Outline
Safety, 749
Technique, 750
T2-Weighted Imaging, 750
T1-Weighted Imaging, 750
Balanced Steady-State Free Precession, 750
Diffusion-Weighted Imaging, 751
Other Magnetic Resonance Acquisitions, 751
Maternal Imaging, 751
Fetal Imaging, 752
Anomalies of the Central Nervous System, 752
Anomalies of the Face, Neck, and Thorax, 755
Abnormalities in the Fetal Abdomen, 762
Tumors, 763
Monochorionic Twins, 765
Abnormal Placentation, 765
Emerging Concepts, 768
Conclusions, 769
Summary of Key Points
- •
Magnetic resonance imaging (MRI) does not use ionizing radiation, and no known biologic risks are associated with this imaging modality.
- •
MRI is an important tool for the evaluation of maternal complications in pregnancy, including the evaluation of atypical advanced ectopic pregnancies, suspected appendicitis, pelvic masses, hepatic and genitourinary diseases, and placenta accreta.
- •
In the fetus, MRI is a useful adjunct to the sonographic diagnosis of suspected central nervous system (CNS) lesions, particularly isolated mild ventriculomegaly.
- •
MRI may provide additional information for certain fetal thoracic lesions, can aid in predicting prognosis in diaphragmatic hernias, and can provide additional information in the setting of complex fetal genitourinary abnormalities.
- •
MRI studies add important information for the potential fetal surgery candidate and may direct crucial decisions for delivery management, as with the ex utero intrapartum treatment (EXIT) procedure.
- •
MRI has been found to be useful in the diagnosis of abnormal placental implantation in cases with an equivocal diagnosis on ultrasound imaging.
- •
Advances in MRI technology continue to allow for better resolution of fetal structures, and it is likely that this technique will have increasing application in prenatal diagnosis and evaluation of fetal abnormalities.
The role of MRI in managing the obstetric patient is increasingly expanding. Although sonography is the primary imaging modality for evaluation of the pregnant patient, MRI has been in use for the past 30 years and is now considered an established adjunct imaging modality for obstetric management. Maternal obesity, oligohydramnios, fetal positioning, and fetal ossification can hamper ultrasound evaluation. Advantages of MRI include lack of ionizing radiation, superior soft tissue contrast, wide field of view imaging, and image acquisition in multiple true orthogonal planes. Various sequences allow us to discern soft tissue, fluid, hemorrhage, fat, and meconium. Additional sequences allow us to identify regions of hypoxia/ischemia. Limitations of MRI include maternal anxiety and claustrophobia, maternal habitus, and fetal motion when evaluating for anomalies. In this chapter we address the utility of MRI as an adjunctive second-line imaging modality in evaluating the symptomatic pregnant patient, the fetus, and the abnormally adherent placenta.
Safety
No known biologic risks are associated with MRI. No delayed sequelae from MRI examination have been encountered, and it is expected that the potential risk for any such delayed sequelae is extremely small or nonexistent. MRI utilizes no ionizing radiation, although concerns include exposure to fluctuating electromagnetic fields, high sound intensity levels, and effects on fetal heart rate patterns during the procedure. Several animal and tissue studies have been conducted to determine the biologic effects of electromagnetic fields with conflicting reports. An early study looked at the long-term effects of repetitive exposure to a static 1.5-tesla (T) magnetic field on human lung fibroblasts. Study and control group proliferations were similar, indicating no adverse effect from repetitive MR exposure.
Few human studies on risks of MRI and early development have been completed. A large epidemiologic retrospective study compared spontaneous abortion rates, infertility rates, incidence of low birth weight, and premature delivery among nurses and technologists working with MRI before and after employment. There were no increased incidences of adverse outcomes in the MR-exposed group. Two studies reported follow-up on children exposed to echo-planar MR in utero. The first by Baker and associates completed a 3-year follow-up on 20 children with no demonstrable increase in the occurrence of disease or disability. These patients were imaged with a 0.5-T superconductive magnet from 21 weeks to term. In the second case-controlled prospective observational study of 20 infants exposed to echo-planar imaging, pediatric assessments completed on these infants at 9 months of age were normal.
In reference to the sound intensity and exposure, one study assessed the sound level experienced by the fetal ear during an MR procedure by having a volunteer swallow a microphone connected to a thin lead and filling the stomach with a liter of fluid to represent an amniotic sac. There was at least 30-dB attenuation in intensity from the body surface to within the fluid-filled stomach, which reduced the acoustic sound pressure down from the dangerous threshold of 120 dB to an acceptable level of less than 90 dB. This level is lower than the 135 dB experienced when vibroacoustic stimulation is used. No evidence of hearing loss was found in one study after 450 babies were exposed to this noise intensity from vibroacoustic stimulation.
Studies have also looked at fetal heart rate patterns during MR procedures and reported no change in heart rate patterns or incidence of fetal movements. Recommendations from the latest document from the American College of Radiology concerning safe practices for MRI have determined that MRI can be performed in pregnancy if the data are needed to affect the care of the fetus or mother during pregnancy. Because of the potential for dissociation of the chelate molecule in the amniotic fluid, MRI contrast agents should not be routinely administered. The longer the chelate molecule remains in a protected space such as the amniotic sac, the greater the potential for dissociation of the potentially toxic gadolinium ion, which can lead to nephrogenic systemic fibrosis in at-risk adults. Gadolinium is listed as a category C drug in pregnancy based on Food and Drug Administration classifications, implying that it should be administered only if potential benefits warrant use of the drug despite potential risks. It is recommended that women undergoing MRI during pregnancy provide informed written consent documenting that they understand the potential risks and benefits of the procedure.
Technique
Fetal and maternal studies are generally performed on a 1.5-T magnet or less, and it is recommended that informed consent be obtained for all patients in the settings of either institutional review board (IRB)-approved protocols or as a clinically indicated procedure. In addition, all women complete a written MRI safety screening questionnaire including information regarding metallic implants, pacemakers, or other metal- or iron-containing devices that may impact the study. Iron supplementation may rarely cause artifact in the colon but does not usually affect the fetal resolution. Portable devices and monitors must be evaluated using U.S. Food and Drug Administration labeling criteria to prevent ferrous objects from entering unsafe zones within the MRI areas. Anxiolytics are not routinely administered because significant maternal anxiety is a very rare event (less than 1% in our population), but in those unique circumstances, short-acting benzodiazepines may be administered. The recommended weight limit is approximately 400 pounds (but dependent on body habitus) on most closed current MR units; therefore, the extremely obese woman may not be a candidate for MRI.
Women are placed feet first in the supine or left lateral decubitus position. A torso coil is used in most circumstances, with the occasional use of either the body or cardiac coil, depending on maternal size, fetal size, and area of interest. A triplanar balanced steady-state free precession (bSSFP) sequence through the entire maternal uterus is initially obtained for localization purposes and planning subsequent sequences, which have a large field of view (350-400 mm), using 6-mm slice thickness with 3-mm interslice gap.
T2-Weighted Imaging
T2-weighted image acquisitions are utilized to define fetal anatomy and dysmorphologic features that have certain tissue characteristics based on a specific spin echo sequence using relatively long repetition time (TR) and long echo time.
Half-Fourier single shot RARE (rapid acquisition with relaxation enhancement) has become the mainstay of maternal pelvic and fetal imaging because of its high tissue resolution and rapid acquisition times. This ultrafast single shot imaging has an average acquisition time of less than 1 second, depending on imaging parameters. Other names for this sequence include single shot fast spin echo (SSFSE) and half-Fourier acquisition single shot turbo spin echo (HASTE). Specifics of the sequence vary, but for the single shot acquisition one signal and one measurement is acquired, the TR range is 800 to 1100 ms, echo time is 60 to 90 ms, flip angle is 130 to 150 degrees with 0- to 2-mm gap, and field of view is 300 to 350 mm.
Fetal images may be obtained contiguously or interleaved, both having distinct advantages. Fetal movement degrades MR images, and contiguous slices may allow for better visualization of some contiguous structures in some slices, even if movement occurs during other slices in the acquisition. The interleave method allows for improved signal-to-noise ratio by obtaining every other image with an interslice gap and repeating process to include that missed gap; this prevents excitation of adjacent and consecutive sections.
T1-Weighted Imaging
The use of T1-weighted gradient echo sequences provides additional information, owing to very short T1 relaxation times and allowing for improved visualization of subacute blood, fat, and proteinaceous fluid as well as fetal liver and meconium. Hyperintense pituitary and thyroid glands can also be seen. Late in gestation the fetal bones have a hypointense appearance. The images are less sensitive in defining some solid organ anatomy because of less tissue contrast.
In the setting of obstetric imaging, T1-weighted sequences are utilized to assess for the presence and distribution of T1 hyperintense meconium, to determine the size and location of the fetal liver, and to evaluate fetal or maternal hemorrhage or fat-containing lesions. Some T1-weighted sequences require more than one single shot with short TR (range 7.7-112 ms), echo time (4.2-5 ms), and flip angle (130-150 degrees). T1-weighted sequences may be obtained by single shot T1-weighted sequences or three-dimensional (3D) imaging techniques, with or without fat suppression. Because of improved signal-to-noise ratio, breath-hold T1-weighted gradient recalled echo sequences are generally used. T1-weighted breath-hold images are acquired with 5.5-mm slice thickness and 10% (0.5-mm) gap with field of view range of 300 to 350 mm.
Balanced Steady-State Free Precession
bSSFP acquisitions have manufacturer acronyms such as True-FISP and FIESTA (fast imaging with steady-state precession and fast imaging employing steady-state acquisition) and are complex in terms of how they derive their contrast, the result of which is high T2 weighting in fluid and high T1 weighting in tissue. Vessels are displayed quite well, especially when surrounded by dense tissue such as liver parenchyma or the vertebral disk spaces. Tissue contrast tends to be quite good, with slice thickness for bSSFP in the range of 5 to 6 mm and field of view ranging from 260 to 300 mm.
Diffusion-Weighted Imaging
Diffusion-weighted imaging (DWI) is based on echo-planar imaging that produces images of water diffusion; this diffusion of water may be restricted in certain disease states, such as hypoxic edema (ischemia) of the brain. Diffusion anisotropy has the potential to characterize premyelination structures and can identify the corpus callosum and other white matter tracts, which will be discussed in Emerging Concepts.
Other Magnetic Resonance Acquisitions
Many other acquisitions can be applied, some of which will be discussed in Emerging Concepts and are briefly reviewed. Short tau inversion recovery (STIR) images may provide improved resolution of tissue characteristics when the water contents of structures are similar. Additional sequences such as fluid attenuated inversion recovery (FLAIR), hydrography, blood oxygen level–dependent (BOLD) imaging, and certain echo-planar imaging may be performed as needed.
Maternal Imaging
In an effort to image the pregnant patient with abdominal pain rapidly and efficiently, the MRI protocol includes multiplanar T2-weighted SSFSE images (with and without fat saturation) of the abdomen and pelvis, gradient echo sequences, and a single plane T1-weighted sequence.
A wide range of pathologic conditions can cause abdominal pain in the pregnant patient. Clinical diagnosis is often challenging because of nonspecific leukocytosis, compression and displacement of organs by the gravid uterus, and nonspecific nausea and vomiting. It is important to differentiate obstetric from nonobstetric causes of the acute abdomen in an effort to minimize maternal and fetal morbidity and mortality risks. Conditions that necessitate surgical intervention must be identified.
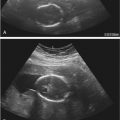
Ectopic pregnancy continues to be the leading obstetric cause of death in early pregnancy. MRI has been shown to be useful in delineating unusual implantation sites, including pregnancies implanted in the interstitium, cervix, cesarean scar, and rudimentary horn of an anomalous uterus, and within the peritoneal cavity ( Fig. 23-1 ). T1-weighted sequences can depict blood products, and the presence of hemoperitoneum and hematosalpinx are highly suggestive of ectopic pregnancy in a patient with a positive pregnancy test and no intrauterine gestational sac. The large field of view and multiplanar capability of MRI allow exact delineation of advanced first trimester and second trimester ectopic pregnancies, which might not be appreciated on ultrasound imaging. Late presentation of ectopic pregnancy can be catastrophic and fatal because of risk of rupture and hemorrhage. The late presentation of an advanced cesarean scar pregnancy that has grown cephalad might mimic a normal intrauterine pregnancy. Demonstration of myometrial tissue surrounding a gestational sac without continuity to the cervix can help discriminate between a pregnancy in a bicornuate uterus versus a rudimentary horn pregnancy.

Acute appendicitis is the most common nonobstetric cause for surgical intervention during pregnancy. Early diagnosis is essential, as surgical delay is associated with increased risk of appendiceal perforation, leading to an increased rate of maternal morbidity and fetal loss ( Fig. 23-2 ). Other gastrointestinal causes of abdominal pain include inflammatory and infectious bowel disease, bowel obstruction, and diverticulitis ( Fig. 23-3 ). Symptoms (abdominal pain, nausea, vomiting) are similar to those that occur with normal pregnancy. Comprehensive cross-sectional imaging is essential for evaluation, and MRI is preferable to computed tomography because of lack of ionizing radiation.


Physiologic dilatation of the urinary tract occurs in up to 90% of pregnant patients. MR urography can differentiate obstructive hydronephrosis caused by renal calculi from physiologic hydronephrosis if renal enlargement, perinephric fluid, and abrupt caliber change of the ureter are demonstrated ( Fig. 23-4 ).

Hepatobiliary causes of abdominal pain include HELLP syndrome (hemolysis, elevated liver enzymes, low platelet count) and acute fatty liver of pregnancy, hepatitis, gallbladder disease, and pancreatitis. MRI and MR cholangiopancreatography can be useful in demonstrating choledocholithiasis and pseudocysts in the patient suspected of having pancreatitis.
Gynecologic causes of pain during pregnancy include ovarian cysts, masses, and torsion and uterine abnormalities, such as leiomyomas and congenital müllerian anomalies. Fluid, solid components, septations, nodularity, edema, hemorrhage, and fat content can be seen on MRI. If a primary pelvic mass is suspicious for malignancy, MRI can depict ascites, peritoneal implants, and lymphadenopathy.
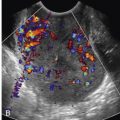
The hypercoagulable state of pregnancy, venous compression by the gravid uterus, and hormone-induced venodilatation put the pregnant patient at increased risk for venous thromboembolic disease, most of which occurs in the lower extremities. However, thrombosis can also develop in the hepatic, mesenteric, gonadal, and pelvic veins, and these sites can be demonstrated by MRI.
Fetal Imaging
Fetal MRI has emerged as an accepted and essential adjunct imaging modality for prenatal diagnosis. Not only does fetal MRI provide additional information useful in patient counseling and management, it also helps determine mode of delivery, location of delivery (community versus tertiary center, labor and delivery room versus operating room), multidisciplinary involvement (pre-, peri-, and postnatal), and decisions regarding fetal intervention. These issues include in utero procedures as well as requirement for airway establishment during delivery.
The fetal MRI examination is conducted to evaluate the fetus within the gravid uterus. Detailed analysis is performed, including placental location, cervical length, amniotic fluid volume, placental cord insertion, and fetal anatomic evaluation. Imaging may be focused on a specific region of interest, but the entire fetus is typically evaluated. Imaging begins with a triplanar scout sequence (axial, coronal, and sagittal to the mother), followed by three-plane orthogonal imaging of normal and pathologic fetal anatomy. Multiplanar T2-weighted images, as noted earlier, are the mainstay of fetal imaging sequences with bSSFP, T1-weighted images, DWI, and other acquisitions applied, depending upon clinical circumstances.
Anomalies of the Central Nervous System
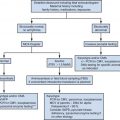
In 1997, Levine and colleagues reported the utility of MRI as an adjunctive modality for CNS anomalies suspected at ultrasound examination ( Fig. 23-5 ). MRI not only corroborated ultrasound findings but also demonstrated additional findings that could alter patient counseling and management. The goal of MRI is to perform an anatomic survey documenting normal intracranial structures and to identify and characterize abnormalities in order to provide a comprehensive, precise diagnosis. In a study evaluating the utility of MRI with respect to gestational age, it was found that if an MRI examination was performed prior to 24 weeks’ gestational age, decisions influenced the continuation or termination of a pregnancy, particularly in the United States. Management decisions that occurred if an MRI examination was performed after 24 weeks of gestation were those relating to delivery location, mode of delivery, and postnatal care. Major CNS indications for referral to fetal MRI include ventriculomegaly, midline defects, posterior fossa abnormalities, and neural tube defects. Additional indications include, but are not limited to, cortical abnormalities, encephaloclastic abnormalities, cystic masses, and tumors ( Table 23-1 ).

| Brain and Spine |
|
| Skull, Face, and Neck |
|
| Thorax |
|
| Abdomen, Retroperitoneum, and Pelvis |
|
| Complications of Monochorionic Twins |
| Fetal Surgery Assessment |
Ventriculomegaly is defined as dilatation of the lateral ventricle greater than 10 mm at the level of the atria and is one of the most common indications for fetal MRI. Ventriculomegaly may be isolated or may be associated with an underlying cause including obstruction from congenital aqueductal stenosis, dysmorphologic appearance such as agenesis of the corpus callosum, intracranial hemorrhage, or syndromal ventriculomegaly, as part of a complex malformation ( Fig. 23-6 ). MRI has been shown to be useful in fetuses with ventriculomegaly, allowing for assessment of degree of enlargement, search for underlying cause, and presence/absence of additional findings ( Fig. 23-7 ). Because the prognosis is related to the presence of additional abnormalities, prenatal detection of associated anomalies is essential. Sonographically occult findings can include agenesis of the corpus callosum, cortical malformations, cerebellar abnormalities, and destructive changes.


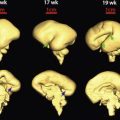
Corpus callosal agenesis is a disorder of prosencephalic midline development. The corpus callosum can be visualized on midline sagittal MRI after 20 weeks’ gestational age. Fetal MRI has been able to delineate a normal corpus callosum in 20% of cases referred for suspected callosal agenesis and has detected occult abnormalities including gyral abnormalities, posterior fossa abnormalities, brainstem anomalies, and periventricular nodular heterotopia in up to 63% of cases ( Figs. 23-8 and 23-9 ).


Holoprosencephaly is a complex anomaly of the prosencephalon manifested by lack of cleavage of midline structures. This malformation is classified into subtypes representing a continuum of midline nonseparation. In decreasing order of severity, they include alobar, semilobar, lobar, and middle interhemispheric variant types of holoprosencephalies ( Figs. 23-10 and 23-11 ).


Fetal MRI can help distinguish an abnormal from a normal posterior fossa and can aid in determining true defects from normal variants. Prognosis and neurodevelopmental outcome depend on the degree of vermian dysplasia and presence of additional supratentorial anomalies ( Figs. 23-12 and 23-13 ). Diagnoses of Dandy-Walker malformation, vermian agenesis, and brainstem anomalies portend a poor prognosis; cerebellar hypoplasia is associated with an uncertain prognosis; and mega cisterna magna, Blake pouch cyst, and retrocerebellar arachnoid cyst are associated with a good neurologic prognosis ( Fig. 23-14 ).