- Comminuted
- Transverse/oblique/spiral
- Impacted
- Greenstick
- Avulsion
- Burst/crush
- Insufficiency
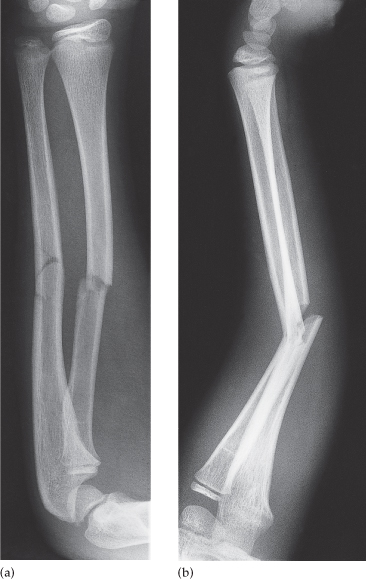
In any case of trauma it is essential to take at least two views, preferably at right angles to one another. Sometimes a fracture or dislocation will be seen on only one view and so may be missed unless two views are taken. Similarly, the position of a fracture should never be assessed from a single view (Fig. 14.1).
Fig. 14.1 Value of two views for demonstrating the position of fractures. (a) The fractures of the radius and ulna show little displacement on the frontal projection. (b) The lateral view, however, shows a marked angulation.
Computed Tomography
The major advantages of multidetector computed tomography (MDCT) over plain films are:
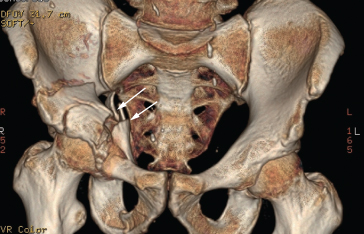
- Better assessment of fractures in bones of complex shape, such as the spine and pelvis (Fig. 14.2) which can be aided by three-dimensional reformation of the CT images. Reconstructions to any desired plane (Fig. 14.3) or three-dimensional images (Fig. 14.4) are easily obtained on modern equipment. In the spine, fractures of the pedicles, laminae and articular facets as well as fragments displaced into the spinal canal are particularly well seen (see Fig. 14.3). In fractures of the pelvis, especially those around the hip joints, CT shows the relationship of the fractures to the joint as well as loose fragments within the joint (see Fig. 14.2). CT is invariably performed in acetabular fractures and fracture dislocations of the hip. It is also very helpful in tibial plateau fractures and fractures of the ankle, calcaneum and midfoot.
- Assessment of the extent of soft tissue damage, haematomas and of internal visceral injuries.
- In general, less manipulation of the patient is required, so that the examination of the severely injured individual is more comfortable and often safer. The examination is quick, an important factor in patients with serious internal injuries.
Fig. 14.2 Fracture of the pelvis. (a) A section through the acetabular roof demonstrating an anterior column injury with displaced bony fragments. (b) A coronal section showing the fracture of the acetabulum extending to the iliac bone superiorly. The fractures and their displacement were much better demonstrated with CT than with radiographs of the pelvis.
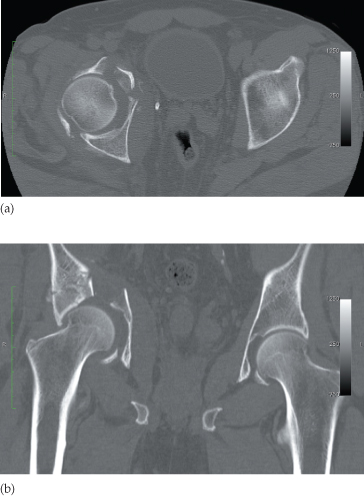
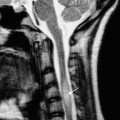
Fig. 14.3 CT scanning in a spinal fracture. (a) A comminuted fracture of C7 with displacement of a large bone fragment (arrow) into the spinal canal. (b) Sagittal reconstruction also shows the fractured vertebra and displaced fragment (arrow).
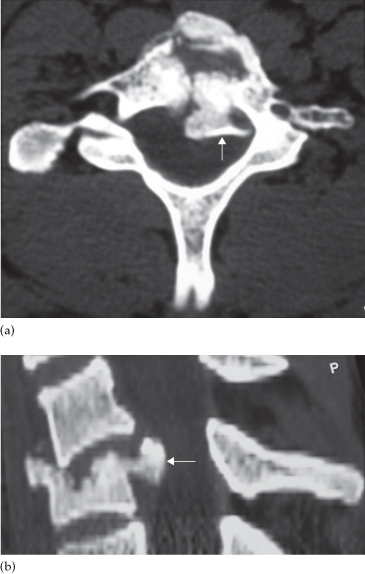
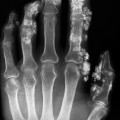
Fig. 14.4 Multiple fractures. Three-dimensional MDCT reformat demonstrating a complex pelvic fracture (arrows), useful in aiding operative planning.
Magnetic Resonance Imaging
Even though cortical bone does not produce a magnetic resonance signal, a fracture can be seen as a dark line across the bright signal of the fat in the marrow on a T1-weighted magnetic resonance imaging (MRI) scan (Fig. 14.5a). Increased signal intensity is seen within the bone, representing haemorrhage and/or oedema on a T2-weighted scan; a STIR sequence has a T2-weighted appearance with saturation of the fat signal intensity (Fig. 14.5b). Sometimes a bone bruise may be visible on MRI (Fig. 14.6) even though there is no discernible fracture on a conventional radiograph.
Fig. 14.5 Coronal MRI of the wrist. (a) T1-weighted image and (b) STIR image, showing a low signal intensity fracture line in the waist of the scaphoid (arrows). The STIR image shows surrounding haemorrhage and oedema as high signal either side of the fracture.
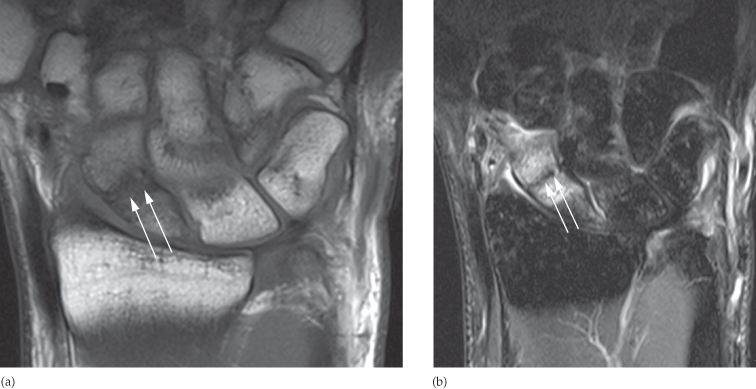
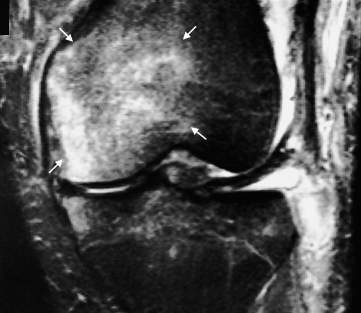
Fig. 14.6 Bone bruise. MRI in a patient who suffered severe soft tissue damage to the lateral side of his knee. The high signal in the medial femoral condyle (arrows) is due to a bone bruise. The plain films of the knee showed no bony injury.
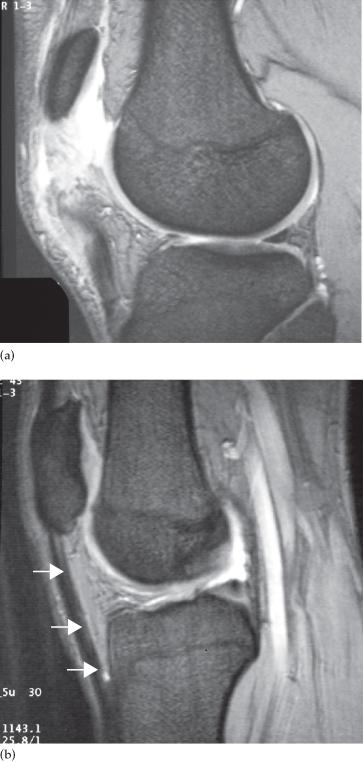
Magnetic resonance imaging is also very useful for demonstrating injury to soft tissues such as muscle, tendons and ligaments and is particularly useful in knee injuries (Fig. 14.7). MRI is the best examination for sports injury and repetitive strain injuries.
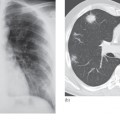
Fig. 14.7 Rupture of patella tendon. (a) MRI showing diffuse high signal in the region of the patella tendon. (b) Normal patella tendon (arrows) for comparison.
It is also the best method for demonstrating scaphoid fractures and its use has been advocated as the initial investigation.
Radionuclide Bone Scanning
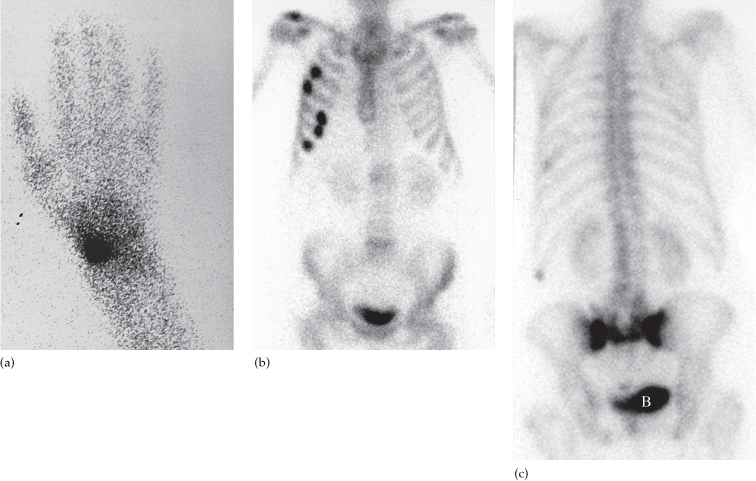
Fractures may not be visible on plain films, and, in these instances, radionuclide bone scanning maybe helpful (Fig. 14.8), although increasingly MRI is often used in these circumstances. The bone scans show increased activity at injured sites within 2–3 days. Increased activity persists for as long as the fractures are healing, often lasting several months. Multiple fractures occasionally give a picture resembling metastases, but usually the distribution suggests injury.
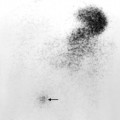
Fig. 14.8 Radionuclide bone scans in trauma. (a) Fracture of the scaphoid. There is increased activity in the scaphoid in this patient who suffered continuing pain after trauma to the wrist. In spite of normal x-rays, the bone scan indicates there is a fracture that was not visible on the radiographs. (b) Fractures in five of the ribs on the right. The distribution of increased uptake is diagnostic of injury. (c) Insufficiency fracture. There is increased uptake in the sacrum in this elderly woman who had a normal pelvic x-ray. B, isotope in the bladder.
Imaging Fractures
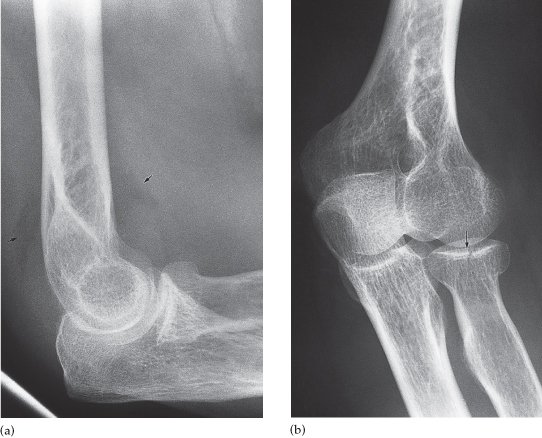
Frequently, a fracture is very obvious but in some cases the changes are more subtle. Fractures may be recognized or suspected by several signs (Box 14.2). A fracture line may been seen as a lucent line. This may be very thin and easily overlooked (Fig. 14.9). Occasionally, the fracture appears as a dense line from the overlap of the fragments (Fig. 14.10). A step in the cortex may be the only evidence of a fracture (Fig. 14.11). Interruption of bony trabeculae is of use in impacted fractures where there is no visible lucent line. This is, however, a difficult sign to evaluate (Fig. 14.11). Bulging or buckling of the cortex is a particularly important sign in children, where fractures are frequently of the greenstick type (Fig. 14.12). Soft tissue swelling may be a valuable guide to the presence of an underlying fracture. A joint effusion may become visible following trauma, and, as in elbow fractures, displaced fat pads may indicate an occult radial head fracture (Fig. 14.13).
- Fracture line (lucent or dense)
- Step in the cortex
- Interruption of bony trabeculae
- Bulging or buckling of the cortex
- Soft tissue swelling
- Joint effusion
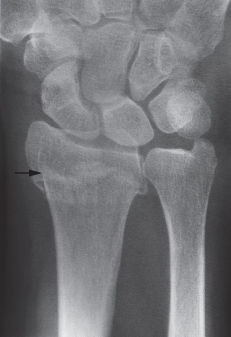
Fig. 14.10 Fracture of the lower ulnar metaphysis appearing as a sclerotic line (small black arrow).
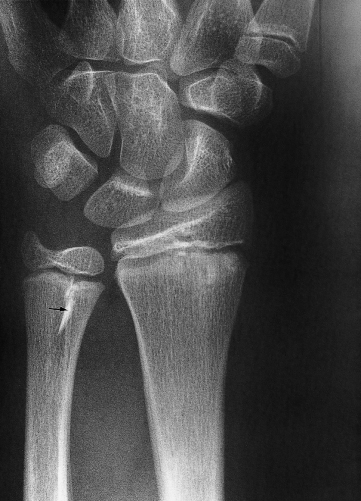
Fig. 14.12 Greenstick fracture of the lower end of the radius in a child. There is buckling of the cortex (arrows).
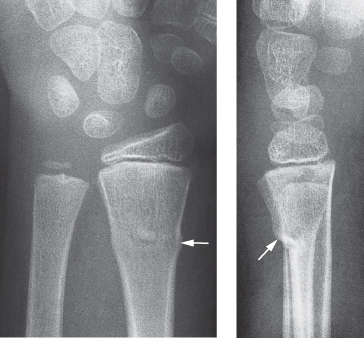
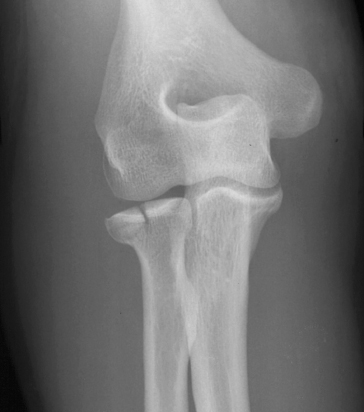
Fig. 14.13 Elbow effusion with fracture of the radial head. (a) The anterior and posterior fat pads (arrows) are displaced away from the humerus, which almost invariably means a fracture is present. (b) Oblique view in this patient showing the fracture of the radial head (arrow) which was only demonstrated on the oblique view.
Some injuries are likely to produce fractures in more than one site. With tibial fractures, for example, the fibula is frequently also broken but the fractures may be a considerable distance apart. Certain bones (e.g. the pelvis and mandible) often fracture in two sites, only one producing severe symptoms. In these situations, all likely fracture sites should be included on the films.
Imaging Dislocations
The joint surfaces no longer maintain their normal relationship to each other. Careful evaluation for an associated fracture should be performed.
Further Plain Film Views
Injuries may sometimes be invisible even with two views taken at right angles to each other. If the radiographic findings are equivocal, or if there is clinical suspicion of bony injury with normal radiographs, then further films should be taken as follows:
- Different projections, e.g. oblique views (Fig. 14.14).
- Stress films. A film taken with a joint under stress may show that it is unstable due to ligamentous damage. Stress films are helpful in ankle injuries when forced inversion and eversion may show movement of the talus (Fig. 14.15).
- Flexion and extension views. In the cervical spine, injury may cause alteration in the alignment of the posterior borders of the vertebral bodies. This is usually much more obvious on a film taken with the neck flexed (Fig. 14.16

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree