In this review, limitations affecting the results of presurgical mapping with blood-oxygen-level-dependent (BOLD) functional magnetic resonance imaging (fMRI) are discussed. There is a great need to standardize fMRI acquisition and analysis methods and establish guidelines to address quality control issues. Several national and international organizations are formulating guidelines and standards for both clinical and research applications of BOLD fMRI. Consensus regarding management of these issues will likely both improve the clinical standard of care and enhance future research applications of fMRI.
Key points
- •
Blood-oxygen-level-dependent (BOLD) functional magnetic resonance imaging (fMRI) has the potential to become a more universal standard of care in presurgical planning for localization of eloquent cortex at risk during surgical resection.
- •
BOLD imaging is affected by a series of technical issues limiting the widespread clinical use of BOLD fMRI.
- •
Extensive and standardized quality control tools need to be established for appropriate interpretation of both clinical and research fMRI studies.
- •
Newly developed methods can overcome current BOLD imaging issues and enhance future research and clinical application of BOLD fMRI.
Introduction
Over the last 20 years, blood-oxygen-level-dependent (BOLD) functional magnetic resonance imaging (fMRI) has been effectively used for clinical presurgical mapping. However, there are important technical limitations and special considerations that one must be aware of to avoid pitfalls in both clinical and research applications of BOLD fMRI.
Introduction
Over the last 20 years, blood-oxygen-level-dependent (BOLD) functional magnetic resonance imaging (fMRI) has been effectively used for clinical presurgical mapping. However, there are important technical limitations and special considerations that one must be aware of to avoid pitfalls in both clinical and research applications of BOLD fMRI.
Image acquisition
Susceptibility Artifacts
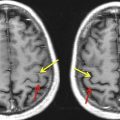
Most clinical and research fMRI studies are performed by using a two-dimensional T2*-weighted gradient recalled echo (GRE) sequence with echo planar imaging (EPI) readout. The rationale behind this choice is the high sensitivity of this pulse sequence to BOLD-related susceptibility changes and its ability to scan the whole brain with adequate spatial (2–3 mm) and temporal (2–3 seconds) resolution to monitor brain activation over time. However, T2* GRE EPI shows high sensitivity as well to intravoxel dephasing caused by macroscopic magnetic field gradients generated by the difference in magnetic susceptibility of multiple tissues contained in 1 voxel. The different magnetic fields experienced by the spins make them precess at different frequencies and, over time, dephasing leads to signal loss. This effect is strong and results in signal loss in regions of the brain characterized by strong susceptibility differences at the junctions between air and tissues such as the orbitofrontal cortex (from the paranasal sinuses) or the medial temporal and the inferior temporal lobes (from the petrous apices and mastoid air complexes), as shown in Fig. 1 . These regions are important in visual and cognitive processing, including language and memory function. The effect of this signal loss is a reduced sensitivity to brain activation in these regions, which may not be recognized when the statistical maps are overlaid on less distorted high-resolution T1-weighted anatomic images. Such susceptibility-related signal loss may result in regional false-negative activation on BOLD presurgical mapping studies. The amount of signal loss has been shown to be dependent on the image orientation, echo time (TE), and spatial resolution. Because the magnetic field gradients are generated along the slice selection, phase encoding, and readout directions, in-plane dispersion is experienced as well as through-plane dispersion of the voxel magnetization. Spatial resolution also counts, because the larger the voxel size, the larger the difference in Larmor frequencies among the spins contained in the voxel and, in turn, the faster the signal dispersion. Reducing voxel size reduces the effects of susceptibility artifacts but at the cost of temporal resolution or reduced brain coverage. The spin dephasing increases along time; therefore, the strength of signal loss depends also on the TE. In principle, one could reduce the TE, but this is at the expense of reducing BOLD sensitivity in other regions of the brain less affected by susceptibility artifacts. In current clinical studies, a tradeoff between spatial/temporal resolution and BOLD sensitivity needs to be achieved.
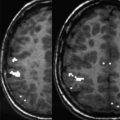
In clinical functional imaging, additional potential sources of susceptibility artifacts include vascular clips, stent grafts, or craniotomy hardware related to previous surgery. These devices can induce strong macroscopic field gradients and generate dramatic signal loss, as shown in Fig. 2 for a patient who underwent presurgical mapping after previous surgery. Furthermore, tumor-related or surgery-related hemorrhages can also cause susceptibility artifacts. Adequate presurgical mapping may still be possible in most of these cases, as reported by Peck and colleagues. However, statistically significant decreased volume of motor activation in the tumor hemisphere was reported in their group of patients with previous surgery compared with another group of patients who had no previous surgery. The investigators explained this difference in volume of activation as a consequence of the loss of BOLD signal and sensitivity to neural activation caused by the artifacts as a result of previous surgery. It is therefore advisable as a general quality control step in clinical functional imaging studies to visually inspect the raw EPI images to assess areas potentially affected by susceptibility artifacts, which may be areas prone to false-negative activation.
Susceptibility artifacts represent one of the major limitations of BOLD functional imaging performed using T2* EPI sequences. In the past, other sequences, such as spin echo EPI or fast low-angle shot or T1-weighted sequences have been explored to overcome this problem. However, they have been plagued by reduced sensitivity to neural activation, inadequate temporal resolution, or the need for exogenous contrast media. Multiple variants of the T2*-weighted BOLD sequences have been proposed in the last few years that attempt to reduce susceptibility artifacts and maintain sensitivity throughout the brain. From a clinical functional imaging standpoint, multiecho EPI and slice-dependent TE sequences with parallel imaging acquisition are promising. With the increasing availability of multichannel coils at high field (3 T), they can be easily implemented on clinical scanners, because they are slightly modified versions of the standard product GRE sequences. In addition, postprocessing requires only minor changes from standard streamlined processing pipelines.
Geometric Distortions
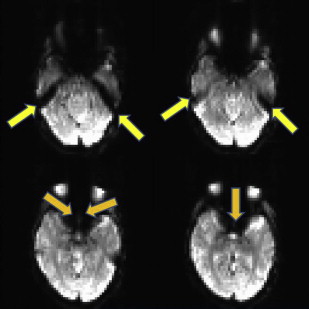
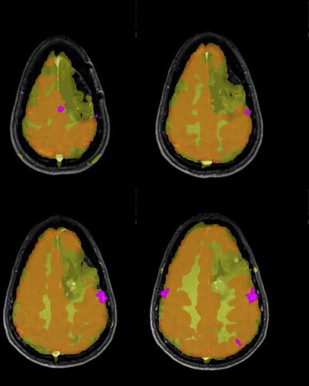
EPI sequences provide the great advantage of acquiring an entire image in a fraction of a second. It is mainly for this reason that they have been the chosen sequences for most research and clinical brain functional imaging studies. One of the main drawbacks for these sequences is the high sensitivity of EPI to geometric distortions, which manifest as mislocalization of the signal in the phase encoding direction and signal intensity loss. The most prominent source of distortion for EPI sequences is the effect of magnetic field inhomogeneities. The low bandwidth in the phase encoding direction (∼20 Hz/pixel) compared with the larger bandwidth in the readout direction (>1000 Hz/pixel) makes EPI signal sensitive to small variations of the main magnetic field. A field inhomogeneity of 100 HZ can then lead to a 5-pixel displacement (∼15 mm in a typical fMRI experiment with 3-mm voxel size). Regions of the brain close to air–soft tissue interfaces, such as the paranasal sinuses, are the most prone to distortions because of strong magnetic susceptibility variation. A minor source of distortions is the imperfect linearity of the gradient waveforms. The most critical consequence of geometric distortions is the suboptimal coregistration of the fMRI data with the structural images acquired in the same session, especially if simple rigid body or even affine algorithms are used, as shown in Fig. 3 . When activation maps are overlaid on structural images, the localization of neuronal activation can be inaccurate in some areas of the brain, if proper algorithms with local linear and nonlinear warping are not used for coregistration. Two strategies are commonly used to reduce and correct for image distortions: one consists of acquiring field maps by using a gradient echo sequence with 2 echoes. The field value is calculated voxelwise by the difference in phase between 2 gradient echo images acquired at the 2 different echoes. The local field variation can then be used to estimate the amount of shift to apply to each pixel to correct for distortion. As shown in Fig. 4 , the correction of geometric distortion results in more accurate coregistration with the structural images and consequently, more accurate activation localization. The point spread function (PSF) is another popular technique to correct geometric distortions. It uses acquisitions with additional phase encoding gradients applied in the x, y, or z directions to map the one-dimensional, two-dimensional, or three-dimensional PSF of each voxel. These PSFs encode the spatial information about the distortion and the overall distribution of intensities from a single voxel. The measured image is the convolution of the undistorted density and the PSF. Measuring the PSF allows the distortion in geometry and intensity to be corrected. Both these techniques require additional scan time and processing time, and, for this reason, they are not routinely applied in presurgical clinical functional imaging examinations. For this reason, a manual coregistration approach has been suggested by Nennig and colleagues in their proposed pipeline for standardized clinical fMRI data processing. This approach has been shown to provide reliable results but is still operator dependent and requires dedicated personnel. In current and future generations of clinical scanners with the increasing availability of multiple channel coils (16–32 channels), parallel imaging can play a crucial role as a tool for reduction of geometric distortions. Reducing by half or more the number of acquired k-space lines reduces the rate of k-space transversal that is proportional to the magnitude of susceptibility-induced distortions.
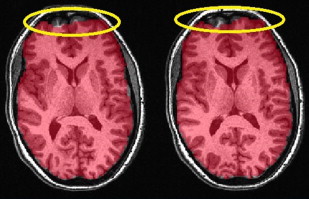
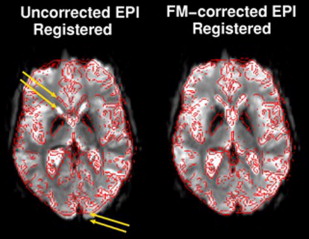
Image analysis and interpretation: false-positive and false-negative activation
Venous Effects
Brain mapping with BOLD fMRI detects transient changes in blood flow, blood volume, and blood oxygenation after the onset of neuronal activation. Although the whole brain coverage and noninvasive nature of BOLD fMRI are advantages, the associated hemodynamic changes can propagate from the capillary beds adjacent to the site of neuronal activation downstream into large draining veins far from the areas of activated neurons, resulting in uncertainty of localization of neural activity by BOLD fMRI. The BOLD contrast arises from a complex interplay between cerebral blood flow, cerebral blood volume, and cerebral metabolic rate of oxygen consumption on a spatial scale that lumps together hundreds of thousands of neurons in each MR imaging voxel. For this reason, the BOLD baseline signal and its increase on neuronal activity have been shown to be highly dependent on the characteristics of the local vasculature. At 1.5 T and 3 T, BOLD activation is predominantly located in veins and venules in spatial proximity to activated neurons, and since the earliest days of BOLD fMRI, it became clear that the strongest BOLD signal change was generated in draining veins. BOLD activation maps then lack spatial specificity because of these so-called venous effects. Venous effects increase the probability of type I error (false-positive or spurious activation), particularly in areas of high vascular density or in close spatial proximity to large draining veins, such as the sagittal sinus and adjacent cortical veins ( Fig. 5 ). Likewise, the attempt to improve spatial specificity by increasing the statistical threshold may increase the chance of type II error (false-negatives), because the weaker parenchymal signal may wash out. Multiple solutions have been suggested in the literature to reduce or suppress the venous BOLD signal. Some methods attempt to discriminate the venous signal by exploiting the differences between BOLD percentage signal change and response latency between voxels with low and high vascular density. Other methods use venous vascular masks generated from different sequences, such as susceptibility weighted imaging to remove BOLD venous signal. With the introduction of ultrahigh field magnets (≥7 T fields), activation maps are expected to be less affected by the venous effect. Multiple studies suggest an improvement not only in sensitivity but also in specificity for BOLD signal at 7 T and higher field, because the increase of BOLD signal change in the parenchyma is predicted to be higher in microvasculature than macrovasculature, because the BOLD contrast is proportional to B0 for large vessels and proportional to B0 2 for small vessels and capillaries. However, in a recent study aimed to quantitatively compare BOLD activation at 3 and 7 T, a statistically significant higher increase of specificity compared with sensitivity was not found.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree