retina in the absence of CNV (Fig. 3.14).80 Recognition of this feature may avoid unnecessary treatment with anti-VEGF drugs. SD-OCT imaging has the resolution to evaluate the retinal layers overlying drusen. A thinning in the photoreceptor layer can be observed in up to 97% of cases, with average photoreceptor layer thickness reduced by 27% compared to age-matched control eyes. The inner retinal layers usually remain unchanged. These findings demonstrate a degenerative process with photoreceptor loss leading to visual impairment.83 Fig. 3.13 Early non-neovascular age-related macular degeneration. (A) Color fundus image of the right eye of a 61-year-old man with drusen and pigmentary changes in the macula. (B) Foveal B-scan showing the drusen as elevations of the retinal pigment epithelium (RPE). The inner and outer segment junction of the photoreceptors adjacent to the drusen appears disrupted (arrow). (C) Fundus autofluorescence illustrating that drusen cannot be reliably identified by this imaging modality. (D) RPE segmentation map showing drusen in a unique three-dimensional perspective. (E) RPE elevation map providing the drusen area (1.37 mm2) and volume (0.063 mm3). Fig. 3.14 Drusenoid retinal pigment epithelium detachment (DPED). (A) Color fundus image of the right eye of a 66-year-old man with a DPED and pigmentary changes in the macula. (B) Foveal B-scan showing the confluent drusenoid material as a large elevation of the retinal pigment epithelium (RPE). Intraretinal pigment migration can be observed (arrow). (C) Fundus autofluorescence image (Heidelberg retina angiograph, Heidelberg). (D) RPE segmentation map showing the DPED in a three-dimensional perspective. (E) RPE elevation map providing the DPED area (3.87 mm2) and volume (0.508 mm3). The acquisition of dense raster scans comprised of a large number of lower-density B-scans combined with the use of segmentation algorithms results in the ability to generate maps of the RPE, which provides information on RPE geometry and therefore a unique perspective of drusen. A novel algorithm developed to identify RPE deformations such as drusen has been shown to be highly reproducible in the measurement of drusen area and volume.13 The algorithm creates a drusen map from a scan pattern of 40 000 uniformly spaced A-scans organized as 200 A-scans in each B-scan and 200 horizontal B-scans, covering an area of 6 × 6 mm centered in the fovea. The algorithm uses the actual RPE geometry and compares this RPE map to a virtual map of the RPE free of any deformations (RPE floor). The algorithm creates a difference map from these two maps, which permits reproducible measurements of drusen area and volume (Fig. 3.6). This algorithm was used to study the natural history of drusen in AMD.14 Drusen were shown to undergo three different growth patterns. In most eyes, drusen were found to increase in volume and area. Drusen could also remain stable or they could dramatically decrease over time. When these drusen decreased, they could evolve into GA or neovascular AMD, or they could decrease, resulting in no apparent residual anatomic defect in the macula. The RPE cells are capable of hypertrophy and proliferation in response to different stimuli and in many cases an intraretinal pigment migration may occur (Fig. 3.14B). The Age-Related Eye Disease Study research group reported a severity scale defining large drusen (≥125 µm) and pigment abnormality in the macula as being a risk factor for disease progression in patients with intermediate AMD.84,85 This pigmentary abnormality can be observed on OCT imaging as small discrete hyperreflective lesions within the neurosensory retina, usually within the outer nuclear layer.86 Typical drusen in AMD are seen as deposits between the RPE and the inner collagenous layer of Bruch’s membrane. OCT imaging is also useful for the assessment of a variety of conditions characterized by variant forms of drusen. These deposits can also be seen on top of the RPE and are known as “subretinal drusenoid deposits.”87,88 They appear on OCT imaging as granular hyperreflective material between the RPE and the IS/OS junction and are also well visualized on blue-light reflectance imaging and autofluorescence imaging (Fig. 3.15). Fig. 3.15 Subretinal drusenoid deposits. (A) Color fundus image of a 76-year-old woman shows multiple yellowish, small, and round lesions. (B) Fundus autofluorescence clearly shows these deposits as small and multiple hyperautofluorescent spots. (C) On optical coherence tomography, these lesions appear as multiple areas of granular hyperreflectivity between the retinal pigment epithelium and the inner segment–outer segment junction (arrows). Another form of drusen, known as “cuticular drusen,” appears as numerous, uniform, round, yellow-white punctuate accumulations under the RPE. Cuticular drusen are usually seen on OCT imaging as elevations of the RPE with occasional disruption of the overlying IS/OS junction and ELM.81 Although cuticular drusen, subretinal drusenoid deposits, and soft drusen are composed of common components, they are distinguishable by multimodal imaging because of differences in location, morphology, and the optical properties of the drusenoid material and the RPE. Late non-neovascular AMD: geographic atrophy: The natural history of GA has been described as a progressive condition that evolves through stages with loss of vision occurring over many years.89–91 Multiple imaging modalities have been used to document and quantify the area of GA. Until recently, color fundus photography was used as the standard method to image GA; however, the use of color photos can be challenging due to the reported difficulty in detecting and accurately delineating GA.91,92 Other imaging modalities such as fluorescein angiography, fundus autofluorescence, and SD-OCT imaging are now used to evaluate and quantify GA (Fig. 3.16). Although these imaging modalities provide different information, none has been shown to be superior to the other. Fig. 3.16 Geographic atrophy (GA). (A) Color fundus image of the left eye of a 74-year-old male with central GA secondary to age-related macular degeneration. (B) Fundus autofluorescence (Spectralis, Heidelberg) showing a central area of hypoautofluorescence corresponding to the GA seen on the color image. (C) Late-phase fluorescein angiography showing a central window defect corresponding to the GA. (D) Horizontal B-scan through the foveal center demonstrating retinal thinning, loss of the retinal pigment epithelium (RPE), and photoreceptors. The loss of photoreceptors (yellow arrows) often extends beyond the margins of the RPE loss (white arrows). Observe the increased light penetration in the areas where the RPE is absent (bracket) and thin choroid (arrowhead). (E) Optical coherence tomography fundus image (courtesy of Cirrus, Carl Zeiss Meditec), showing the GA as a bright area. GA is seen clinically as one or more well-demarcated areas of hypopigmentation or depigmentation due to the absence or severe attenuation of the underlying RPE. The larger, deeper choroidal vessels are more readily visualized through the atrophic areas, and are accompanied by varying degrees of photoreceptor and choriocapillaris loss. Associated retinal atrophy is seen as thinning or loss of the outer nuclear layer and the absence of ELM and IS/OS junctions.93,94 The loss of photoreceptors often extends beyond the margins of GA, with the ELM and IS/OS junctions disappearing while bridging across the GA margin.95 Evaluation of these junctional zones may provide information about the pathogenesis of GA, and the role of RPE, photoreceptor, and choriocapillaris loss in the initiation and propagation of this condition.95 SD-OCT has been shown to be useful in detecting some of these morphologic alterations (Fig. 3.16D). With the use of SD-OCT enhanced depth imaging (EDI) protocols, it is now possible to visualize the structure of the choroid in greater detail.29 EDI demonstrated that subfoveal choroidal thickness decreases with age and axial length.31 In a subset of elderly patients complaining of unexplained vision loss, abnormal choroidal thinning was identified, and this condition was named “age-related choroidal atrophy.”96 Future studies are necessary to confirm if this represents a new clinical entity or a subtype of AMD. In contrast, the choroidal thickness appears to be unaffected in early non-neovascular AMD patients.97 SDOCT can also be used to quantify the areas of GA and monitor the progression of the disease. GA is currently imaged with SDOCT by using the OFI, which represents a virtual fundus image resulting from the en face summation of the reflected light from each A-scan. This en face OCT fundus image identifies GA as a bright area due to the increased penetration of light into the choroid where atrophy has occurred in the macula. The absence of the RPE and choriocapillaris is responsible for this increased penetration of light associated with GA. The OFI was shown to correlate well with the GA seen on clinical examination, color fundus imaging, and autofluorescence imaging (Fig. 3.16E).12,98,99 More recently, a newer algorithm provides an enhanced (partial) OFI, which is the summation of the reflected light from beneath the RPE (Fig. 3.17
Optical Coherence Tomography
Physical principles of optical coherence tomography
Device (manufacturer)
Axial resolution; scanning rate
Special characteristics
3D-OCT 2000
(Topcon, Tokyo, Japan)
5 µm; 27 kHz
Fundus camera
Bioptigen SD-OCT
(Bioptigen, Research Triangle Park, NC)
4 µm; 20 kHz
Designed for research applications
Cirrus HD-OCT
(Carl Zeiss Meditec, Dublin, CA)
5 µm; 27 kHz
RTVue-100
(Optovue, Fremont, CA)
5 µm; 26 kHz
SOCT Copernicus
(Optopol, Zawiercie, Poland)
6 µm; 27 kHz
Spectral OCT SLO
(Opko, Miami, FL)
6 µm; 27 kHz
Microperimetry
Spectralis OCT
(Heidelberg Engineering, Heidelberg, Germany)
8 µm; 40 kHz
Eye-tracking, fluorescein angiography, ICG angiography, autofluorescence
Quantitative analysis of OCT datasets
Normal macular anatomy
SD-OCT in retinal disorders
Vitreomacular traction
Epiretinal membrane
Macular hole
Age-related macular degeneration
Non-neovascular AMD
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree