Chapter 4 Pulmonary Diseases in the Immunocompromised Host with or without Acquired Immunodeficiency Syndrome
PULMONARY INFECTIONS IN THE IMMUNOCOMPROMISED PATIENT WITHOUT HUMAN IMMUNODEFICIENCY VIRUS INFECTION
The lung is a frequent target of infection in the immunocompromised host, and mortality rates associated with pulmonary disease often are as high as 40% to 50%. Infection is the most frequent cause of the radiographic abnormality; however, conditions such as extension of malignancy (e.g., lymphoma, metastases), drug reactions, or other noninfectious processes may be diagnostic possibilities (Table 4-1). Unfortunately, noninvasive diagnostic methods such as sputum smears and cultures that are used for evaluation of pneumonia are less useful in the immunocompromised host. The clinician must therefore choose between using invasive techniques to determine the exact cause of the pneumonia or employing empirically chosen therapy. The former choice is not without hazard in debilitated patients; the latter is complicated by the broad range of possible causes and appropriate therapies.
Table 4-1 Radiographic Patterns in HIV-Negative Immunocompromised Patients with Pulmonary Disease
| Lobar or Segmental Consolidation | Nodules with Rapid Growth ± Cavitation | Diffuse Lung Disease |
|---|---|---|
| Causes | Causes | Causes |
| Radiographic features | Aspergillus | Pneumocystis jiroveci pneumonia |
| Diagnosis | Mucormycosis | Cytomegalovirus |
| Differential diagnosis | Diagnosis | |
| Differential diagnosis |
BAL, bronchoalveolar lavage; TBB, transbronchial biopsy; TNB, transthoracic needle biopsy.
Radiologic Patterns
Table 4-1 summarizes the radiographic patterns for HIV-negative immunocompromised patients with pulmonary disease.
Lobar or Segmental Consolidation: Bacterial Pneumonia
Causes
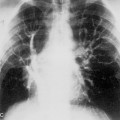
The Legionnaires’ disease bacterium (Legionella pneumophila) and Pittsburgh pneumonia agent are causes of acute bronchopneumonia in the immunocompromised host, particularly in renal transplant recipients. The clinical and radiographic appearance of Legionella pneumophila in these patients is usually identical to that seen in the normal host. Multilobar consolidation is common. However, the Pittsburgh agent (Legionella micdadei) typically produces circumscribed areas of pneumonia, creating a nodular appearance on the chest radiograph (Fig. 4-1).
Nodules with Rapid Growth or Cavitation: Fungal Pneumonias
Causes
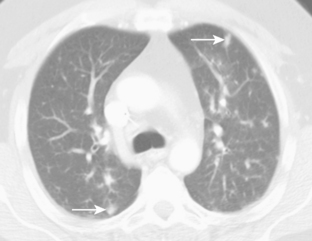
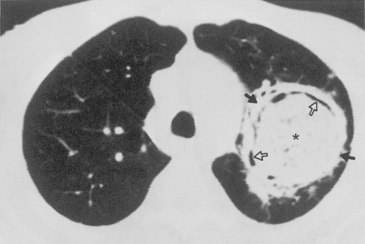
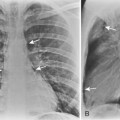
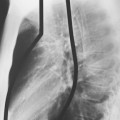
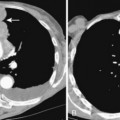
Aspergillus pneumonia is the most common fungal pulmonary infection in immunosuppressed patients. Aspergillus causes an invasive necrotizing pneumonia resulting from invasion of blood vessels with accompanying pulmonary infarction. The roentgenographic features consist of multiple nodular areas of consolidation that often abut the pleural surfaces (Fig. 4-2). These areas frequently cavitate and may show crescentic radiolucencies around the parenchymal opacities (i.e., air crescent sign) that may mimic mycetoma. This sign also can be identified on CT studies (Fig. 4-3). Another characteristic finding is a pulmonary mass surrounded by a zone of lower attenuation with ground-glass opacification (i.e., halo sign), probably produced by adjacent hemorrhage. The diagnosis of Aspergillus pneumonia usually requires invasive procedures, such as needle aspiration or open lung biopsy.
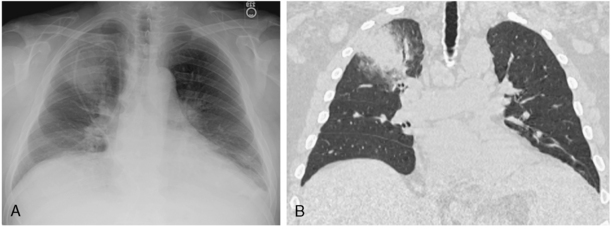
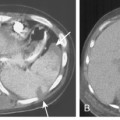
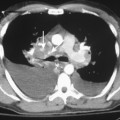
Pulmonary disease caused by Mucor is clinically and radiographically indistinguishable from that caused by Aspergillus. It is frequently seen in patients with lymphoproliferative disease, leukopenia, and diabetes, and it is detected in patients after antibiotic use. The organism likewise has a predilection for blood vessel invasion and pulmonary infarction (Fig. 4-4).
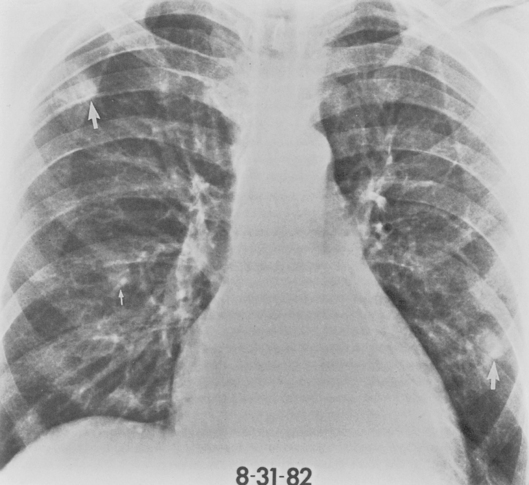
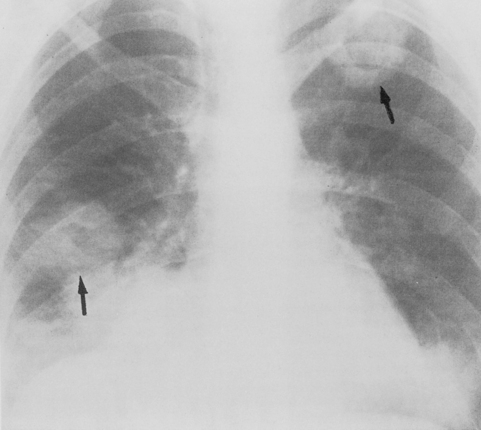
Nocardia asteroides is an opportunistic bacterium that causes pneumonia in the compromised host with diseases or conditions in which cellular immunity is depressed. Antecedent corticosteroid therapy is a common history, but white blood cell counts are often normal. Nocardia usually does not cause a fulminant, rapidly progressive pulmonary infection. The usual radiologic appearance is that of single or multiple nodules with or without cavitation (Fig. 4-5). They may extend to the pleural surface with associated pleural effusion or chest-wall invasion. The diagnosis can occasionally be made on sputum smears or cultures, but invasive procedures are usually required.
Diffuse Lung Disease
Causes
The prevalence of pneumonia caused by Pneumocystis jiroveci has decreased because of widespread prophylaxis in immunocompromised patients with or without HIV infection, but it remains a major cause of morbidity and mortality. Once classified as a protozoan, the organism is now considered to be a fungus. The pneumonia it produces is acute and fulminating. The classic radiographic manifestation is initially a bilateral, perihilar or diffuse, symmetric interstitial pattern, which may have a finely granular, reticular, or ground-glass appearance. If left untreated, it may progress over 3 to 5 days to a homogeneous, diffuse alveolar consolidation (Fig. 4-6). Hilar adenopathy and pleural effusion are distinctly unusual. Computed tomography (CT), particularly high-resolution CT (HRCT), may show areas of disease due to Pneumocystis pneumonia when the appearance on a standard chest radiograph is normal. Involved areas of the lung show ground-glass opacification without obliteration of normal pulmonary vessels. Because it is not possible to culture these organisms, the diagnosis depends on morphologic identification of Pneumocystis
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree