Pain from sacral insufficiency fractures or metastatic tumor to the sacrum, refractory to radiation and/or chemotherapy, can be extremely debilitating to affected patients. Conservative medical therapy with rest, limited ambulation, and pain medication has been the mainstay of treatment. Open surgical fixation is reserved for severe fracture that does not heal with rest. A minimally invasive treatment, sacroplasty, is gaining popularity and uses image-guided, percutaneous injection of surgical cement into the fracture. This article reviews the incidence, causes, diagnosis, presentation, and treatment options for sacral fractures, and describes detailed technical steps of the sacroplasty procedure.
Sacral fractures
Incidence
Spontaneous, pathologic fractures of the sacrum are now well known, but were only first described as recently as 1982 by Lourie. Such injuries are often termed as “insufficiency fractures,” indicating that the bone strength is insufficient to withstand normal mechanical and physiologic forces. The incidence of sacral insufficiency fractures (SIFs) is substantially less than that of osteoporotic fractures involving the lumbar and thoracic spine; however, in a published series evaluating elderly patients with acute onset of low-back pain and negative radiographs, bone scintigraphy diagnosed 102 sacral fractures over a 2-year period at a single institution. In a second study, using retrospective review of 1017 consecutive bone scans in patients older than 70 years, SIFs were identified in 194 patients (19%). Weber and colleagues reported an incidence of 1.8% of SIFs (n = 20, total = 1015) in a female population, older than 55 years, admitted to a single department with low-back pain. In an active, single practice setting that treated approximately 2000 patients with osteoporotic vertebral fractures, 80 patients with sacral insufficiency fractures were identified and treated, which was a relative incidence of at least 4%.
Reported data for incidence of localized sacral fractures from tumor destruction are limited at this time.
Causes of Sacral Fractures
Sacral fatigue or stress fractures occasionally occur in long-distance runners. Sacral pathologic (insufficiency or tumor related) fractures may occur in patients with fragile or abnormal bone owing to osteoporosis, disorders of calcium metabolism, osseous metastatic disease, and prior radiation therapy. Postmenopausal women with osteoporosis have been reported to be the most common group to suffer from sacral insufficiency fractures.
Diagnosis
Sacral fractures present clinically with pain. Pain localizes mainly to the sacral and buttock region, but may be referred to the low lumbar spine. Pain is usually acute in onset and exacerbated by weight bearing, which is typically relieved by rest or the supine position. Referred pain to the hips or groin is common. For osteoporotic fractures, there can be concurrent fractures of the ischial and pubic rami, or coexistent vertebral compression fractures. There is usually no related neurologic abnormality. Physical examination may show sacral tenderness on lateral compression, and there is restricted mobility that is indistinguishable from sacroiliac arthropathy and other spinal and pelvic pathologic conditions.
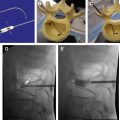
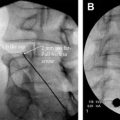
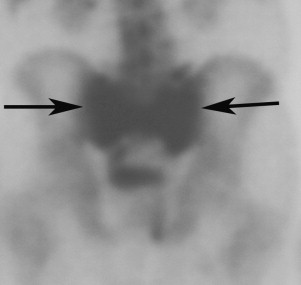
Conventional plain radiographs are insensitive, only occasionally showing sclerosis of the sacrum or an actual fracture line. Computed tomography (CT) offers greater sensitivity ( Fig. 1 ); however, acute, nondisplaced fractures without sclerosis may be difficult to visualize. Bone scintigraphy is reliable, but with less anatomic detail compared with CT. Bone scintigraphy demonstrates increased tracer activity in the areas of the fracture ( Fig. 2 ). Ambiguity on bone scan in this region is common because of bowel and bladder activity or uptake around the sacroiliac (SI) joint caused by degenerative change. Careful attention to findings on the posterior nuclear images is therefore helpful when evaluating the sacral region.
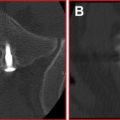
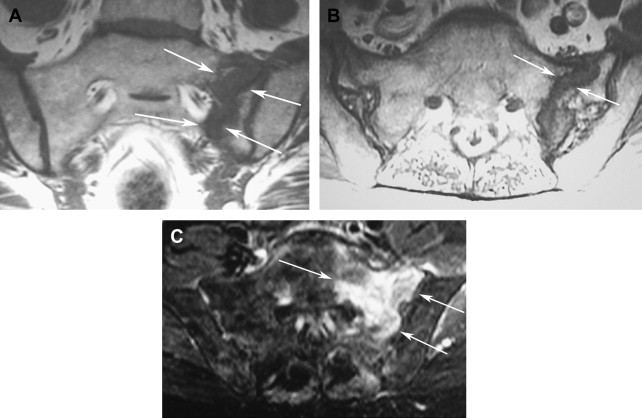
Magnetic resonance (MR) imaging is sensitive and specific in demonstrating sacral fractures. Marrow edema is visible as decreased signal on the T1-weighted images. T2-weighted short tau inversion recovery (STIR) or fat-saturated T2-weighted images show increased signal related to edema ( Fig. 3 ). Axial or coronal images show the fractures in 2 complementary planes. Although sacral insufficiency fractures demonstrate enhancement after intravenous contrast agent administration, this often is not required because the imaging findings on the unenhanced MR imaging examination are fairly characteristic. In a patient with tumor history, sacral fractures may be misinterpreted as metastatic disease, which sometimes can be differentiated with diffusion-weighted images.
Sacral Fracture Types
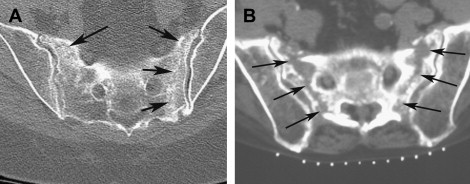
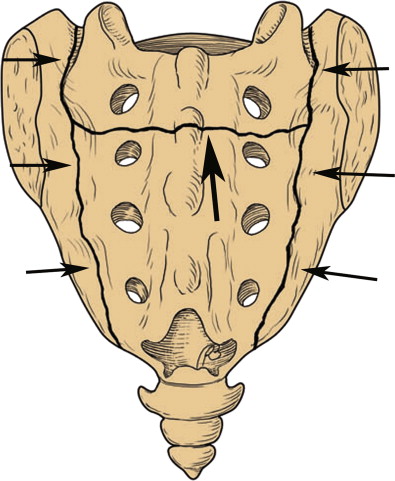
The classic appearance of a sacral insufficiency fracture on skeletal scintigraphy is an H-shaped uptake pattern ( Fig. 4 ). This pattern results from fractures of the sacral ala bilaterally with a horizontal fracture component that extends through the central sacrum (usually at about the S2 level). The fracture may present with only the vertical component on one or both sides of the sacrum. The horizontal fracture may also accompany a single unilateral component. The complexity of the fracture makes the diagnosis difficult based solely on the bone scan, thus anatomic confirmation with CT or MR imaging is always preferred.
Functional Outcome of Sacral Fractures
Functional outcome data are limited for sacral insufficiency fractures with few reported series. In a series of 60 hospitalized patients (mean age of 83 years) with pelvic insufficiency fractures, of which 16 involved the sacrum, the average hospital stay was 45 days. After 1 year, only 36.6% had the same level of self-sufficiency as before the fracture and 25% were discharged to institutions with a 1-year mortality of 14.3%. Less bleak outcomes have also been reported. In a series of 20 patients with sacral insufficiency fractures (mean age of 74 years), 17 patients had resolution of local pain and 3 patients required analgesics for pain control. In another series of 20 hospitalized patients with sacral insufficiency fractures (mean age 79), with analgesics and physical therapy, pain resolved for all 20 patients at 9 weeks. In patients with sacral fractures associated with tumor, functional outcomes data are more limited because these are patients with shorter life expectancy directly related to the tumor.
Treatment options
Medically, patients with insufficiency fractures are treated with medications to reverse osteoporosis and to decrease pain. Early mobilization is recommended to stimulate osteoblastic bone-forming activity by weight bearing or muscle tension strength. Prolonged immobilization leads to bone resorption, complications of sacral decubitus ulcers, and deep venous thromboses. Rehabilitation therapy is recommended. Surgery is usually not considered for insufficiency fractures because most patients are treated conservatively, as surgery is rarely indicated. However, surgery remains available with attempts to bridge the fractured ala areas with metallic screws placed via a lateral approach through the SI joints.
For pain and fractures from invasion of tumor, surgery is extensive, with high morbidity and complications. Patients with metastatic lesions who continue to have pain after radiotherapy and chemotherapy are considered for surgery. However, surgical fusion with hardware would be extensive, involving the lumbar spine to bilateral ileum, and postoperative complication rates for spine surgery from metastatic disease are high.
Percutaneous sacroplasty is an image-guided percutaneous injection of bone cement into the sacral fracture site. The proposed mechanism of pain relief is fracture stabilization that eliminates motion of the fractured bone. Although the concept of vertebroplasty (injection of cement into compressed thoracic and lumbar vertebral bodies) theoretically applies to sacral fractures as well, even experienced practitioners were initially hesitant to apply vertebroplasty techniques at the sacral level. This hesitance was, in part, due to constraints imposed by the complex sacral anatomy. The inherent difficulty in fluoroscopic visualization of important sacral landmarks, including the spinal canal and neural foramina, makes the detection of cement leaks into these spaces difficult. The authors recommend specific training and the use of CT guidance for sacroplasty. In a prospective, multicenter, observational study of 52 patients treated with sacroplasty, there was a statistically significant immediate improvement with dramatic decrease in pain after the procedure.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree