Part 5 Postprocedural Cases
Case 120
Clinical History
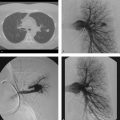
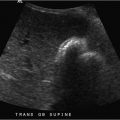
A 57-year-old man with right leg and groin pain after cardiac catheterization (Fig. 120‑1 ).

Key Finding
Postprocedure arterial access complications (local).
Top 3 Differential Diagnoses
Hematoma. The most frequent complication of diagnostic angiography, minor hematomas occur in up to 10% of groin arterial access cases. Minor hematomas are typically self-limited. Major hematomas, defined as those requiring transfusion, surgical evacuation, or delay in discharge, occur in 0.5% of femoral arterial punctures. Differentiating hematoma from pseudoaneurysm can be done by physical examination; however, confirmation with ultrasound is the norm.
Pseudoaneurysm. Also known as false aneurysms, these areas of dilated vessel lack all three arterial layers. Therefore, blood flows out of the vessel, is contained by juxta-arterial tissues (connective tissue and hematoma), and is returned to the original vessel. This creates the classic “yin-yang” or “to-and-fro” appearance on ultrasonography. Causative etiologies include infection, trauma (both penetrating and blunt), and iatrogenia. Iatrogenic pseudoaneurysms form at a rate of 0.1 to 0.2% in diagnostic angiography and up to 2% in interventional cases. Risk factors include a “high” or “low” puncture of the femoral artery, the use of anticoagulants or antiplatelet agents, thrombocytopenia, thrombolysis cases, and the use of large devices. On physical examination, the presence of a pulsatile mass is virtually diagnostic. The presence of a bruit is helpful, but is not specific, as other complications may present with a bruit (e.g., dissection, stenosis, arteriovenous fistula). Pseudoaneurysms are painful and may lead to rupture, distal embolization, or overlying skin ischemia. Treatment options include ultrasound-guided compression, percutaneous thrombin (or collagen) injection under ultrasound guidance, endoluminal coils or stent-graft placement, or surgery. Percutaneous options are usually the first-line treatments. Surgery is required with rapid expansion, distal ischemia, neurologic deficits, failure of percutaneous interventions, infection, or compromised soft tissue viability.
Arteriovenous fistula (AVF). This abnormal communication, between an artery and an adjacent vein, occurs secondary to access of ipsilateral vessels or unnoticed puncture of the vein when accessing the artery. Fistulae form rarely after diagnostic arteriography, at rates less than 0.2%. Clinically, patients complain of leg pain and swelling, and a bruit is audible on physical examination. Ultrasound interrogation will reveal arterialized flow in the adjacent vein. The site of communication can usually be determined. Treatment options include stent-graft placement, coiling of the communication, or open surgery.
Diagnosis
Pseudoaneurysm.
Pearls
Groin hematomas are the most common puncture site complication; the vast majority are self-limiting.
Pseudoaneurysms present as pulsatile masses with a classic “yin-yang” or “to-and-fro” appearance on duplex ultrasound.
AVFs most often result from inadvertent venous puncture during arterial access.
AVFs reveal arterialized flow in the adjacent vein; they can be treated endovascularly or with surgery.
Suggested Readings
Lenartova M, Tak T. Iatrogenic pseudoaneurysm of femoral artery: case report and literature review. Clin Med Res 2003;1(3):243–247 Singh H, Cardella JF, Cole PE, et al; Society of Interventional Radiology Standards of Practice Committee. Quality improvement guidelines for diagnostic arteriography. J Vasc Interv Radiol. 2003;14(9 Pt 2):S283-S288Case 121
Clinical History
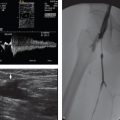
A 55-year-old man who presents with progressive left leg pallor and pain after undergoing a left common femoral artery access for a right leg interventional procedure earlier today (Fig. 121‑1 ).

Key Finding
Post groin arterial access leg ischemia.
Top 3 Differential Diagnoses
Arterial occlusion (thrombus/embolus). During and after groin artery access, a variety of thromboembolic issues can arise. Thrombi can form along the catheter or sheath, and become dislodged, embolizing down the leg. To combat this, sheaths and catheters are frequently flushed with saline, often mixed with heparin. Injury to the artery such as dissection is rare, but can also lead to foci of clot formation with local occlusion or subsequent distal embolization. Risk factors for acute thrombosis or embolus leading to limb ischemia include small-caliber artery, female sex, peripheral artery disease, diabetes, larger-sized sheaths/catheters, long dwell times of devices, and inadvertent cannulation of the smaller superficial or profunda femoral artery instead of the common femoral artery. Patients postprocedure with a symptomatic groin site or ipsilateral leg ischemia should undergo prompt evaluation, often with duplex ultrasound. Depending on ischemia severity, and occlusive lesion morphology, prompt endovascular or open procedures are necessary to repair the insult.
Steal (arteriovenous fistula). In those with arteriovenous fistula post groin arterial access, flow diversion into the vein can be extensive enough to steal blood from the distal limb. This steal syndrome is rare, and can present with intermittent claudication or even more severe issues with heart failure or distal limb ischemia. Duplex ultrasound will often be diagnostic, and is readily available. Fistulae leading to steal should be promptly closed.
Closure device malfunction. Leg ischemia postclosure device can occur due to a variety of circumstances and depends on the closure device used. Rates of complication are low, but when patients present with symptoms postclosure device placement, operators must have a high index of suspicion. One popular device, the Angio-Seal (St Jude Medical, St Paul, MN), deploys a collagen footplate and plug on the inside and outside of the arteriotomy, respectively. Multiple types of complications have been reported, such as deploying the plug within the lumen, attempted closure of the smaller superficial femoral artery leading to footplate occlusion of the vessel, embolization of device components down the artery, and disruption of the arterial wall leading to dissection or bleeding. Each of these complications can lead to ischemia of varying degrees. Duplex ultrasound may be able to identify the plug within the lumen, with the associated suture material, but clinical suspicion must remain high in order to make the diagnosis, especially in delayed presentations. Open surgical removal and arterial repair is the treatment of choice for malpositioned or malfunctioning closure devices, although some operators attempt to angioplasty vessels that are physically occluded by certain devices.
Diagnosis
Left femoral arteriovenous fistula.
Pearls
Duplex ultrasound is usually the first study in patients with postprocedure leg ischemia, being able to diagnose local occlusive disease, steal, or some closure device issues. Despite a negative ultrasound, one should maintain a high index of suspicion. When symptoms persist, or when they intensify, angiography or open exploration should be strongly considered.
Ultrasound-guided femoral artery access helps to avoid complications such as inadvertent venous traversal and high or low arterial cannulation.
Case 122
Clinical History
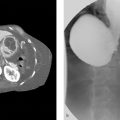
A 73-year-old man with a 6-cm infrarenal abdominal aortic aneurysm for endovascular aortic repair (EVAR) (Fig. 122‑1 ).

Key Finding
EVAR complications.
Grouping of complication types is based on those reported by The Society of Vascular Surgery and the American Association of Vascular Surgery.
Top 3 Complication Types
Deployment-related. Issues that occur during the physical deployment of the endograft include aortic dissection, arterial perforation, peripheral embolization, and access site complications (e.g., hematoma, pseudoaneurysm, and infection). Malposition of an endograft over a renal artery origin is a relatively common deployment-related complication, occurring 2 to 4% of the time, especially in short aneurysm necks. This should be dealt with immediately to avoid renal infarction, given the lack of collateral flow to the kidneys. A graft “pull-down” (placing a stiff guidewire from one groin access, over the bifurcation of the endograft, and out the other groin access, then pulling down), can move the device caudally, uncovering the renal artery origins. Bare metal stents may also open a partially covered renal artery. Relative to open repair, the rate of local, deployment-related complications is slightly higher in EVAR.
Systemic. Systemic complications include resultant injury to the heart, lungs, kidneys, brain, blood (e.g., blood loss), and gastrointestinal tract, all likely related to physiologic changes due to anesthesia, manipulation during the procedure, and the presence of the graft. Another systemic complication is that of “postimplantation syndrome,” which consists of fever, leukocytosis, elevated inflammatory markers (e.g., C-reactive protein), abdominal tenderness, all in the absence of infection. Unlike deployment-related complications, systemic complications are less common with EVAR than in open repair.
Implant-related. Problems specific to the graft device include future aneurysm rupture, migration, endoleak, graft limb issues, aortoenteric fistula, graft infection, and peripheral ischemia.
Migration: Endografts may move over time, caudally from the proximal fixation, or cranially from the iliac limbs. Any movement of 5 to 10 mm is considered significant. Rates of migration range from 5 to 15%, increasing in frequency with length of time implanted, and continued degeneration of the aneurysm sac. Treatment is usually accomplished with endograft aortic cuff extenders with or without fixation using large Palmaz type stents or endoanchors.
Endoleaks: Endoleaks are diagnosed when there is blood flow still within the aneurysm sac post-EVAR. These can be seen immediately at angiography, or in a delayed manner, usually diagnosed by contrast-enhanced CT. Endoleak types are numbered as follows: (I) leak from graft ends (a, proximally; b, distally); (II) sac filling via branch vessels; (III) leak through a defect in the graft fabric; (IV) graft porosity; and (V) endotension. Treatment may be graft extension, sac or branch vessel embolization, or relining the graft with another device.
Graft limb complications: Uncommon problems arise with the iliac limbs due to kinking, twisting, stenosis, or thrombosis. The prevalence of graft limb complications is about 2 to 3%. Patients typically present with lower extremity ischemic symptoms. Endovascular treatment with angioplasty and/or stent placement, or thrombolysis when needed, is usually adequate.
Diagnosis
EVAR coverage of right accessory renal artery leading to renal infarction. Associated type II endoleak.
Pearls
According to the EUROSTAR collaboration, patients post-EVAR continue to have an approximately 1% rupture rate per year.
The most common type of endoleak is type II. Most of these resolve spontaneously. When persistent, they require embolization.
Suggested Reading
Grande W, Stavropoulos SW. Treatment of complications following endovascular repair of abdominal aortic aneurysms. Semin Intervent Radiol. 2006;23(2):156–164Case 123
Clinical History
A 59-year-old man with head and neck cancer with request to place a gastrostomy tube prior to initiating radiation treatments (Fig. 123‑1 ).

Key Finding
Early complications of percutaneous gastrostomy tube placement.
Top 3 Differential Diagnoses
Peritonitis. The severe complications of peritonitis—fever, leukocytosis, and abdominal pain with signs of peritoneal irritation—carry a high mortality rate. It is important to identify this complication early on and initiate supportive care and broad-spectrum antibiotics. Abscesses can be drained percutaneously. Abdominal free air is not synonymous with peritonitis, and should be observed if noted in isolation. When free air is seen with peritonitis, altered mental status, or in patients who are difficult to examine, advanced imaging or surgical exploration is reasonable. Beyond simple infection, causes of postgastrostomy peritonitis are as follows:
Tube dislodgement prior to tract maturity: Tract maturation occurs between 1 and 3 weeks. Tubes that become dislodged during this time period require prompt attention. Attempts to replace the tube within 24 to 48 hours are successful 80 to 90% of the time, especially in the setting of t-fastener gastropexy. If the tube cannot be replaced, bowel rest and nasogastric tube decompression is recommended for 48 hours, and a new tube can be placed thereafter. If peritonitis develops, supportive therapy and antibiotics are the minimal treatment, and surgical consultation is recommended. Tube migration distally into the gastrointestinal tract can lead to balloon blockage gastric outlet obstruction, or maladministration of feedings. Fluoroscopic-guided tube repositioning often fixes these issues.
Peritoneal leakage around tube: Leakage of gastric contents into the peritoneum around gastrostomy tubes occurs when the gastropexy is loose, due to over distention of the stomach, or due to the need to oversize the gastrostomy site to allow tube placement (e.g., using a 22-F peel away sheath to advance an 18-F balloon anchoring tube into the stomach). Treatment options include suction decompression of the stomach, tightening the t-fasteners to improve the gastropexy, repositioning the intragastric balloon toward the gastrostomy, and upsizing the tube, in addition to treating peritonitis.
Colon perforation: Although rare, traversal of the colon during gastrostomy tube placement can lead to peritonitis. The transverse colon is typically the site injured. T-fasteners can lead to colon obstruction. Patients with minimal symptoms can be managed conservatively, leaving the now colostomy tube in place until tract maturation. If there is peritonitis, surgery is typically necessary.
Hemorrhage. Hemorrhage is a complication common to surgery, and is treated, based on severity, location, and comorbidities. Treatment is with fluid resuscitation, transfusion of blood products, embolization, endoscopy, or open surgery.
Aspiration. Aspiration leading to pneumonitis or pneumonia is one of the more likely complications postgastrostomy, carrying a high mortality when it occurs. Prevention is the key, and is best accomplished by avoiding oversedation, keeping the stomach decompressed immediately postprocedure, limiting feedings if high residuals or ileus are noted, and maintaining head elevation. When aspiration occurs, initial pulmonary toilet is followed by observation for at least 48 hours. Start antibiotics if the patient does not improve during this observation period.
Diagnosis
Early gastrostomy tube dislodgement with percutaneous replacement.
Pearls
Abdominal free air postpercutaneous gastrostomy tube placement occurs about 5 to 10% of the time. Free air not related to peritonitis can be observed, and typically resolves within 2 to 3 weeks.
Major complications after radiologic gastrostomy occur between 1 and 5% of the time, a rate that is less than that of endoscopic or surgical gastrostomy tube placement.
Suggested Readings
Covarrubias DA, O’Connor OJ, McDermott S, Arellano RS. Radiologic percutaneous gastrostomy: review of potential complications and approach to managing the unexpected outcome. AJR Am J Roentgenol 2013;200(4):921–931 Perona F, Castellazzi G, De Iuliis A, Rizzo L. Percutaneous radiologic gastrostomy: a 12-year series. Gut Liver 2010;4 Suppl 1 :S44-S49Case 124
Clinical History
A 71-year-old man who is chronically dependent on a gastrostomy due to prior stroke, is brought in for evaluation of a broken gastrostomy tube hub (Fig. 124‑1 ).

Key Finding
Late gastrostomy tube complications.
Top 3 Differential Diagnoses
Migration/dislodgement. Tubes may migrate distally, causing gastric outlet obstruction. Fluoroscopic-guided reposition is simple and effective in this situation, with attention to ensuring proper fixation in the future. When a late gastrostomy tube becomes dislodged, tube replacement within a 24- to 48-hour time course is nearly 100% successful. Bedside replacement with a smaller tube (e.g., a Foley catheter) can hold open the gastrostomy until fluoroscopic replacement is feasible. After bedside replacement, contrast injection and abdominal radiography should be done to ensure proper location.
Exit site issues/leakage. Having a small amount of pericatheter leakage is typical of gastrostomy tubes, requiring a few dressing changes per day. More voluminous leakage, or that associated with skin breakdown, requires attention. Leakage often occurs early on until a tight tract forms. Later on, leakage may be due to mechanical widening of the tract, excessive gastric pressurization (e.g., in gastroparesis), or formation of a fistula between the gastric lumen and the tract. Leakage will often lead to skin breakdown. Treatments include local measures such as absorbing agents (e.g., pectin powder) and skin protectants (e.g., zinc oxide), or proton pump inhibitors to decrease the acidity of the leaking fluid. Tightening the balloon or bumper retention device can help temporarily, but excessive tension can induce ischemia and impair tract healing. This can lead to “buried bumper syndrome” creating a large soft tissue defect. Upsizing the tube can help, but if the cause of leakage persists (e.g., excessive gastric pressure), leakage will continue along a larger hole. Finding a tract fistula is difficult, requiring a high index of suspicion, and a careful tract-o-gram. Abandoning sites complicated by fistulae, and placing a new gastrostomy, is the most effective strategy in this type of leakage. When peristomal infection occurs, antibiotics are effective.
Tube occlusion. Prevention of tube occlusion is important to maintain adequate nutrition and hydration for tube-dependent patients. Occlusion is often related to instillation of crushed medications or inadequate flushing. Flushing regimens should recommend 30 to 60 mL of fluid flushed after tube feeds, or every 3 to 4 hours in the setting of continuous feeds. When obstruction occurs, tube irrigation with saline via a syringe is the first step. Care should be taken to not use excessive force when flushing to avoid tube rupture. Instilling occluded tubes with carbonated beverages or pancreatic enzymes can also clear tubes. If these methods fail, the tube should be exchanged over a wire. A stiff hydrophilic wire tends to be the most effective device to pass through occlusion material. If unable to pass a wire through the tube lumen, passing a wire and catheter along the tube can help preserve the tract. In the setting of a well-matured tract, simply removing the occluded tube, and replacing it with another, works without issue on most occasions.
Diagnosis
Tube breakage with retention balloon dysfunction.
Pearls
Buried bumper syndrome occurs late (median 35 months) when the tube retention mechanism is too tight for the tract. This leads to the retention device eroding into the soft tissues of the abdominal wall and major tissue breakdown. The incidence of buried bumper syndrome is reportedly up to 20% with chronic tube use.
Peristomal infections occur around gastrostomy tube sites at a rate of 5 to 25%.
Suggested Reading
Huang SY, Engstrom BI, Lungren MP, Kim CY. Management of dysfunctional catheters and tubes inserted by interventional radiology. Semin Intervent Radiol. 2015;32(2):67–77Case 125
Clinical History
A 78-year-old man with bilateral distal ureteral obstruction due to prostate cancer leading to obstructive renal failure (Fig. 125‑1 ).

Key Finding
Early complications postnephrostomy.
Top 3 Complications
Hemorrhage. Postnephrostomy hemorrhage is usually self-limiting. Most patients experience mild hematuria that resolves over the next 1 to 3 days. Small, nonexpanding, minimally symptomatic hematomas are reported about 10 to 15% of the time, and most often can be managed conservatively. However, when there is frank hematuria (with clotted blood, not just bloody urine), a decreasing hemoglobin/hematocrit level, hemodynamic instability, or bleeding that persists beyond 3 to 5 days, one should suspect serious vascular trauma. Such serious bleeding occurs in about 1 to 3% of patients. In serious vascular trauma, the most common injury is to a renal lobar artery and embolization is effective in achieving hemostasis. Additional potential bleeding vessels include renal capsular and intercostal/muscular wall arteries.
Infection/sepsis. Sepsis with fever, leukocytosis, hypotension, and tachycardia, occurs 1 to 2% of the time postnephrostomy. Supportive measures and antibiotics are the mainstay of treatment, often in the ICU.
Postobstructive diuresis. After relief of a complete urinary obstruction (e.g., bladder outlet obstruction or bilateral ureteral obstruction), patients may experience a potentially lethal polyuric response. In this state, copious amounts of salt and water are eliminated via the kidneys. Although it is normal to eliminate volume and salts after relief of obstruction, in up to 50% of patients diuresis will continue even after homeostasis is achieved. This puts patients at risk of dehydration, electrolyte derangements, hypovolemic shock, and death. The diagnosis is made when there is excessive urine production, exceeding 200 mL/h over 2 consecutive hours or greater than 3 L in 24 hours. This may be self-limiting, correcting after 24 hours, or pathologic, extending beyond 48 hours. Careful monitoring of urine output, daily weights, and twice-daily serum electrolytes helps manage the disorder. Most advocate for replacement of 75% of the prior day’s loss typically using half-normal saline. Excessive resuscitation will exacerbate the condition. Nephrology consultation is helpful.
Additional Considerations
Early tube dislodgement: Dislodgement of recently placed nephrostomy tubes is problematic. In studies of nephrostomy tube replacement, risk factors for failure to reinsert via the same tract include an immature tract (≤ 100 days), and prolonged time from dislodgement (> 24 hours). If reinsertion is unsuccessful, new nephrostomy tube placement is usually necessary.
Adjacent organ injury: In specific clinical scenarios, nephrostomy tube placement needs to be directed via an upper pole access in order to aid future operators in stone extraction. This often necessitates an intercostal approach, via the 10th or 11th intercostal space. When this is necessary, pleural or pulmonary injury can happen. This can lead to pneumothorax. More commonly, this approach is complicated by pleural effusion (in up to 10%), which can be managed conservatively or by small-bore pleural drains. When hemothorax occurs, large-bore chest tubes and embolization may be required.
Diagnosis
Hematuria postnephrostomy.
Pearls
Major complications of percutaneous nephrostomy tube placement occur 4 to 5% of the time, with hemorrhage and sepsis being the most common. This mostly occurs after tract dilation to 30 F, excessive manipulation, or due to an intercostal approach.
The death rate postpercutaneous nephrostomy has been reported in the range of 0.05 to 0.3%.
Suggested Reading
Hart CYT, Ryu JH. Complications of percutaneous nephrostomy tube placement to treat nephrolithiasis. Hosp Physician 2002:43–46Case 126
Clinical History
A 55-year-old woman with pelvic malignancy leading to right ureteral obstruction, being treated with chronic right nephrostomy tube drainage, reports that the tube fell out 2 days ago (Fig. 126‑1 ).

Key Finding
Late issues with percutaneous nephrostomy.
Top 3 Issues
Leakage. Urine leakage typically indicates catheter obstruction, catheter injury, or that the nephrostomy tube is retracted into the tract. Leakage is very uncomfortable for the patient, and those caring for the patient, and should be addressed urgently. Physical examination and bedside tube flush will usually uncover the issue and is sometimes able to mitigate the situation (e.g., kink in catheter, tube obstruction with debris). Catheter exchange under fluoroscopy will almost always stop the leak. Urinary leakage can lead to skin breakdown and infection. If this becomes severe, local skin issues may necessitate loss of the current access site. Prevention of skin breakdown is a must.
Dislodgement. Nephrostomy tube dislodgment is very common, with an incidence of up to 35%. Tubes may present partially dislodged, leading to leakage or obstruction, or completely dislodged. When tubes are completely out, prompt attention is required to preserve the access site, and to avoid complications such as sepsis. Reinsertion is successful well over 90% of the time, especially if attempts are done in the first 24 to 48 hours, and the tract is mature.
Encrustation/obstruction. Tube obstruction can occur due to mineral deposits or debris. To prevent this, some advocate for daily saline flushes. Adequate hydration can help keep tubes open with plentiful urine output, and dilution of minerals. Infection and obstruction are related, another reason to maintain the nephrostomy tubes as clean as possible. To avoid obstruction and encrustation, most recommend routine nephrostomy tube exchanges at 1 to 4 month intervals. Tubes can become so encrusted that they are unable to be removed from the patient, leading to chronic infection and obstructive issues.
Additional Consideration
Bacterial colonization/infection: Chronic urinary tubes are almost always complicated by asymptomatic bacteriuria. Therefore, maintaining tube patency is important to prevent bacteremia and urosepsis. Additionally, bacteremia can occur during tube manipulation. Most advocate periprocedural antibiotics for tube exchanges or urinary manipulation, especially in patients with valvular heart disease.
Diagnosis
Nephrostomy tube dislodgement for percutaneous replacement.
Pearls
Urinary tube leakage should be addressed urgently due to patient discomfort and potential for skin breakdown with associated infection and potential loss of the access site.
Patients with urinary tubes should be regularly followed to ensure that the tubes are exchanged at adequate intervals to prevent encrustation, usually every 1 to 4 months.
Suggested Reading
Collares FB, Faintuch S, Kim SK, Rabkin DJ. Reinsertion of accidentally dislodged catheters through the original track: what is the likelihood of success? J Vasc Interv Radiol. 2010;21(6):861–864Case 127
Clinical History
A 64-year-old woman with history of pancreatitis and chronic abdominal pain, now with new-onset fever (Fig. 127‑1 ).

Key Finding
Management of abscess drainage catheters.
Image-guided drainage of infected fluid provides a clear benefit to patients with localized infection, in the setting of sepsis or not. Technical success is well over 90%, with defervescence and hemodynamic stability typically within 1 to 2 days. What to do with drains after placement is similarly important, as mismanagement can lead to increased morbidity and mortality. Interventional radiology teams should closely follow these patients to ensure proper care and to address any issues.
Top 3 Clinical Scenarios of Abscess Drain Management
Completed. In the typical drainage catheter scenario, there is a gradual decrease in tube output over time. If there is a sudden decrease in output, or leakage of fluid around the tube, the catheter should be flushed with 3 to 5 mL of normal saline to unclog the device. Drain removal can be done when the fluid collection has resolved, and the patient has improved. The specific goals are as follows: (1) catheter output should slowly taper to less than 10 mL/d, (2) patient defervescence, (3) resolution of leukocytosis, and (4) tolerating enteric feedings. If these goals are met, the catheter can be removed. CT imaging is not necessary prior to drain removal unless the drain was in a difficult place to access, or the drain traverses solid organ parenchyma. Additionally, one should consider pulling the drain over a wire if the course of the drain is through parenchyma, to ensure that there is no bleeding. This clinical scenario is the desired goal of percutaneous drainage.
Undrained fluid. During follow-up, certain patients may continue to have fever or leukocytosis, despite a slow tapering of the drain output to less than 10 mL/d. In this clinical scenario, the patient usually has persistent undrained fluid. This undrained fluid may be thick fluid within the targeted drainage site, loculated fluid within or adjacent to the original fluid collection, or a fluid collection at a completely different site. One management option at this point is to perform intracavitary fibrinolysis by injecting fibrinolytic medications. A simple protocol for doing this is to inject 2 mg of tissue plasminogen activator, suspended in 10 mL of fluid, into the targeted collection via the current drain. Allow the fibrinolytic to dwell for about 1 hour by capping the drain, and then opening the drain back to a bag or suction bulb. The goal in doing this is to address thick fluid within the cavity, or to open up loculations within or adjacent to the targeted collection. Another management option is early imaging, usually with CT, to look for the location of the suspected undrained fluid. After the CT, intracavitary fibrinolysis, tube upsize, or new tube placement at a remote site, can be done depending on the results of the imaging study. Ultimately, the goal will be to meet the criteria in the “completed” scenario.
Fistula. Still another clinical scenario is when the patient improves, with defervescence and normalization of leukocytosis, but the drainage output remains elevated. This is typical of fistulous communication between a hollow viscous and the targeted fluid collection. Other signs of fistula include seeing tube feeds in the output, fecalized output, or the character of the fluid being chylous, pancreatic, biliary, or that of urine. Sometimes fluid chemistry is necessary to determine the source. Fistulas are classified as low output when less than 200 mL/d, and high output when over 500 mL/d. This classification scheme can also help suggest the source of the output, the likelihood of closure, and associated mortality. Full diagnostic evaluation of the fistula requires contrast injections into the fluid collection cavity, and/or CT scanning using a variety of enteric or IV contrast agents (sometimes in a staged approach).
Diagnosis
Infected pancreatic pseudocyst with persistent pancreatic leak.
Pearl
Low-output fistulas (< 200 mL/d) are most likely to be from the colon; high-output fistulas (> 500 mL/d) are more likely to be from the small bowel.
Suggested Reading
Lorenz J, Thomas JL. Complications of percutaneous fluid drainage. Semin Intervent Radiol. 2006;23(2):194–204Case 128
Clinical History
A 50-year-old woman with right leg pain (Fig. 128‑1 ).

Key Finding
Contrast reactions.
Most patients who receive iodinated contrast media have no ill effects, and when reactions do occur, most are mild and self-limited. The overall incidence of contrast reactions for low osmolality agents ranges from 0.2 to 0.7%. Moderate reactions have pronounced symptoms that require medical management. Severe reactions occur after 0.01 to 0.02% of agent administrations, and are life-threatening, often requiring more advanced medical assistance (e.g., Code Blue). Management is based on the type of reaction and the severity.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree