Part 4 IR Anatomy
Case 108
Clinical History
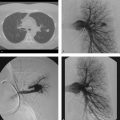
A 58-year-old woman with bilateral lower extremity claudication and diminished femoral pulses (Fig. 108‑1).

Key Finding
Collateral pathways in aortic occlusion.
Top 3 Collateral Pathway Categories
Systemic–systemic collaterals. These arteries arise from the common embryologic dorsal artery; this common origin allows for collateral communication to form in the setting of aortic occlusion. During development, these arteries divide and become intercostal arteries, lumbar arteries, iliolumbar, and superior gluteal arteries. Examples of common systemic–systemic collateral pathways in the setting of aortic occlusion include (1) descending thoracic aorta-intercostal-deep circumflex-external iliac artery, and (2) abdominal aorta-lumbar-iliolumbar-internal iliac- external iliac artery. Pathways form based on patient anatomy and the level and length of aortic occlusion.
Visceral–visceral or visceral–systemic collaterals. These collateral arterial pathways arise from the ventral aspect of a common embryologic structure—the paired ventral embryo-logic aorta. Ultimately, these vessels develop into the celiac axis, superior mesenteric, and inferior mesenteric arteries. Patient-specific anatomy, along with the morphology of aortic occlusion, will determine the configuration of the collateral pathway. When occlusions occur focally between the celiac axis and the superior mesenteric artery, blood flow may connect the celiac and superior mesenteric via the gastroduodenal or dorsal pancreatic arteries. In a small number of patients, a direct connection may exist, namely the arc of Buhler. In more distal aortic occlusions, specifically those below the superior mesenteric artery, typical collateral pathways include (1) aorta-superior mesenteric-marginal artery-superior rectal-internal iliac-external iliac artery, (2) aorta-superior mesenteric-arc of Riolan-inferior mesenteric artery-aorta, and (3) aorta-superior mesenteric-marginal artery-superior rectal-middle sacral-aortic bifurcation. A number of other collateral possibilities exist, but they will follow this common embryologic categorization.
Unusual pathways. Although essentially a systemic–systemic collateral pathway, Winslow’s pathway is unusual in that the origin of the pathway starts high in the thorax. Thus, angiographers must have a high clinical suspicion, and may need to perform angiograms cephalad to the arch. With Winslow’s, the collateral blood flow in distal aortic occlusion begins in the subclavian artery, through the internal mammary, to the inferior epigastric, and ultimately into the common femoral artery. Identification of this pathway is necessary prior to abdominal or thoracic surgery in order to help surgeons avoid bleeding or sacrificing this pathway leading to worsening of lower extremity ischemia. Other unusual collateral pathways in aortic occlusion include the lateral system that develops from the renal and gonadal arteries.
Diagnosis
Aortic occlusion with visceral–systemic collateral flow and flow via Winslow’s pathway.
Pearls
CTA is helpful in preoperative planning in suspected aortic occlusive disease, with 95% sensitivity and 96% specificity for stenosis of at least 50%.
Winslow’s pathway was originally thought to be seen only in the setting of aortic coarctation but has now been seen in adult-onset aortic occlusive disease.
Suggested Readings
Hardman RL, Lopera JE, Cardan RA, Trimmer CK, Josephs SC. Common and rare collateral pathways in aortoiliac occlusive disease: a pictorial essay. AJR Am J Roentgenol 2011;197(3):W519–W524 Prager RJ, Akin JR, Akin GC, Binder RJ. Winslow’s pathway: a rare collateral channel in infrarenal aortic occlusion. AJR Am J Roentgenol 1977;128(3):485–487Case 109
Clinical History
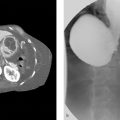
Worsening dysphasia (Fig. 109‑1).

Key Finding
Vascular rings and slings.
Top 3 Differential Diagnoses
Aberrant right subclavian artery. A left aortic arch with aberrant right subclavian artery occurs with a frequency of 0.4 to 2%. It results from involution of the embryonic aortic segment between the right subclavian and common carotid arteries. The right subclavian becomes the last aortic branch, reaching the right thorax by passing behind the trachea and esophagus in 85% and between them in 15%. The majority of cases are asymptomatic, although dysphagia (dysphagia lusoria) may occur.
Double aortic arch. A double aortic arch accounts for 40% of all thoracic vascular rings. It is caused by persistence of the right and left fourth branchial arches and is rarely associated with congenital heart disease. The aortic arches pass on both sides of trachea, joining posteriorly behind the esophagus and anteriorly in front of the trachea. The vascular ring produces tracheal and/or esophageal compression with possible airway compromise and dysphagia. Imaging findings include a right arch which is higher and larger than the left. Esophagography shows a reverse S pattern, with bilateral esophageal impressions on frontal views and a posterior impression on lateral views. Angiography demonstrates a right arch supplying the right subclavian and right common carotid and the left arch supplying the left common carotid and subclavian arteries.
Right arch with aberrant left subclavian artery. The most frequent branching pattern for a vascular ring with a right aortic arch is an aberrant left subclavian artery originating as the last branch of the aortic arch from a retroesophageal diverticulum of Kommerell. This occurs in 0.05 to 0.1% of the population. The ring is completed by a left-sided ligamentum arteriosum. A right aortic arch occurs when the right dorsal aorta remains patent and the left dorsal aorta regresses abnormally. Vascular rings encircle the trachea and/or esophagus and may cause esophageal compression and dysphagia. Cardiac defects, such as tetralogy of Fallot, occur in 5 to 10%. Radiographic findings include the presence of a right aortic knob, slight leftward deviation of the lower trachea, and posterior indentation of the trachea. Esophagography may show a right-sided indentation of the esophagus and posterior indentation of the esophagus. Cross-sectional imaging and angiography demonstrate the right arch and aberrant left subclavian artery arising from the protuberant diverticulum of Kommerell, which should not be mistaken for a pseudoaneurysm.
Additional Diagnostic Consideration
Pulmonary sling: About 5% of thoracic vascular rings are due to a pulmonary artery sling. The left pulmonary artery arises from the posterior aspect of the right pulmonary artery and passes between the trachea and esophagus to reach the left hilum. The left pulmonary artery thus forms a sling around the distal trachea and the proximal right main bronchus. There may be associated malformations of the bronchotracheal tree, as well as cardiovascular abnormalities. This may mimic a mediastinal mass on chest radiographs. CT or MR imaging may establish the diagnosis.
Diagnosis
Double aortic arch.
Pearls
The majority of aberrant right subclavian arteries pass posterior to the esophagus and are asymptomatic.
A double aortic arch joins posteriorly behind the esophagus and anteriorly in front of the trachea.
Right arch with aberrant left subclavian artery is a complete ring, causing tracheal/esophageal compression.
Pulmonary sling (left pulmonary artery arising from the right) passes between the trachea and esophagus.
Suggested Reading
Castañer E, Gallardo X, Rimola J, et al. Congenital and acquired pulmonary artery anomalies in the adult: radiologic overview. Radiographics 2006;26(2):349–371Case 110
Clinical History
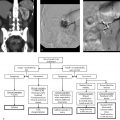
Three different patients undergoing angiography of the liver for chemoembolization (Fig. 110‑1).

Key Finding
Aberrant hepatic artery anatomy.
The standard branching pattern to the hepatic arterial supply begins at the celiac trunk, where the splenic, left gastric, and common hepatic arteries trifurcate. The common hepatic artery becomes the proper hepatic artery at the bifurcation with the gastroduodenal artery. Finally, the proper hepatic artery bifurcates into the left and right hepatic arteries.
A replaced artery is a substitute or “replacement” for the normally positioned artery, which is absent. An accessory artery is one that provides additional supply to an organ, or acts as an “accessory,” to the normally positioned artery.
Top 3 Differential Presentations
Aberrant right hepatic artery. The most common nonclassic location for the right hepatic artery origin is as the first branch from the superior mesenteric artery. Additional locations of aberrant origin include off the celiac, the left hepatic, gastroduodenal, or aorta.
Aberrant left hepatic artery. Although arising from the common/proper hepatic artery in the large majority of patients, the left hepatic artery may originate from the left gastric artery about 12% of the time. Other origins for the left hepatic artery include directly off the celiac trunk, or the gastroduodenal artery.
Aberrant common hepatic artery. There is rare, but broad variety to the origin of the common hepatic artery. The normal origin location arises from the celiac trunk in up to 85% of patients. About 3% of all individuals have the common hepatic artery originate directly from the aorta, with similar numbers originating from the left gastric or superior mesenteric arteries. Rarely is the origin from the right renal.
Diagnosis
Examples of aberrant hepatic artery anatomy.
Pearls
About 60% of patients have standard hepatic arterial anatomy; 20% have a variant left hepatic artery origin; 15% have variant right hepatic arterial origins; and 4% have a variant origin to the common hepatic artery.
Having both the left and right hepatic arteries replaced in the same individual occurs about 1% of the time.
CT and MR angiography are excellent tools in delineating aberrant liver anatomy and should be considered important in the work-up and evaluation prior to hepatic endovascular and surgical procedures.
Suggested Reading
Covey AM, Brody LA, Maluccio MA, Getrajdman GI, Brown KT. Variant hepatic arterial anatomy revisited: digital subtraction angiography performed in 600 patients. Radiology 2002;224(2):542–547Case 111
Clinical History
A 65-year-old man, with hepatocellular carcinoma in segment 8, status post oily transarterial chemoembolization (TACE), with residual enhancement of the medial aspect of the tumor on 1-month follow-up imaging (Fig. 111‑1).

Key Finding
Extrahepatic or parasitized blood supply to hepatocellular carcinoma (HCC).
Factors that should increase suspicion of extrahepatic collateral arterial supply to HCC include (1) exophytic tumor growth, (2) invasion of adjacent organs, (3) connection to hepatic ligaments/bare area, (4) presence of dilated extrahepatic arteries on CT/MR, (5) a peripheral stain defect in the tumor after oily chemoembolization, (6) local recurrence peripherally, and (7) sustained elevation of tumor markers post-TACE.
Top 3 Vascular Considerations
Right inferior phrenic. When there is extrahepatic supply to an HCC, the most common culprit vessel is the right inferior phrenic artery. This supply has been seen and embolized in as many as 25% of patients undergoing TACE (repetitive) in a large series of about 3,000 patients. The phrenic arteries may have a common trunk, or be individual, and most commonly originate off the celiac trunk or the aorta. Rarely, the inferior phrenic arteries originate from the renal arteries, left gastric artery, or a hepatic artery.
Omental artery. Arising from the right or left gastroepiploic arteries (off the gastroduodenal or splenic, respectively), the omental artery is typically a small vessel (2–3 mm in diameter). In parasitizing HCC, the vessel hypertrophies and becomes readily apparent on angiography. This is an infrequent finding, seen in up to 6% of HCC patients undergoing TACE.
Adrenal artery. Another infrequent vessel providing extrahepatic arterial supply, seen in up to 5% of HCC patients during TACE, is the adrenal artery. The adrenal supply may be off the inferior phrenic, the lateral aorta, or directed superiorly from the ipsilateral renal artery.
Additional Consideration
Rare extrahepatic arterial supply in HCC: There are other extrahepatic arteries (not included above) that have been seen on TACE to supply HCC. This happens less than 2% of the time. Arteries to consider in this setting include intercostals, the cystic, either internal mammary (right greater than left), the left inferior phrenic, renals, branches of the superior mesenteric artery, either gastric, and the lumbars.
Diagnosis
Extrahepatic blood supply to HCC from the right inferior phrenic artery.
Pearls
When an HCC is high in segment 7, abutting the diaphragm, selective angiography of the right inferior phrenic is important to ensure complete treatment during chemoembolization procedures. When in segments 2 and 3, abutting the left diaphragm, one should check the left inferior phrenic artery.
High-quality CTA or MRA is important in pre-TACE planning, helping the interventionalist to create a tailored embolization approach in each patient with HCC.
Suggested Reading
Kim HC, Chung JW, Lee W, Jae HJ, Park JH. Recognizing extrahepatic collateral vessels that supply hepatocellular carcinoma to avoid complications of transcatheter arterial chemoembolization. Radiographics 2005;25(Suppl 1): S25-S39Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree