MR imaging plays an important role in the detection and characterization of several pediatric disease entities that can occur in the emergent setting because of its cross-sectional imaging capability, lack of ionizing radiation exposure, and superior soft tissue contrast. In the age of as low as reasonably achievable, these advantages have made MR imaging an increasingly preferred modality for diagnostic evaluations even in time-sensitive settings. In this article, the authors discuss the current indications, techniques, and clinical applications of MR imaging in the evaluation of pediatric emergencies.
Key points
- •
MR imaging is valuable in the confirmation and characterization of emergent pediatric disorders because of its ease of performance, superior soft tissue contrast, multiplanar capabilities, lack of ionizing radiation, and ability to detect multiple lesions with minimal patient manipulation.
- •
Emergent MR imaging can be critical when clinical, laboratory, and ultrasound or computed tomography findings are nonspecific.
- •
Recognition of some of the characteristic imaging features of emergent pediatric disorders is important because it can allow rapid institution of therapy, which reduces morbidity and mortality.
- •
MR imaging can be an effective and efficient method for the timely and appropriate management of pediatric disease entities that can occur in the emergent setting.
Introduction
MR imaging in the pediatric population can be difficult to obtain because of noise, fear, claustrophobia, and inability to remain motionless. Sedation or general anesthesia is often required to obtain quality images. Despite these limitations, the uses of pediatric MR imaging have expanded over time to include a growing number of emergency indications. This shift is a reflection of the intrinsic advantages of MR imaging in depicting soft tissue disease processes and bone marrow edema as well as the heightened awareness of radiation exposure associated with alternative work-up strategies that use computed tomography (CT). Additionally, rapid MR imaging protocols have reduced the length of MR imaging examinations while still obtaining clinically usable information. The use of single-shot fast spin-echo (SSFSE) MR imaging, for example, has been shown to be useful in the evaluation of ventriculostomy shunt malfunction in children treated for hydrocephalus. Similarly, rapid protocols can also be used to diagnose appendicitis without the need for CT imaging. In this article, the authors discuss a variety of pediatric emergencies that can be diagnosed with MR imaging and review the clinical features that are specific for each disease entity.
Introduction
MR imaging in the pediatric population can be difficult to obtain because of noise, fear, claustrophobia, and inability to remain motionless. Sedation or general anesthesia is often required to obtain quality images. Despite these limitations, the uses of pediatric MR imaging have expanded over time to include a growing number of emergency indications. This shift is a reflection of the intrinsic advantages of MR imaging in depicting soft tissue disease processes and bone marrow edema as well as the heightened awareness of radiation exposure associated with alternative work-up strategies that use computed tomography (CT). Additionally, rapid MR imaging protocols have reduced the length of MR imaging examinations while still obtaining clinically usable information. The use of single-shot fast spin-echo (SSFSE) MR imaging, for example, has been shown to be useful in the evaluation of ventriculostomy shunt malfunction in children treated for hydrocephalus. Similarly, rapid protocols can also be used to diagnose appendicitis without the need for CT imaging. In this article, the authors discuss a variety of pediatric emergencies that can be diagnosed with MR imaging and review the clinical features that are specific for each disease entity.
Imaging technique
Pediatric magnetic resonance (MR) evaluation in the emergent setting is typically performed on a 1.5T or 3T MR scanner with patients supine, using a phased array receiver coil. Higher field strength imaging on a 3T MR scanner offers several benefits for pediatric imaging, including improved signal-to-noise ratio (SNR) and contrast-to-noise ratio, with potential for improved spatial and temporal resolution. However, 3T MR imaging also has disadvantages that can be particularly challenging for abdominal imaging, including increased susceptibility artifacts from air within bowel loops as well as increased energy deposition, which can approach patients’ specific absorbed ratio limits in children. Nevertheless, the current trend in academic centers in the United States is toward higher-field-strength pediatric MR imaging.
The receiver coil used for pediatric MR depends on the body part being imaged. For brain imaging, a 32-channel head coil is typically used at the authors’ institution. For spine imaging, whole-body coils that include coil elements embedded in the scan table are used. The receiver coil used in pediatric musculoskeletal imaging also depends on the body part being imaged, with either a flex coil or a whole-body coil in combination with a body matrix coil used. The receiver coil for pediatric abdominal imaging should fit snugly around patients to maximize spatial resolution and SNR. This fit can be difficult, as various adult MR coils need to be fitted to pediatric patients ranging from infants to adolescents. A head coil is often used for infants and small children, whereas a body coil is typically used for larger children and adolescents. Whole-body coils have also been used with success.
Compared with adult imaging, patient motion is a more substantial problem with pediatric MR imaging. Sources of motion include voluntary motion from patients’ muscular movements in the scanner as well as involuntary motion predominantly due to respiratory motion in children unable to suspend respiration on command. In order to minimize scan times, parallel and fast imaging techniques can be used. An additional consideration unique to children is the prevalence of orthodontic hardware, the presence of which can render susceptibility-weighted imaging (SWI) and diffusion-weighted imaging (DWI) nondiagnostic. Therefore, removal of braces should be considered in cases whereby these sequences are vital (eg, acute stroke).
Patient preparation
Patients older than 6 years are usually able to cooperate with MR imaging after an explanation of the procedure and reassurance. Distraction techniques including the use of MR imaging–compatible music and video players as well as scanning during off hours to minimize ambient noise and activity can also be helpful. Similarly, newborns and young infants may tolerate MR imaging without the need for sedation, if they are well fed and comfortably swaddled. For young children less than 6 years of age, conscious or deep sedation may be required to relieve patient anxiety and minimize patient motion during imaging.
Sedation
Several sedation medications are currently used for pediatric MR imaging, including chloral hydrate, pentobarbital, propofol, and midazolam. Advantages of sedation in children who cannot tolerate MR imaging while awake include reduction in scan time and improvement in image quality. Generally, the least amount of sedation necessary for patients to tolerate MR imaging is administered, both to minimize post-MR imaging side effects and to facilitate patient induction and emergence from sedation. As always, the proper balance should be maintained between adequate sedation for patient comfort and scan performance and minimization of potential neurologic and cognitive effects associated with prolonged anesthesia.
Intravenous Contrast Administration
Contrast agents used for clinical pediatric MR imaging are gadolinium chelated, extracellular contrast agents that cause T1 shortening within blood vessels and perfused tissues. The typical dose for intravenous administration is 0.1 mmol/kg.
Imaging sequences
Brain
A minimal MR imaging examination of the brain (termed brain screen at the authors’ institution) includes sagittal T1, axial T2 fluid-attenuated inversion recovery (FLAIR), axial T2 fast spin echo, coronal T2, and axial DWI. Additional sequences are obtained based on the clinical indication ( Table 1 ). When the clinical question is limited to change in ventricle size for chronically shunted patients, SSFSE imaging is adequate for this evaluation, requires only a few minutes of scanner time, and can typically be performed without sedation in the authors’ experience ( Fig. 1 ).
| Indication | Sequences |
|---|---|
| Hydrocephalus | Axial SSFSE |
| Stroke |
|
| Sinus thrombosis |
|
| Encephalitis and meningitis Cerebral abscess/empyema |
|
| New intracranial neoplasm |
|
Spine
The MR imaging sequences for the evaluation of pediatric spine emergencies also depend on the disease process, whether it is infectious/inflammatory, neoplastic, or traumatic in cause ( Table 2 ).
| Indication | Sequences |
|---|---|
| Discitis/osteomyelitis |
|
| Transverse myelitis/acute neurologic abnormality |
|
| New primary tumor |
|
| Metastatic disease |
|
| Trauma |
|
Body
Septic arthritis, osteomyelitis, and appendicitis are the 3 most common clinical indications for emergent body MR imaging in pediatric patients. In the case of acute hip pain, the following sequences are obtained for evaluation of septic arthritis and/or osteomyelitis: coronal T1, coronal fast spin-echo inversion recovery (FSEIR), sagittal oblique proton density with fat saturation, axial T2 with fat saturation, and postcontrast axial and coronal T1 with fat saturation. The main MR imaging sequences for the evaluation of osteomyelitis in other regions of the body include coronal T1, axial T2 with fat saturation, sagittal FSEIR, postcontrast axial T1 with fat saturation, and postcontrast coronal/sagittal T1 with fat saturation ( Table 3 ).
| Indication | Sequences |
|---|---|
| Septic arthritis (hip) |
|
| Osteomyelitis |
|
| Appendicitis |
|
Appendix MR imaging sequences are acquired from the level of the kidneys through the urinary bladder without sedation or contrast media. Although there is variability in the published literature about the necessary sequences, most institutions include multiplanar T2-weighted single-shot turbo spin-echo (ie, half-Fourier acquisition single-shot turbo spin-echo) without fat saturation and at least a single-plane short-tau inversion recovery (STIR) sequence. At the authors’ institution, additional sequences that have been considered useful include a coronal T2-weighted 3-dimensional (3D) turbo spin-echo (TSE) sequence with multiplanar reconstructions and/or axial true fast imaging with steady-state precession. All sequences except 3D TSE are obtained with breath holding, whereas 3D TSE is acquired during shallow free breathing (see Table 3 ).
Spectrum of disorders
Vasculopathic Disorders
A stroke is a syndrome of spontaneous, prolonged neurologic dysfunction secondary to brain injury. Like adults, childhood strokes can be divided into those due to insufficient arterial inflow (arterial ischemic stroke [AIS]) and those due to thrombosis of the venous outflow (cerebral venous thrombosis [CVT]). Although hemorrhagic strokes are more common in children than adults, composing up to 30% to 50% by some estimates, the immediate care of these patients often revolves around managing mass effect from the hematoma or hydrocephalus from decompression into the ventricular system, something usually done adequately and with maximal efficiency with head CT. Therefore, it is not further discussed in detail here.
Arterial ischemic stroke
The incidence of AIS in children has been estimated in the range of 0.2 to 7.8 cases per 100,000 children per year, with neonates having roughly 10-fold higher incidence. Well-known risk factors include underlying congenital heart disease and sickle cell anemia, the latter increasing the risk of an ischemic event by roughly a factor of 100 times. Most children presenting with AIS are previously well, and up to 30% to 50% of AIS cases have been historically thought to lack an identifiable risk factor. However, the increasing recognition of children with some kind of risk factor, such as an arteriopathy (eg, moyamoya, spontaneous intracranial dissection), prothrombotic state (eg, hyperhomocysteinemia), or recent infection, have reduced the number of cases with no known cause.
Unlike adults, significant numbers of pediatric patients with AIS present with seizure. This feature is particularly striking in neonates whereby seizures may affect up to 40% of newborns with territorial infarcts. These newborns may also have vague findings, such as generalized encephalopathy or abnormalities of tone. In older children, focal neurologic symptoms are more common; but the clinical picture may be obfuscated by nonspecific symptoms, such as headache or altered mental status. These atypical presentations may be a contributor to considerable delays in diagnosis of pediatric AIS and also represent an impediment to investigation of stroke interventions.
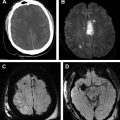
In the authors’ institution, emergent evaluation of a potential AIS is generally sought using brain MR imaging, MR angiography (MRA) (brain and neck), and in many cases MR venography (MRV) because this combined MR work-up provides the quickest means of unambiguously differentiating AIS from alternative diagnoses, such as CVT, posterior reversible encephalopathy syndrome, or acute disseminated encephalomyelitis (ADEM). Imaging typically begins with diffusion imaging early in the examination to detect sites of infarction should patients prove too unstable or uncooperative to continue imaging. As is the case for adults, diffusion restriction is visible within minutes of thromboembolic compromise of inflow, and T2 signal abnormality develops after the first 6 to 8 hours with peak mass effect anticipated a few days after the onset of infarction. The vascular imaging typically allows for definition of the level of inflow compromise ( Fig. 2 ); special sequences, such as fat-saturated or vessel wall imaging, may be used in certain circumstances, such as suspected dissection. SWI is a useful adjunct to diffusion imaging as it can detect areas of petechial hemorrhage, intraluminal clot, and areas of increased oxygen extraction from decreased blood flow ( Fig. 3 ). In the dictation, special comment should be made on the general geographic extent of the infarction and involvement of critical structures, such as the corticospinal tract, because these imaging markers correlate with long-term disability.


Therapy for AIS begins with correction of any exacerbating conditions, such as decompensated cardiac function or prothrombotic states. Investigation for predisposing risk factors, such as prothrombotic conditions, is usually undertaken unless a known risk factor accounts for the stroke. Additional therapies then depend on underlying causes of the stroke. For arterial dissection, therapeutic anticoagulation with heparin/warfarin is generally begun in patients with infarction, though anticoagulation for intracranial dissections is more controversial. For patients with sickle cell, transfusions are usually performed to optimize oxygen carrying capacity. At this time, thrombolytic agents are considered experimental in children, though clinical trials are currently evaluating their use. Aspirin is often used as a medication for secondary prevention, but special considerations are required for minimizing the risk of Reye syndrome. The therapies available for AIS as well as expected outcomes for specific causes of AIS are comprehensively reviewed in the American Heart Association’s (AHA) scientific statement on pediatric stroke.
Cerebral venous thrombosis
CVT is abnormal clot formation within the cortical veins, deep cerebral veins, or the dural sinuses. When of hemodynamically significant size or occlusive, these clots cause venous hypertension, which can result in congestion of the drained brain parenchyma, in some cases manifesting as venous ischemia or hemorrhage (up to 40%). CVT is estimated to be twice as common in children as in adults, with an estimated incidence of slightly less than 1 case per 100,000 children per year; neonates represent the most susceptible population accounting for more than 25% of all affected children ascertained in the largest study of childhood CVT. Myriad causes may trigger CVT episodes, including prothrombotic states (eg, malignancy; nephrotic syndrome; lupus anticoagulant; medications, such as l -asparaginase or oral contraceptives), dehydration, and underlying infection/inflammation (eg, inflammatory bowel disease or complicated otomastoiditis). Although focal neurologic deficits or headaches often accompany CVT, the clinical picture may be confusing in very young patients whereby altered sensorium or seizure may be the primary manifestation. Additionally, more insidious symptoms, such as elevated intracranial pressure, communicating hydrocephalus, or papilledema (ie, vision loss), can be the primary symptom in some patients.
Therefore, imaging is often pivotal in correctly identifying the cause of nonspecific symptoms, such as headache, seizure, or altered mental status; the appropriateness of MRV in infants should always be considered as part of the evaluation for unexplained seizure, and scrutiny of the venous flow voids is required when MR imaging for headache has no other positive findings. In the authors’ institution, MR imaging and MRV are the preferred means of evaluating and following CVT because of the absence of radiation and the ability to characterize parenchymal complications in detail as well as evaluate the cerebral venous system. Clot within the cerebral sinuses evolves in a manner paralleling parenchymal hematomas, being initially T1 isointense and T2 hypointense followed by evolution to more T1/T2 hyperintense signal over a course of a few weeks. Deoxygenated hemoglobin from clot is most conspicuous on SWI or T2* gradient echo (GRE) sequences ( Fig. 4 ). MRV can be performed using the time-of-flight technique (T1 hyperintense clot can spuriously appear as flow due to the T1 weighting of this sequence, and in-plane flow can falsely simulate clot) or phase contrast technique (artifacts related to aliasing or flow below the velocity encoding can occur). Postcontrast 3D gradient echo sequences, such as MPRAGE or SPGR, circumvent many of the technical limitations of noncontrast MRV sequences and offer isotropic resolution, often enabling more confident determination of the extent/occlusiveness of a thrombus. It should be noted that isolated cortical vein thrombosis is frequently best seen on susceptibility and conventional noncontrast 3D gradient echo sequences. These technical details have been recently reviewed elsewhere.
Treatment of CVT in children begins with management of systemic contributors to the thrombosis, including cessation of offending medications, rehydration, and treatment of inciting infections (eg, mastoidectomy and antibiotics for complicated sinusitis). However, the use of anticoagulation is more controversial because of the frequent co-occurrence of intracranial hemorrhage with CVT and lack of age-specific randomized controlled trials. Based on a large case series, however, the AHA’s Stroke Council endorses the use of therapeutic anticoagulation for CVT experienced in children outside the neonatal age group. For neonates, the AHA does not recommend use of heparin/warfarin unless there is radiographic evidence of thrombus propagation or multiple thrombosed dural sinuses. However, there is considerable heterogeneity in actual practice patterns regarding neonatal CVT treatment.
Posterior reversible encephalopathy syndrome
Posterior reversible encephalopathy syndrome (PRES) merits consideration in a discussion of pediatric stroke because it is also a disorder that may present with headaches, altered mental status, and seizure. Although studies of children with PRES are scarce, the risk factors seem similar to those for adult PRES and include use of immunosuppressive medication (eg, tacrolimus, cyclosporine), some chemotherapeutic agents, and hypertensive crisis. However, there is some evidence that these children may lack the severity of hypertension common to adults presenting with this diagnosis. Regardless of cause, the treatment of PRES is generally to remove the inciting cause.
Although frequently subtle on CT examinations, the MR imaging manifestations of PRES are usually quite obvious. Typical features are areas of subcortical and cortical signal abnormality within the perfusion watershed ( Fig. 5 ) with occasional involvement of deeper structures, such as the deep white matter, deep gray matter, brainstem, and cerebellum. Small areas of diffusion restriction or hemorrhage occur in a minority of cases and probably account for the rare cases where imaging findings are not truly reversible. It has been suggested that pediatric PRES may present in a more florid manner than typical adult PRES, but many adult cases of PRES have been reported with similarly extensive findings.
Infectious and inflammatory disorders
Brain
Meningitis
Meningitis is inflammation of the leptomeninges by a virus (most commonly enteroviruses), bacterium, autoimmunity, or drug/chemical exposure. In an emergency setting, bacterial meningitis is the most feared type of meningitis because of mortality rates approaching 30% to 50% in some studies and significant morbidity in those who survive. It is also frequently a difficult diagnosis for the referring physician, reportedly representing the most common cause of malpractice litigation among pediatric emergency department physicians: symptoms are neither sensitive nor specific, featuring headache, fever, seizure, and mental status changes seen with many of the other infectious disorders discussed in this section. The incidence of bacterial meningitis among children of all ages is reported as 1 to 3 per 100,000 individuals per year, but this frequency can be 2 orders of magnitude higher among newborns and in some developing countries. The most common neonatal causes of meningitis are group B streptococcus ( Streptococcus agalactiae ), gram-negative rods, such as Escherichia coli , and Listeria monocytogenes . The most common causes of bacterial meningitis among older children are bacteria known to colonize the upper respiratory tract, including Haemophilus influenza type b, Streptococcus pneumonia , and Neisseria meningitidis , though the epidemiology is evolving because of vaccination campaigns against these organisms. Complicated sinusitis/otomastoiditis are also important though infrequent causes of meningitis in older children.
Cerebrospinal fluid (CSF) sampling is critical in confirming an acute infection, suggesting a bacterial cause, and eventually identifying a specific organism by CSF culture. Such an analysis is recommended in febrile seizure patients with additional features, such as encephalopathy or without immunization against the major meningitic bacteria, H influenza type b and S pneumonia . Although antibiotic therapy before lumbar puncture can diminish the yield from CSF culture, the typical bacterial meningitis CSF pattern can still be observed: high protein, low glucose, and polymorphonuclear cell-predominant white blood cells. Once the diagnosis of bacterial meningitis is secured, broad-spectrum antibiotics are continued until culture identifies a specific agent and its sensitivities; steroids are occasionally also used.
Imaging is performed to exclude parenchymal complications (ie, infarction, abscess), empyema, ventriculitis, or hydrocephalus from bacterial meningitis rather than to confirm the diagnosis itself. Although some articles have suggested remarkable sensitivity for diagnosing meningitis by imaging alone, expert opinion is that imaging remains much less sensitive in detecting meningeal inflammation than CSF sampling. MR imaging with diffusion imaging provides the most sensitive evaluation for parenchymal changes, allowing early detection of areas of microinfarction from meningitis; DWI/FLAIR allows differentiation of proteinaceous exudates from purulent material ( Fig. 6 ). MR imaging can also be used to follow disturbances of CSF resorption (ie, hydrocephalus) that ensue in many cases of bacterial meningitis. In cases of tuberculous meningitis, areas of leptomeningeal nodularity, thick basilar exudates, and parenchymal tuberculomas may suggest this specific diagnosis. As detailed later, MR imaging has additional advantages in definitively characterizing parenchymal abscess versus cerebritis and effusions from empyema. Should there be concern regarding arterial or venous occlusion in the setting of meningitis, MRA and MRV can be added on during the examination with minimal prolongation in total examination time.
Encephalitis and acute disseminated encephalomyelitis
Encephalitis is a condition of parenchymal inflammation due to an infectious agent other than pyogenic bacteria or due to autoimmune inflammation. Although relatively uncommon in the population as a whole, encephalitis is relatively common in children, particularly young children, with quoted incidences of 10.5 and 27.7 cases per 100,000 children yearly for all children and infants, respectively. The list of potential causes is lengthy and includes viruses (respiratory, such as influenza A; enteroviruses; arboviruses, such as West Nile; and herpes viruses, such as herpes simplex virus type 1 [HSV-1], varicella zoster, and Epstein-Barr viruses) and nonpyogenic bacteria, such as Mycoplasma , Bartonella , and Borrelia species. Depending on the case series, a significant minority or most cases will ultimately remain without a defined infectious agent as the cause. Of interest, the California Encephalitis Project suggests that Mycoplasma pneumoniae –associated encephalitis may represent the single most common infectious cause of childhood encephalitis and autoimmune (ie, anti–N-methyl-d-aspartate receptor) encephalitis may represent the single most common noninfectious cause excluding ADEM. ADEM is a monophasic demyelinating event associated with recent vaccination or infection, most commonly seen in children whereby the incidence is up to 0.8 cases per 100,000 individuals per year.
The encephalitides and ADEM present with fever, headache, other symptoms of elevated intracranial pressure (nausea, vomiting), changes in mental status, and seizure making it difficult to distinguish the different encephalitides among each other and from other possibilities, such as meningitis. However, neuropsychiatric manifestations and movement disorders are a much more pronounced feature of autoimmune encephalitis, with elevations in temperature usually occurring in the setting of autonomic instability. Also, ADEM symptoms are reported to have more subacute onset than infectious encephalitis, over a couple of days. Laboratory analysis (serum and CSF) is a critical component of an encephalitis work-up and typically reveals CSF pleocytosis with a mononuclear/lymphocyte predominance. ADEM CSF studies usually show normal or more modest levels of CSF pleocytosis and may have transient oligoclonal immunoglobulin G detected.
MR imaging is the most sensitive imaging modality for detecting encephalitis, in the case of HSV-1 being positive in more than 90% of cases. The advantages of MR imaging are particularly striking with the use of diffusion imaging, which can detect areas of nascent cytotoxic injury before the onset of florid parenchymal edema. In most cases, a specific viral agent cannot be suggested based on the imaging pattern alone. One exception is reactivation pattern HSV-1 infection, which classically involves one or both temporal lobes with associated diffusion restriction and hemorrhage ( Fig. 7 ). In this regard, the radiologist may play an important role in diagnosis because polymerase chain reaction (PCR) of CSF from early HSV-1 encephalitis may occasionally be falsely negative. This pattern stands in contrast to the neonatal, HSV-2 encephalitis pattern, which is less spatially restricted and frequently involves areas outside the limbic system (eg, deep gray matter, brainstem, cerebellum). There is a propensity for deep gray matter structures by the respiratory viruses and some arboviruses, such as West Nile. The pattern of involvement may also suggest a noninfectious cause because ADEM typically has less mass effect, smaller lesions, and a propensity for subcortical/deep white matter. MR imaging of the spine also commonly reveals additional lesions in up to 28% of ADEM cases. However, ADEM also commonly involves the deep gray matter as well as the deep white matter; hyperacute lesions may restrict diffusion, raising uncertainty about the diagnosis.
When encephalitis is clinically suspected, it is standard to administer acyclovir until HSV PCR has returned negative with administration of antibiotics for indeterminate CSF cell count (ie, possible bacterial meningitis) or suspected exposure to atypical bacteria (eg, doxycycline for suspected tick exposure). This practice reflects the fact that therapy for most encephalitides besides herpes viridae (mainly HSV-1 and HSV-2) are only supportive at the present time. Should the clinical history, CSF analysis, and imaging support a diagnosis of ADEM instead, steroids are commonly given because there is evidence of reduced duration of symptoms and possibly better outcomes with immunosuppression.
Cerebritis and cerebral abscess
Cerebritis is an infection of the brain parenchyma by pyogenic bacteria and, left untreated, will result in progressive liquefaction/necrosis of the involved brain tissue until surrounded by a capsule that indicates an organized abscess. The incidence of cerebral abscess is difficult to estimate. However, it is clearly quite rare with case series suggesting incidence in the range of 1 case per million inhabitants per year. Risk factors for cerebritis and cerebral abscess include cyanotic congenital heart disease, meningitis, trauma, and intracranial complications from otomastoiditis or sinusitis; making allowances for roughly one-third of cases with polymicrobial or nongrowing cultures, gram-positive cocci seem to be the most common single infectious agents. Demographic risk factors include male sex and age in the early first or second decade. It should be noted that intracranial complications from bacterial sinusitis are estimated to be 4%, and abscess is less common than empyema (see later discussion). Although abscess is also an unusual complication of meningitis, certain organisms are known to have a high propensity for this complication; neonatal Citrobacter meningitis can be complicated by abscess in greater than 70% of cases. Symptoms of abscess include headache (particularly older children), irritability (particularly young children or infants), altered mental status, and seizure. However, fever is present in only half of cases, and the white blood cell count may be normal.
Cerebritis and abscess have been studied experimentally and with radiologic-pathologic correlation, demonstrating evolution through 4 discrete stages (beginning with early cerebritis and ending with an encapsulated abscess) over a period of 2 weeks. The imaging abnormalities recapitulate these stages with ill-defined T2 signal abnormality and absence of enhancement in early cerebritis, followed by incomplete peripheral enhancement in late cerebritis. By the time an organized abscess has formed, a well-defined rim of T2 hypointensity and enhancement (usually thinner on the ventricular side of the abscess) can be defined with a center of restricted diffusion ( Fig. 8 ). As expected, extensive edema surrounds the abscess and manifests as facilitated diffusion.