Second and Third Trimester Pregnancy
Over the last 30 years, ultrasound has played an essential role in the care of the obstetric patient. The body of knowledge and expertise in obstetric sonography is robust. Ultrasound is the primary imaging modality for evaluation of uterine, cervical, and amniotic fluid abnormalities; placental and umbilical cord problems; and determination of gestational age, fetal congenital abnormalities, multiple gestation, and fetal presentation.1 While many parts of the examination are not relevant in the emergency setting, there are some findings that may be critical to the acute care of an obstetric patient. This chapter discusses the use of point-of-care ultrasound to evaluate pregnant patients in their second and third trimesters. During this time period, the major indications for its use are in the initial assessment of vaginal bleeding, labor, trauma, and abdominal pain. Emphasis will be placed on a focused or goal-directed ultrasound examination to rapidly measure fetal cardiac activity, estimate gestational age, and exclude placenta previa. Additional applications include assessment of cervical length and fetal position, and for the evaluation of traumatic and nonobstetrical causes of abdominal pain.
 CLINICAL CONSIDERATIONS
CLINICAL CONSIDERATIONS
In early pregnancy, point-of-care ultrasound is commonly used to help rule out ectopic pregnancy by confirming an intrauterine pregnancy. The clinical indications for point-of-care ultrasound in early pregnancy, the limited information sought, and the recommended technique are widely agreed upon and well described (see Chapter 14, “First Trimester Pregnancy”). In contrast, the role of point-of-care ultrasound in the second and third trimesters of pregnancy is not as well established. Yet, emergency care providers are frequently faced with evaluating patients who are in the latter part of pregnancy and present to the ED with trauma, vaginal bleeding, or abdominal pain. Depending on the practice setting, obstetrical consultation may not be readily available and patients may not have had prenatal care. Increasingly, point-of-care ultrasound is readily available in the emergency setting and clinicians are adept at its use. There are clearly a number of clinical situations during the second and third trimesters of pregnancy where a rapid, goal-directed ultrasound examination can expedite the diagnosis and improve the care of mother and fetus.
A discussion of the use of point-of-care ultrasound in the second and third trimesters of pregnancy must begin by addressing the following questions:
1. What are clinical indications for point-of-care ultrasound in the second and third trimesters of pregnancy?
2. Which focused ultrasound applications are reasonable for acute care providers to perform during late pregnancy?
3. What are the goals of the point-of-care ultrasound exam in the second and third trimesters of pregnancy?
4. Is ultrasound safe, and what are the alternative diagnostic modalities in pregnancy?
What are the clinical indications for point-of-care ultrasound in the second and third trimesters of pregnancy? The concept that point-of-care ultrasound should remain goal-directed helps define its appropriate use in the latter part of pregnancy. In this setting, the focused concept takes on particular importance for the following reasons. First, since the scope and quantity of information potentially available using ultrasound in late pregnancy are enormous, the clinician performing ultrasound in the emergency setting must have a distinct, clinically relevant goal in mind before performing the exam. It would be inappropriate, for example, to assess fetal cardiac morphology in the emergency setting. Second, the medicolegal ramifications of basing clinical decisions on point-of-care ultrasound in the obstetric patient mandate caution. In one analysis of malpractice claims involving diagnostic ultrasound, obstetric ultrasound constituted 75% of the cases.2 Not only should the information sought with each application be carefully limited, but ultrasound should be used only for emergency indications where the immediate benefit of the information outweighs the possibility of a missed diagnosis.
Major clinical indications for the use of point-of-care ultrasound that seem to satisfy these constraints include the initial evaluation of a pregnant trauma patient, mid-and late-trimester vaginal bleeding, labor, and abdominal pain during pregnancy.
Which focused ultrasound applications are reasonable for acute care providers to perform during late pregnancy? Several standard point-of-care ultrasound applications are indicated in late pregnancy and are considered safe and accurate in this setting. These include the focused assessment with sonography for trauma (FAST) examination to assess for significant hemoperitoneum, a right upper quadrant ultrasound examination to assess for gallstones and signs of cholecystitis, and a renal ultrasound examination to evaluate for severe hydronephrosis. Ultrasound may also be used to rapidly visualize fetal cardiac activity. It is also reasonable to estimate gestational age by measuring biparietal diameter (BPD) or femur length. In addition, transabdominal ultrasound can be used to evaluate for possible placental abruption, to exclude placenta previa, and to ascertain the position of the fetus. Transvaginal ultrasound may be used to clarify the relationship of the placenta to the internal os and to measure cervical length.
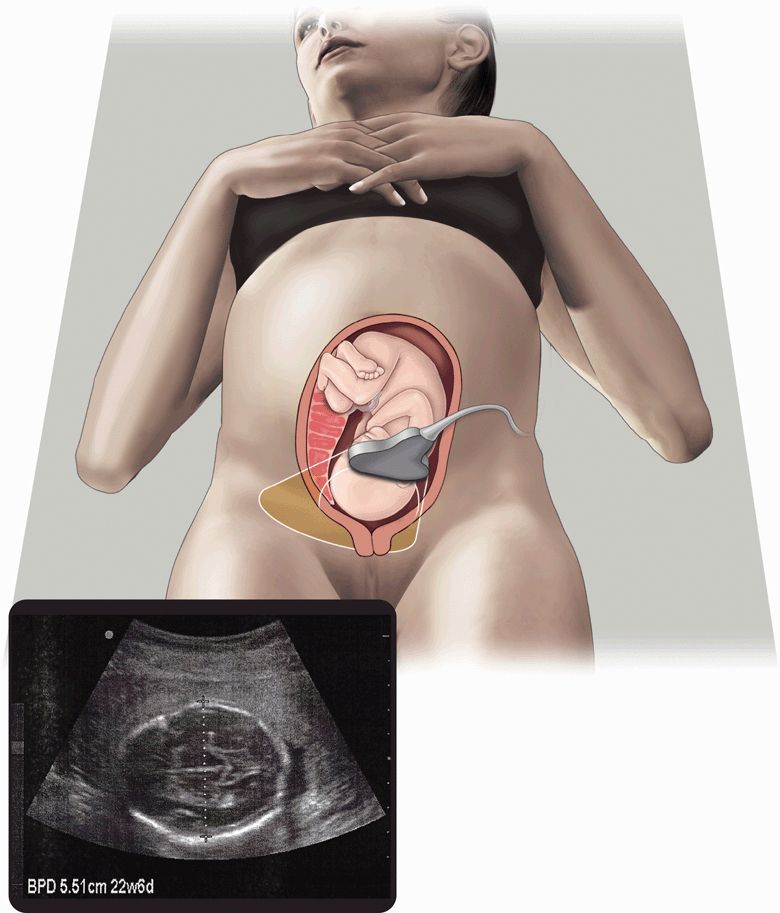
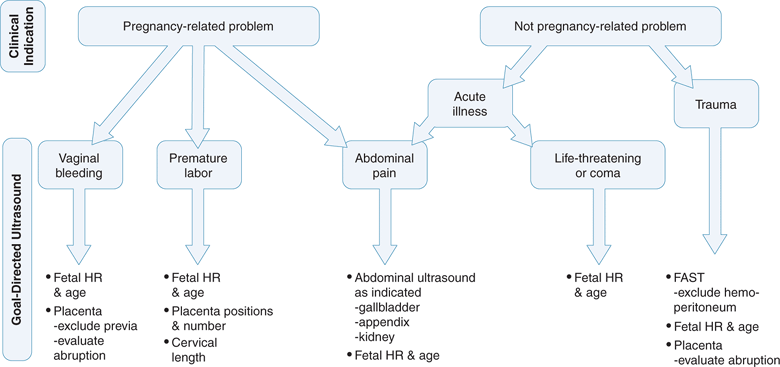
What are the goals of the point-of-care ultrasound exam in the second and third trimesters of pregnancy? In other words, given the patient’s clinical problem, what clinically important question(s) can be answered rapidly with a focused ultrasound examination? For example, in a trauma patient who is comatose and appears to be pregnant, the following questions may be answered with point-of-care ultrasound: Is the patient pregnant? Is there evidence of free intraperitoneal fluid (from hemoperitoneum or uterine rupture)? Is the fetus alive? What is the gestational age of the fetus (and might it survive in the extrauterine environment)? (Figure 15-1) Is there obvious retroplacental hematoma? Figure 15-2 presents a goal-directed approach to point-of-care ultrasound in the second or third trimester of pregnancy based on the patient’s clinical problem.
Figure 15-2. Goal-directed ultrasound in the second and third trimesters based on the clinical indication.
Is ultrasound safe, and what are the alternative diagnostic modalities in pregnancy? The emergence of ultrasound as the main imaging modality in pregnancy is based, in part, on it being considered extremely safe for the fetus. Human organogenesis largely takes place before the 10th week of gestation, the time period when diagnostic ultrasound is often used. The absence of an association between ultrasound and fetal structural anomalies supports its safety in early pregnancy.3 The critical period of brain development occurs during the 14th–22nd week of gestation. The theoretical adverse affect of ultrasound on the fetal brain, due to production of thermal energy and cavitation, has been the topic of several important epidemiologic studies, all of which have found no deleterious effects on cognitive development.4,5 By contrast, the risk to the fetus associated with exposure to ionizing radiation, particularly from abdominal CT, is considered significant.1
For assessment of fetal well-being, alternatives to sonography include the handheld Doppler stethoscope for measurement of fetal heart tones and continuous cardiotocography. Cardiotocography is a form of fetal assessment that simultaneously records fetal heart rate, fetal movements, and uterine contractions to investigate fetal well-being.
In the absence of a reliable menstrual history, a very rough estimate of gestational age can be obtained by measuring fundal height. This crude test represents the only alternative to sonography for estimating gestational age when the history is unclear.
For the evaluation of vaginal bleeding in late pregnancy, sonography has supplanted the traditional double setup and potentially harmful physical examination for excluding the diagnosis of placenta previa. Placental abruption is usually diagnosed using clinical information, by excluding the diagnosis of placenta previa with ultrasound, and with the assistance of cardiotocography. The use of magnetic resonance imaging has been described in the workup of relatively stable patients with unexplained vaginal bleeding, but it is not widely used.6
Ultrasound provides information that is complimentary to the physical examination and cardiotocography in the evaluation and management of preterm or precipitous labor. Ultrasound is the only imaging modality used in this setting to assess cervical length, amniotic fluid volume, and fetal position.
Abdominal pain of unclear etiology is an important clinical indication for use of point-of-care ultrasound in late pregnancy where alternative imaging modalities—such as plain radiography and CT—are commonly employed in nonpregnant patients with similar complaints.
 CLINICAL INDICATIONS
CLINICAL INDICATIONS
The clinical indications for performing point-of-care ultrasound in the second and third trimesters of pregnancy are as follows:
1. Determination of gestational age and fetal heart rate
2. Assessment of obstetrical causes of abdominal pain and vaginal bleeding
3. Evaluation of labor
4. Evaluation of traumatic injuries
5. Assessment of nonobstetrical causes of abdominal pain
DETERMINATION OF GESTATIONAL AGE AND FETAL HEART RATE
Fetal assessment begins with measurement of fetal cardiac activity to establish fetal viability. This step is recommended early, not only in evaluation of the pregnant trauma patient7 but also in patients with vaginal bleeding8 and preterm labor.9 The normal fetal heart rate following the first trimester is 120–160 beats perminute (bpm). Sustained bradycardia is often associated with fetal hypoxia and acidemia. Historically, in emergency medicine, fetal cardiac activity was established by measuring fetal heart tones with a handheld Doppler stethoscope.
Point-of-care ultrasound is an attractive alternative to the handheld Doppler stethoscope for initial detection of fetal cardiac activity. Using B-mode scanning of the gravid uterus, locating the fetal heart, and assessing the presence of cardiac motion is relatively straightforward. Using M-mode scanning, the waveform produced by cardiac motion can be recorded. Fetal heart rate is then determined rapidly and accurately with the aid of obstetrics software contained in most ultrasound machines.
Knowledge of gestational age is critical in many clinical settings. This information may be used to guide the decision to perform an emergency cesarean section, such as in the setting of maternal cardiac arrest.10 In addition, gestational age and fetal maturity often influence the management of placenta previa, preterm labor, rupture of membranes, eclampsia, and other severe medical illnesses in late pregnancy. In the setting of trauma, sonographic assessment of gestational age, along with fetal heart rate, can be performed as an adjunct to the initial E-FAST examination. In a stable trauma patient, gestational age may influence the decision to proceed to exploratory laparotomy.11
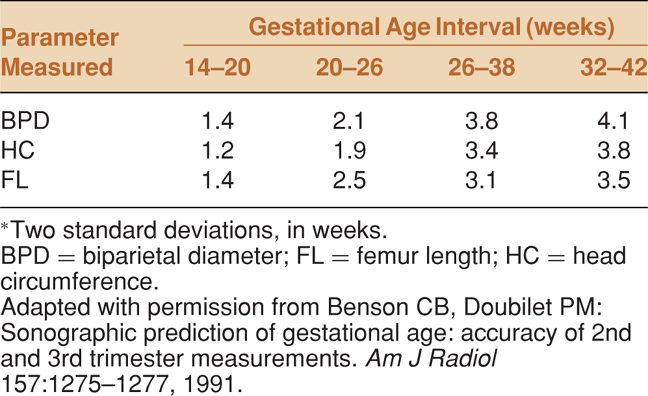
When assessing gestational age, consider the following general points. By current convention, obstetric dating begins with the first day of the last normal menstrual period, referred to as gestational age or menstrual age, and is equal to the fetal or conceptual age plus 14 days. Assume fetal viability when the gestational age is >24 weeks. The pregnancy is considered “term” at 38 weeks. Sonographic estimates of gestational age are progressively less accurate in later pregnancy due to natural variations in the size of the fetus (Table 15-1). A simple rule that reinforces this principle is that the variability (2 SD from the mean) of a gestational age estimate is equal to approximately 8% of the predicted age.12 An ultrasound measurement of BPD that yields a gestational age of 32 weeks has a variability of ±19 days. Nevertheless, estimates based on BPD, obtained as late as 20 weeks, still outperform menstrual history for predicting onset of labor.13 If possible, base gestational age on results of an ultrasound examination performed prior to 20 weeks or on a reliable menstrual history. Beyond 20 weeks, point-of-care ultrasound may be used to estimate gestational age if menstrual dates are unreliable or when the patient cannot provide a history.
In the first trimester, crown-rump length is the preferred biometric measurement for establishing gestational age. In the second and third trimesters, measurements commonly used to estimate gestational age include BPD, head circumference (HC), and femur length. Modern ultrasound machines contain software that will automatically calculate gestational age based on any one of these parameters. In choosing which bio-metric parameter to measure, the established predictive validity of the parameter (Table 15-1) should be considered, and the ease and speed with which it can be obtained. A parameter that has excellent predictive validity according to the obstetric literature, but is difficult to measure and therefore prone to error, may not be well suited to the emergency setting.
In the second trimester, BPD and HC are the most widely used measurements to determine gestational age.12,14 Although in expert hands, HC has a somewhat better predictive validity than BPD, measurement of BPD is preferable in the emergency setting because of the relative ease with which it is obtained. While HC must be calculated in a particular plane, BPD can be measured in any plane, provided the line of measurement intersects the thalamus and third ventricle.12,15 Of particular relevance to emergency medicine providers is a study that directly correlated neonatal survival in premature infants with various biometric measurements obtained by ultrasound shortly before birth. Based on analysis of receiver operator curves, a BPD of >54 mm was the single best predictor of survival.16
In the third trimester, femur length is a frequently used alternative to BPD for estimating gestational age.12,17 In late pregnancy, measurement of BPD may be difficult because the fetal skull is frequently located within the maternal pelvis and can be obscured by acoustic shadowing. The predictive validity of femur length is slightly better than BPD at this stage.14 Femur length is relatively easy to measure because the transducer need only be parallel to the long axis of the femur.12,18
In the late third trimester, identification of an ossified distal femoral epiphysis represents a potentially rapid means of estimating gestational age, and may be useful in the emergency setting. The appearance of this ossification center indicates a gestational age of 29 weeks or greater, whereas its absence means that the gestational age is <34 weeks.19 Similarly, the appearance of an ossified proximal tibial epiphysis suggests a gestational age of at least 35 weeks indicating the fetus is at or very near term.12
ASSESSMENT OF OBSTETRICAL CAUSES OF ABDOMINAL PAIN AND VAGINAL BLEEDING
The primary pregnancy-related causes of abdominal pain in the second and third trimesters are preterm labor, placental abruption, and chorioamnionitis.20–24 Chorioamnionitis refers to an infection of the amniotic fluid, typically following a rupture of membranes, or rarely as a complication of diagnostic amniocentesis. In rare cases, infection may occur without membrane rupture, causing pain, preterm labor, and systemic signs of infection. The diagnosis can be made by ultrasound-guided amniocentesis.25
There are several disorders unique to pregnancy that may cause abdominal pain. Severe pregnancy-induced hypertension can be complicated by HELLP syndrome in 5–10% of cases, which is characterized by hemolysis, elevations in liver function tests, and low platelets. Midepigastric or right upper quadrant pain is present in 25% of cases.25 Spontaneous liver or spleen subcapsular hematomas can also develop, usually, but not always, in association with pregnancy-induced hypertension. Patients may experience right or left upper quadrant pain and mild coagulopathy, but have normal liver function tests. Hematoma rupture results in peritonitis and hemorrhagic shock.24 The diagnosis is often difficult, since this condition may resemble uterine rupture or abruption, and ultrasound may be helpful in differentiating these conditions.
Patients with vaginal bleeding in the second and third trimesters are at high risk, with fetal mortality or adverse outcomes in up to one-third of all cases.25,26,27 Vaginal bleeding beyond 20 weeks’ gestational age complicates 5% of all pregnancies. In 13% of those with bleeding, placental abruption is the cause, and in 7% placenta previa is the cause.28,29 Placenta previa and abruption account for the vast majority of cases requiring transfusion or cesarean section, as opposed to cases of vaginal bleeding caused by early labor, lower genital lesions, or other diagnoses.
Placenta Previa
The term placenta previa refers to a placenta that completely covers the internal cervical os. Placenta previa has traditionally been subdivided into complete, meaning that the entire os is covered by placenta, and partial, meaning the os is partially covered. When the placental edge is located within 3 cm of the internal os, it is termed marginal placenta previa. The term low-lying placenta is useful for describing the case of a placenta located in the lower portion of the uterus in which the exact os–placenta relationship cannot be defined, or for describing an apparent placenta previa when seen in the second trimester (Figure 15-3).
Placenta previa is present at term in only approximately 0.5% of pregnancies. Yet, routine ultrasound in early second trimester has found low-lying placenta in up to 45% of patients and an apparent placenta previa in 5%.30–32 The explanation for this paradox is widely referred to as placental migration, the relatively rapid elongation of the lower uterine segment during the third trimester, which effectively moves the placenta away from the os.33
Maternal risk factors for placenta previa include advanced age, multiparity, non-Caucasian race, previous cesarean section, and prior history of placenta previa.33,34 Placenta previa usually presents as painless vaginal bleeding. However, pain from contractions sometimes accompanies the hemorrhage. The first episode of bleeding typically occurs in the third trimester, but may not occur until after the 36th week in up to one-third of cases.33
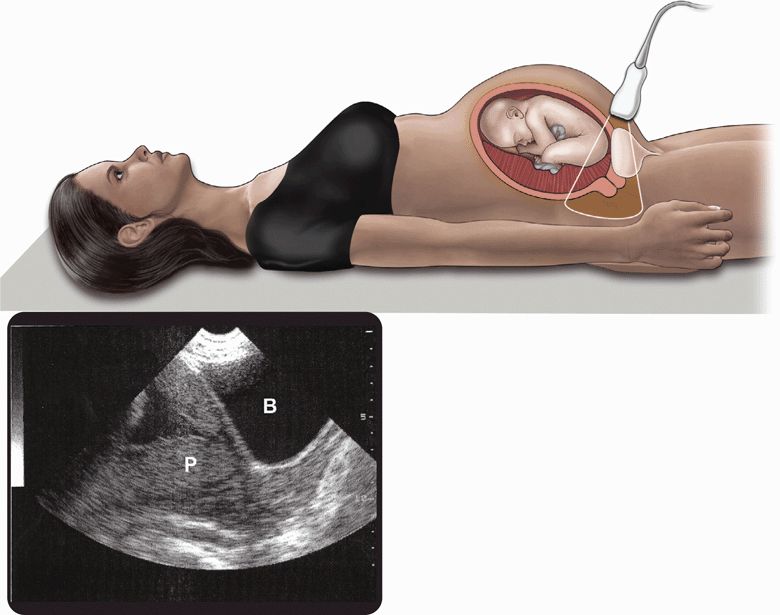
The evaluation of possible placenta previa begins with transabdominal scanning as it is rapid, noninvasive, and reliable for locating the placenta (Figure 15-4) A digital vaginal examination can precipitate severe hemorrhage in the presence of placenta previa. Ultrasound can be used to locate the placenta and exclude placenta previa prior to vaginal examination. The sensitivity for ultrasound diagnosing placenta previa is 92–98%.35,36 When the placenta is visualized at or near the fundus by transabdominal ultrasound, placenta previa is effectively excluded. After excluding the diagnosis of placenta previa, the clinician can then proceed to evaluate the patient for placental abruption. However, if the placenta is clearly seen covering the entire cervical os, particularly in the third trimester, the diagnosis of placenta previa is confirmed. When the placenta appears to be low lying or partially covering the os, or when an adequate view cannot be obtained with transabdominal ultrasound, further evaluation with transvaginal or translabial ultrasound is generally indicated. The diagnosis of placenta previa by transabdominal ultrasound has a high false-positive rate, up to 17% in one large study.37 Placenta previa is often overdiagnosed in the second trimester because it is mimicked by two conditions: (1) an overdistended bladder that compresses the lower uterine segments and (2) focal contractions.38 With the transabdominal approach, the relationship of the inferior edge of the cervix to the internal os is frequently obscured by patient obesity, an overdistended bladder, myometrial contractions, a posterior placenta, or the ossified fetal skull.39,40 In one study of patients with suspected previa, assessment of the placenta–os relationship was impossible in 31% of transabdominal studies.41 Nevertheless, in the case of severe hemorrhage, if findings on transabdominal ultrasound appear to be consistent with placenta previa, the patient should proceed directly to the operating room. A double setup examination may then be performed in the operating room at the obstetrician’s discretion.
Figure 15-4. Placenta previa. Transabdominal longitudinal ultrasound demonstrates partial previa. B = bladder; P = placenta. (Sonogram courtesy of Lori Sens and Lori Green, Gulfcoast Ultrasound)
The approach to management of placenta previa depends largely on the sonographic assessment of fetal well-being and gestational age. Although cesarean section is the definitive treatment, vaginal bleeding due to confirmed placenta previa is frequently managed in an expectant fashion. The rationales for expectant management are as follows: (1) bleeding prior to the third trimester is often self-limited and can be treated by transfusion, if necessary; (2) vaginal bleeding represents little direct risk to the fetus in the absence of significant abruption or maternal shock; and (3) delaying delivery, to maximize fetal maturity, improves perinatal outcome.33 Confirmation of fetal well-being is a prerequisite to expectant management. A rapid initial measurement of fetal cardiac activity can be performed in the emergency setting with transabdominal ultrasound, although cardiotocography is then required for ongoing fetal monitoring. Other measures of fetal well-being, such as amniotic fluid volume and biophysical profile, may have an impact on management.
A determination of gestational age also is required to guide treatment decisions.8 The following general guidelines have been proposed with regard to management of previa based on gestational age. When gestational age is <24 weeks, delivery is indicated only for hemorrhage that is life threatening to the mother. Between 24 and 34 weeks, fetal distress and life-threatening hemorrhage are indications for delivery. Beyond 34–37 weeks, delivery is indicated for fetal distress, significant bleeding, or labor.33
Placental Abruption
Any abnormal separation of the placenta occurring after 20 weeks’ gestation is defined as a placental abruption. Prior to this date, placental separation is considered part of the process of a spontaneous abortion. While it affects <1% of all pregnancies, abruption accounts for more than a quarter of all perinatal mortality.8,33 The epidemiology of placental abruption suggests that a variety of risk factors contribute to its development, many of which relate to more general microvascular disease. One of the strongest associations is with maternal hypertension, both chronic and pregnancy induced.42 Cigarette smoking and cocaine abuse have also been linked to higher rates of abruption.43,44 Trauma is an uncommon but important cause of abruption. Particular attention must be given to victims of motor vehicle crashes, falls from height, and domestic violence.28,33,45
Hemorrhage from abruption begins at the point of separation between the placenta and the uterus, or the placenta and the amnion. The timing and degree of subsequent bleeding from the cervix are dependent on the size of the hemorrhage and its location relative to the placenta. The amount of vaginal bleeding is not a reliable guide to the degree of placental abruption or the severity of hemorrhage. In some cases, patients may experience no vaginal bleeding despite significant placental separation. The amount of vaginal bleeding must never be taken as a guide to degree of internal hemorrhage. Also, the presence of abdominal pain, considered a hallmark symptom for abruption, is absent in nearly half of all cases.8,33 Therefore, when evaluating patients with painless vaginal bleeding in pregnancy, the possibility of abruption must be considered.7,33 The most consistent finding in abruption will be the presence of uterine irritability and contractions.7 These may be unappreciated by both patient and physician without the aid of cardiotocography.
Because neither the character of the bleeding nor the presence of pain can be relied upon to differentiate placental abruption from placenta previa, it is recommended that the evaluation of vaginal bleeding in the second and third trimesters begins with an ultrasound examination to exclude placenta previa. Once the diagnosis of placenta previa is excluded, abruption becomes the major diagnostic consideration.
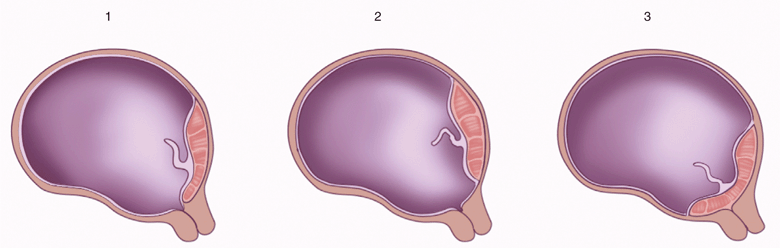
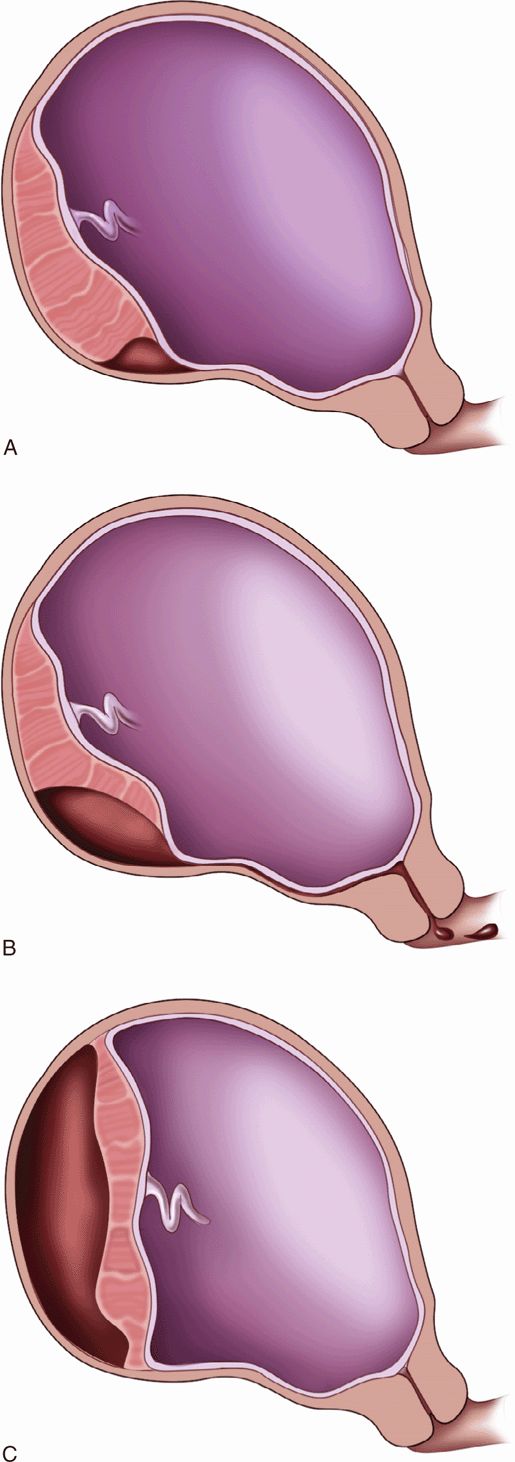
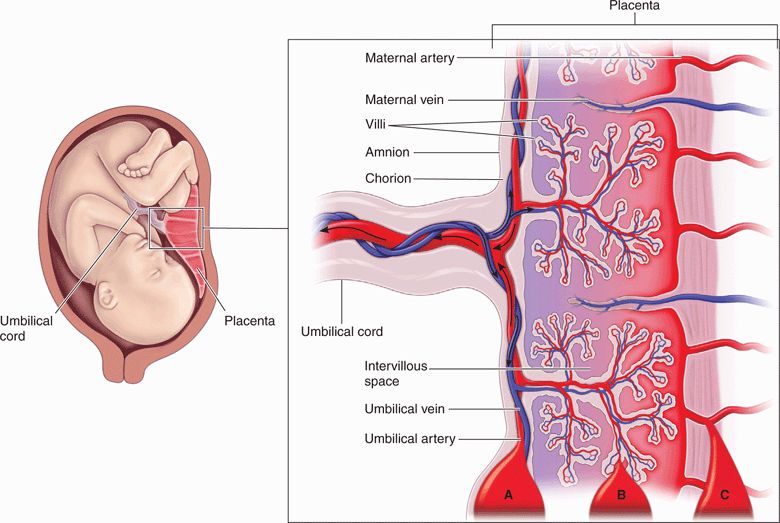
During placental abruption, hemorrhage occurs within a layer of the placenta causing separation from the adjacent uterine wall.33 This hemorrhage and separation may remain partial and self-limited or progress on to complete abruption. The clinical manifestations of abruption and its prognosis are directly determined by the extent to which placental circulation is compromised, which in turn depends on both its size and location. Hemorrhage confined to the edge of the placenta is referred to as a marginal abruption. The degree or size of placental-uterine separation is graded as mild (grade 1), partial (grade 2), or complete (grade 3) (Figure 15-5).27,46 These categories correlate reasonably well with the clinical presentation and prognosis. Grade 1 separations are usually marginal, involve less than a few centimeters of the placental border, and are usually not clinically significant. In contrast, grade 3 abruption can be fatal to both the fetus and the mother. Abruption is further categorized by the anatomic location of the hematoma relative to the placenta: retroplacental (in the decidua basalis, between placenta and uterine wall), subchorionic (between decidua and the membranes), and preplacental (between placenta and amniotic fluid, immediately beneath the amnionic membrane) (Figure 15-6).47 These distinctions also have significant prognostic implications, but primarily for fetal outcome. A retroplacental hemorrhage of 60 mL or more results in a 50% fetal mortality rate, but a similar-sized subchorionic hemorrhage results in a 10% fetal mortality rate. Preplacental hemorrhage is often self-limited and clinically silent, with 30% detected only after delivery.27,47 They appear sonographically as an irregular bulge along the inner border of the placenta. Rupture of these hematomas results in the classic “port-wine” staining of the amniotic fluid.
Figure 15-5. Classification of placental abruption.(A) Grade 1, mild abruption—as in this diagram, usually marginal in location. Presentation may be subtle or subclinical, affords best prognosis.(B) Grade 2, partial abruption—presentation and prognosis determined by location of separation and degree of compromise of maternal–fetal circulation.(C) Grade 3, complete abruption—worst prognosis. In this diagram, the hemorrhage is concealed by the tamponading effect of the placental margins.
Figure 15-6. Diagram of the anatomy of the third trimester placenta. The location of three types of placental abruption is also shown: (A) preplacental, (B) subchorionic, and (C) retroplacental.
Ultrasound is not a sensitive test for placental abruption and the diagnosis remains largely clinical, beginning with a meticulous search for evidence of vaginal bleeding, uterine tenderness, labor, or fetal distress. Signs of labor and fetal distress are the keys to making the diagnosis. Uterine contractions are present in nearly all cases of abruption, although they may be difficult to appreciate by either the physician or the patient. Contractions are characteristically of high frequency but low amplitude. It has been demonstrated that 6 hours of cardiotocographic monitoring after trauma is 100% sensitive for predicting all subsequent complications.7 Even rare cases of late-onset abruption or fetal distress after trauma are heralded by early abnormalities on cardiotocography.48 Hence, the absence of uterine irritability or fetal distress remains an excellent indicator of maternal–placental well-being, suggesting that abruption is either absent or clinically insignificant. Cardiotocography should be a routine part of the initial evaluation where there is a concern for abruption, even in those who are asymptomatic. The stable patient without evidence of uterine irritability for 6 hours may be discharged home with appropriate instructions and follow-up.
Once placental abruption is diagnosed, an immediate cesarean section remains the definitive treatment. Decisions regarding the manner and timing of any intervention depend on an overall assessment of maternal–fetal well-being. If the fetus is immature and the abruption is judged to be mild, an expectant approach may be attempted. Signs of preterm labor may be difficult to distinguish from mild abruption.33 A term fetus or evidence of uterine irritability refractory to medical management should prompt expedited delivery. Similarly, fetal distress or maternal signs of abruption indicate a need for immediate cesarean section.25,33
EVALUATION OF LABOR
Approximately 7% of newborns are premature at birth, which may result in mental or physical impairment.49 Preterm labor is defined as regular uterine contractions accompanied by characteristic changes in the cervix, occurring prior to 37 weeks’ gestation. Assessing the potential for premature delivery is the main goal of the emergency evaluation for preterm labor.
Ultrasound is a safe, rapid, and accurate method of evaluating the cervix for signs of labor. As labor begins, the cervix undergoes effacement followed by dilation. While the digital examination has traditionally been used to evaluate such cervical changes, ultrasound has emerged as a safer and more accurate means to evaluate cervical changes.50 Sonographic measurement of cervical length represents an objective way to quantify effacement.
There are several justifications for using an ultrasound examination rather than the digital examination in this setting. First, a direct contraindication to digital examination may exist, such as placenta previa or ruptured membranes, and digital examination in these settings can produce life-threatening bleeding or chorioamnionitis. A transabdominal or translabial ultrasound examination may negate the need for a digital examination. Second, an ultrasound examination has been shown in numerous studies to be more accurate than digital examination in estimating cervical length and predicting preterm labor.50
An advantage of using ultrasound is that it can visualize the internal cervical os and detect funneling. Funneling is dilatation of the internal cervical os without dilation of the external cervical os, and it is one of the earliest signs of labor. A digital examination cannot palpate the internal os, so it cannot detect funneling. An ultrasound examination is the only practical way to detect this important finding.50–55
There are three methods for imaging the cervix with ultrasound: transabdominal, transvaginal, and translabial. The transabdominal approach is the least reliable method, with successful imaging of the cervix in only 46% of patients without a full bladder and 86% with a full bladder.56 Presenting fetal parts and a large maternal habitus may obscure visualization of the cervix. The transvaginal technique produces the most consistent findings, with visualization of the cervix in up to 100% of patients.51 The translabial ultrasound is considered the most technically difficult method. In the hands of a skilled operator, however, this approach provides an adequate view of the cervix in up to 95% of patients.51,57 In the emergency setting, it is reasonable to begin with transabdominal ultrasound and then proceed to transvaginal or translabial imaging if the cervix cannot be well visualized.
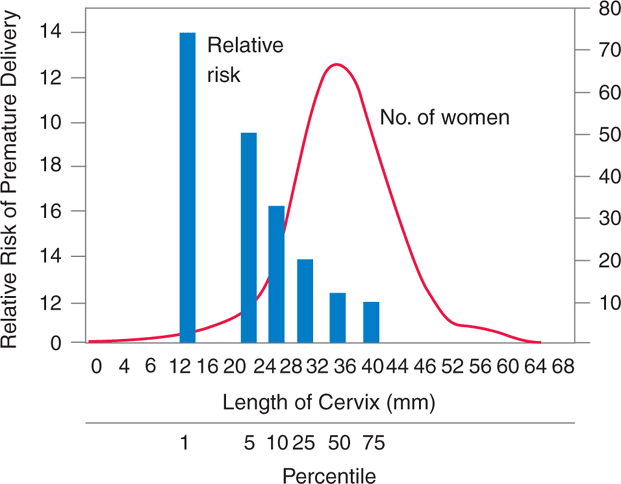
From studies of all three ultrasound methods, it is clear that cervical measurements can predict the risk of preterm delivery. In one study of nearly 3000 patients at 24–28 weeks’ gestation, it was demonstrated that a positive correlation exists between short cervical length (<30 mm) and risk for preterm birth before 35 weeks (Figure 15-7).53 Another study of patients between 16 and 28 weeks’ gestation found a 79% rate of preterm delivery in those with cervical funneling of >50%.58 Subsequent studies have confirmed the predictive value of both short cervical length and the presence of funneling, and have extended the findings to twin pregnancies.55,59 For the most part, patients in these studies were asymptomatic so applicability in the emergency setting is unclear. Also, the impact of such findings on clinical management, such as the need for cervical cerclage, remains uncertain.
Figure 15-7. Relative risk of premature delivery (solid bars) versus cervical length, measured by transvaginal ultrasound at 24 weeks. Cervical length is expressed both in millimeters and percentiles of the normal distribution. The number of subjects versus cervical length is also shown (solid curve). (Reprinted, with permission, from Lams JE, Goldenberg RL, Meis PJ, et al.: The length of the cervix and the risk of spontaneous premature delivery. N Engl J Med 334:567, 1996.)
The use of ultrasound in determining the presence or absence of cervical changes can help the emergency physician risk stratify patients with symptoms suggestive of labor. The goal of such an evaluation is to identify patients who would benefit from admission and tocolysis versus candidates for outpatient follow-up. The utility of cervical sonography in the management of preterm labor was retrospectively evaluated at one facility. Women were hospitalized only if they had a cervical length <30 mm. This protocol produced a decrease in hospital days of 48% without affecting the rate of preterm births.60 The critical cervical length appears to be 30 mm, with preterm delivery much more likely in patients with a shorter cervical measurement by ultrasound.60 Apply cervical sonography in conjunction with clinical variables—such as the results of a biophysical profile (discussed below), the gestational age, and a history of preterm births—to influence management decisions.
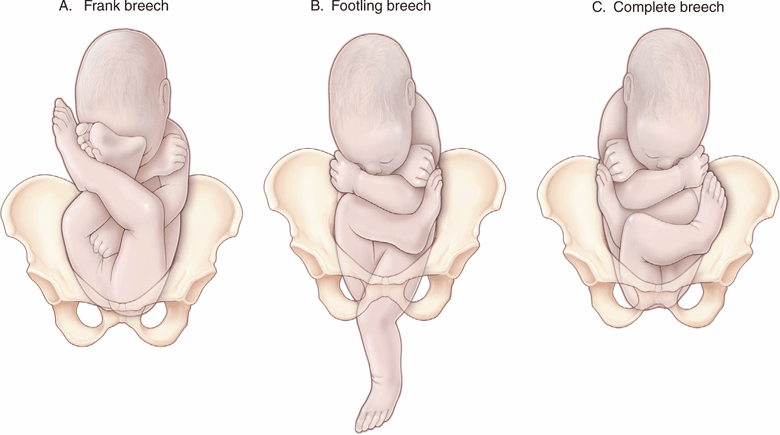
Lie refers to the relationship of the fetus to the long axis of the uterus, while presentation describes what fetal part is nearest the cervix. In normal deliveries, the fetal lie is longitudinal and the presentation is cephalic. Transverse lie and breech presentation—where the fetal sacrum and feet, respectively, are engaged in the pelvis—are referred to as malpresentations. The classification of breech presentations is demonstrated in Figure 15-8.
Figure 15-8. Types of breech presentation. In a frank breech presentation (the most common), the thighs are flexed at the hips with the legs and knees extended. In complete breech (the least common), the thighs are flexed at the hips, and there is flexion of the knees as well. One or both hips and knees are extended in the footling breech. The risk of cord prolapse is greatest with footling breech and least with a frank breech.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


 CLINICAL CONSIDERATIONS
CLINICAL CONSIDERATIONS CLINICAL INDICATIONS
CLINICAL INDICATIONS ANATOMICAL CONSIDERATIONS
ANATOMICAL CONSIDERATIONS GETTING STARTED
GETTING STARTED TECHNIQUE AND NORMAL ULTRASOUND FINDINGS
TECHNIQUE AND NORMAL ULTRASOUND FINDINGS COMMON ABNORMALITIES
COMMON ABNORMALITIES OTHER SELECTED ABNORMALITIES
OTHER SELECTED ABNORMALITIES PITFALLS
PITFALLS CASE STUDIES
CASE STUDIES