Given the rapid expansion of interventional radiology (IR) and wide adoption of its treatments in medical guidelines, there is increasing patient and clinician expectation for IR teams that can provide high-quality treatments in a safe and effective manner. Delivering this advanced level of care is contingent on safe and reproducible processes and pathways for IR clinicians and other team members. Furthermore, a successful interventional radiologist must have strong clinical skills and be familiar with relevant disease processes, alternative treatment options, and possible complications and their initial management.
Lack of proper preprocedural planning and ineffective communication with healthcare team members or the patient are often the reasons for adverse clinical outcomes and patient complaints. The interventional preprocedural care starts the moment a referral is received and continues until the patient is safely transferred to the procedure table. The key steps involved in the preprocedural care are outlined in Fig. 2.1 .
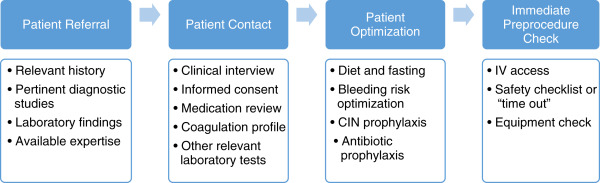
Patient Referral
The procedural planning starts the moment a patient is referred to the IR team. On most occasions, it is a one-to-one conversation between the operator and referring physician. Relevant history and careful review of available imaging and laboratory studies provide the essential framework for agreeing to or rejecting the requested procedure. Risk factors that may require a delay or modification of the proposed procedure or adaptation of an alternative therapeutic approach should also be taken into consideration. Most referrals for interventional procedures are straightforward and involve the interventional radiologist and the referring physician, whereas a multidisciplinary team approach is essential when complex procedures are to be performed, and in specialty settings such as cancer treatments. A multidisciplinary team meeting is the platform for collective decision-making on best available treatment options and for agreement on an alternative plan of management should any failure or complication occur after the procedure. In addition, the operator should fairly assess his or her own expertise and make sure that the assistance/guidance warranted for the procedure is available.
Patient Contact
Clinical Interview
The patient contact should ideally occur at a time and place remote from the procedure because the patient or decision-maker needs to be attentive and focused, and free from undue stress while discussing the procedure. Inpatients should be assessed during a bedside visit, ideally the day before the procedure. Elective cases must be evaluated in a dedicated interventional clinic or combined clinics with other disciplines. Holding the discussion between the patient and the interventional radiologist in a place outside the procedure room greatly helps in establishing a rapport and allaying the patient’s anxiety. Similar to any other clinical discipline, the clinic or bedside visit consists of such general steps as detailed history, focused physical examination, review of drug chart, allergies, and relevant imaging and laboratory studies. In addition, for vascular procedures, the interventional radiologist should evaluate the proposed puncture site, the condition of extremities, and distal pulses. The proposed site should be examined for preexisting pseudoaneurysm, hematoma, inguinal hernias, recent injury, or surgery to the site of puncture. Presence of any of these should result in evaluation of an alternate access site and change to the initially planned approach.
The risk factors for contrast-induced nephropathy such as preexisting renal impairment, intravascular volume depletion, or concomitant use of nephrotoxic drugs should be actively assessed and appropriately addressed. Similarly, relevant history and review of medication should be performed to evaluate for bleeding risk. Appropriate laboratory tests should be discussed with the patient along with steps to optimize them before the procedure. The level of sedation required based on the complexity of the procedure and patient’s pain threshold should be discussed and timely referral to anesthetist done. Before proceeding to the informed consent, the interventionalist should have a thorough understanding of the patient’s underlying medical condition, comorbidities, potential risk, and alternative treatment options and must believe that the proposed procedure is the best available treatment for the patient’s condition.
A careful review and discussion of prior imaging studies is perhaps the most important step in planning the IR procedures. Such a review helps in planning complex procedures in advance, choosing the appropriate equipment, choosing a safer alternative approach or even an alternative treatment, and optimizing any predisposing factors that would complicate the intended procedure. This will also reduce procedure time and the radiation exposure ( Box 2.1 ).
General steps
- •
History and directed physical examination
- •
Review of current medications/allergies
- •
Review of laboratory findings
- •
Discuss relevant imaging studies
- •
Evaluations specific to interventional radiology procedures
- •
Proposed puncture site
- •
Bleeding risk assessment
- •
Risk of contrast-induced nephropathy
- •
Requirement of sedation
- •
Laboratory Test and Coagulation Profile
The risk of hemorrhage and consequent recommendations for laboratory testing depend on the nature of the planned procedure, the patient’s underlying health, and any ongoing antiplatelet or anticoagulant therapy. The risks of bleeding after image-guided procedures can be divided into three categories as suggested by the Society of Interventional Radiology consensus statement. This classification is based on the organ system, access site, size of interventional devices used, and potential for hemorrhage in a location that allows no or poor prompt control of the bleeding when it occurs.
For procedures with low risk of bleeding, the preprocedural value of the international normalized ratio (INR) for prothrombin time is recommended if the patient is taking warfarin or has liver disease. Activated partial thromboplastin time (aPTT) should be measured if the patient is on heparin therapy. Platelet counts and hemoglobin levels are not routinely checked. If the procedure has a moderate risk of bleeding, the preprocedural INR level or aPTT (ongoing intravenous heparin) should be evaluated. With procedures that incur a high risk of bleeding, it is recommended that the INR level or the aPTT (if the patient is receiving intravenous heparin), the platelet count, and the hematocrit level all be checked.
Regardless of the risk of bleeding, the preprocedural platelet count should be at least 50,000/μL. For procedures with low bleeding risks, the INR should be corrected if it is greater than 2. For procedures that have a moderate or high risk of bleeding, the INR should be corrected if it is greater than 1.5. Similarly, for moderate- and high-risk procedures, a prolonged aPTT should be corrected by withholding the unfractionated heparin before the procedure, and/or transfusing fresh frozen plasma with the threshold being 1.5 × control ( Table 2.1 ).
| Bleeding Risk | |||
|---|---|---|---|
| Low, Easily Detected and Controllable | Moderate | High, Difficult to Detect or Control | |
| Procedures | Vascular Dialysis access interventions Venography Central line removal IVC filter placement PICC placement Nonvascular Drainage catheter exchange Thoracentesis Paracentesis Superficial aspiration and biopsy (excludes intrathoracic or intraabdominal sites) Superficial abscess drainage | Vascular Angiography, arterial intervention with access size up to 7F Venous interventions Chemoembolization Uterine fibroid embolization Transjugular liver biopsy Tunneled central venous catheter Subcutaneous port device Nonvascular Intraabdominal, chest wall, or retroperitoneal abscess drainage or biopsy Lung biopsy Transabdominal liver biopsy Percutaneous cholecystostomy Gastrostomy tube: initial placement Ablation: straightforward Spine procedures (vertebroplasty, kyphoplasty, lumbar puncture, epidural injection, facet block) | Vascular TIPS Nonvascular Renal biopsy Biliary interventions (new tract) Nephrostomy tube placement Ablation: complex |
| Laboratory testing | INR: for patients receiving warfarin or known or suspected liver disease aPTT: for patients receiving intravenous unfractionated heparin Platelet count: not routinely recommended Hematocrit: not routinely recommended | INR: recommended aPTT: recommended in patients receiving intravenous unfractionated heparin Platelet count: not routinely recommended Hematocrit: not routinely recommended | INR: routinely recommended aPTT: routinely recommended in patients receiving intravenous unfractionated heparin infusion, no consensus on patients not receiving heparin Platelet count: routinely recommended Hematocrit: routinely recommended |
| Management | INR: correct to < 2 (FFP, vitamin K) aPTT: no consensus Platelets transfusion if counts < 50,000/μL | INR: correct to < 1.5 aPTT: correct if value > 1.5 × control Platelets transfusion if counts < 50,000/μL | INR: correct to < 1.5 aPTT: stop or reverse heparin for values > 1.5 × control Platelets transfusion if counts < 50,000/× l |
Consent
Informed consent is the process in which a patient makes a decision whether to have a given treatment, based on information given by a healthcare provider. Consent is a legal and ethical requirement that must be upheld by all clinicians. The information provided should include the diagnosis and prognosis, purpose and main stages of procedure, common and significant risks/complications, other alternative procedures, key people involved in the procedure and their roles, and the right to refuse treatment and to seek a second opinion. In order to give consent, the patient should be competent, which in general requires being able to understand and retain the information and to weigh the potential risks against benefits. Finally, the decision on consent should be made freely without pressure from anyone. Consent can be withdrawn by the patient at any time before administration of treatment.
Consent in IR
A large proportion of IR procedures are elective and for these cases, consent should be secured in advance of the procedure, so that the patient can have adequate time to consider the information given, ask any questions, and consult any others if they wish to do so. The consent discussion should take place outside of the immediate area of the procedure, preferably on the hospital ward or clinic room. Effective communication is key in giving the patient the correct and relevant information to make a decision. Written leaflets can assist in clear communication of pertinent facts. Consent should ideally be obtained by the person performing the procedure.
Emergency Procedures and Patients Without Capacity to Consent
There are scenarios in which consent by the patient is not possible. First, cases where emergency or life-saving treatment is required, but the patient is incapacitated; for example, being unconscious. In such cases, reasons for the treatment and results should be clearly explained to the patient after the treatment is completed. Second, in patients who are not competent to give consent because of severe mental health conditions or in cases in which the patient is unable to understand or retain the information due to memory conditions such as dementia. In these scenarios, the patient’s best interests should be discussed among relevant clinical teams and the treatment discussed as much as possible with the patient and their immediate family. The refusal of treatment, even in cases apparently unwise or clearly detrimental to the patient, should not affect assessment of a patient’s capacity to consent. In fact, adults should be assumed to be able to give consent unless proved otherwise.
Verbal and Written Consent
Consent may be verbal or written. The clinician’s judgment and convention dictate which of these should be obtained and it is usually the case that written consent is obtained where the procedure is complex or carries significant risk or side effects, and when the procedure is part of a research program. In small procedures such as nasal feeding tube insertions and venograms, verbal consent is usually sufficient.
Patient Optimization
Fasting
Impaired upper airway control after administration of sedatives and analgesics predisposes to aspiration of gastric contents. Thus, fasting is recommended for all nonurgent procedures in which moderate sedation is planned or is likely to be administered ( Table 2.2 ). In urgent and emergent cases, the potential risk for aspiration must be taken into consideration to determine the target level of sedation, timing of the procedure, and airway protection by intubation. Prokinetics (e.g., metoclopramide 10 mg intravenous [IV]) and an H2 blocker (e.g., 10 mg famotidine IV) may be administered to stimulate gastric emptying and reduce the likelihood of aspiration. Similarly, medical conditions in which gastric emptying is delayed or gastric reflux occurs (i.e., pregnancy, diabetic gastroparesis, gastroesophageal reflux disease, hiatal hernia, gastric surgery, peritonitis, and trauma) warrant special consideration in terms of duration of fasting, need for prokinetics/H2 blockers, and low threshold for airway protection.