Functional
Neoplastic
Inflammatory/Infectious
Other
Functional cyst
Surface epithelial–stromal tumors (see separate subclassification below)
Tubo-ovarian (TO) abscess
Endometrioma
Hemorrhagic cyst
Sex-cord–stromal tumor (see separate subclassification below)
Hydro-/hemato-/ pyosalpinx
Ectopic gestation
Corpus luteum
Germ cell tumor (see separate subclassification below)
Polycystic ovaries/ovarian hyperstimulation
Carcinosarcoma
Lymphoma
Paraovarian/paratubal cyst
Metastases
Ovarian torsion
Peritoneal inclusion cyst
Table 28.2 classifies adnexal lesions according to imaging morphologic characteristics into predominantly cystic, predominantly solid, and mixed cystic–solid masses.
Table 28.2
Classification of adnexal pathologies on the basis of imaging morphologic characteristics
Predominantly cystic | Cystic–solid | Predominantly solid | |
|---|---|---|---|
Ovarian | Functional cyst | Mature cystic teratoma | Fibroma |
Hemorrhagic cyst | Cystadenofibroma | Fibrothecoma | |
Endometrioma | Serous/mucinous cystadenoma | Brenner tumor | |
Polycystic ovaries | Endometrioid carcinoma | Granulosa cell | |
Ovarian hyperstimulation | Clear cell carcinoma | ||
Serous/mucinous cystadenoma | Dysgerminoma | ||
Metastases | |||
Ovarian torsion | |||
Non-ovarian | Hydrosalpinx/hematosalpinx/pyosalpinx | Tubo-ovarian abscess | Subserosal/broad ligament fibroid |
Paraovarian cyst | Tubal ectopic pregnancy | ||
Peritoneal inclusion cyst | Degenerated subserosal/broad ligament fibroid |
Table 28.3 classifies primary ovarian tumors according to their cell of origin into epithelial, sex-cord, and germ cell.
Table 28.3
Pathologic classification of primary ovarian tumors
Surface epithelial–stromal | Sex-cord–stromal | Germ cell |
|---|---|---|
Serous | Granulosa cell | Teratoma |
Mucinous | Thecoma | Yolk sac (endodermal sinus) |
Endometrioid | Fibroma | Embryonal |
Clear cell | Sertoli–Leydig cell | Dysgerminoma |
Transitional cell | Choriocarcinoma |
Table 28.4 classifies ovarian tumors according to their type, incidence, age predisposition, and associated serum tumor markers/hormones.
Tumor type | Overall incidence (percentile) (%) | Age predisposition years | Tumor markers |
|---|---|---|---|
Epithelial cell type | 85–95 | (50–70 years: malignant subtype) (10–30 years: benign subtype) | Cancer antigen- 125 (CA-125) |
(a) Serous | 40 | ||
(b) Endometrioid | 20–25 | ||
(c) Mucinous | 20 | ||
(d) Clear cell | 5–8 | ||
(e) Transitional cell | |||
Stromal cell (sex-cord) | 5–8 | ||
(a) Granulosa cell | <5 | No specific age group | Serum inhibin |
(b) Thecoma/fibroma/fibrothecoma | <4 | No specific age group | |
(c) Sertoli–Leydig Cell | <0.5 | <30 | |
(d) Sclerosing Stromal | <0.5 | 10–30 years | |
Germ cell | 3–5 | 10–30 years | |
(a) Teratoma | Mature <45 | Immature: alpha-fetoprotein (AFP) and CA-125 | |
Mature | Immature <20 | SO: thyroxine | |
Immature | Carcinoid: serotonin, | ||
Struma ovarii (SO) | 5-hydroxy-indole acetic acid (5-HIAA) | ||
Carcinoid (very rare) | |||
(b) Yolk sac (endodermal sinus) | 1–3 | 10–30 | AFP |
(c) Embryonal | 3–4 | <30 | AFP and serum human chorionic gonadotropin (hCG) |
(d) Dysgerminoma | 2–5 | <30 | Serum LDH and hCG |
(e) Choriocarcinoma | <1 | <25 years | hCG |
Other | |||
Cystadenofibroma | <1.7 | 15–65 | |
Lymphoma | <1.5 | 30–40 | |
carcinosarcoma | <1 | 50–70 years | CA-125 |
Metastases | 10 |
Imaging Techniques and Utility
Ultrasonography
Ultrasonography, including transvaginal sonography (TVUS), remains the primary modality of choice for evaluating adnexal masses because of its easy availability, high sensitivity, cost effectiveness, and lack of ionizing radiation. It is helpful for differentiating simple benign entities from indeterminate or malignant lesions. Color Doppler sonography may be useful for differentiating benign from malignant neoplasms, with low resistance (low resistive index) flow described in malignant neoplasm [7, 8].
Computed Tomography (CT)
Computed tomography (CT) remains the imaging modality of choice in malignant ovarian neoplasm for evaluating extent of the disease in pretreatment planning, including cytoreduction, and for posttreatment follow-up [9]. CT has a limited role in the primary evaluation and characterization of ovarian lesions except mature teratoma, which can be confidently characterized because of the presence of macroscopic fat and calcifications.
CT of the pelvis should be performed following administration of oral positive contrast to opacify the bowel loops to help delineate the ovaries from the adjacent bowel. Intravenous administration of 125 ml of nonionic iodinated contrast is recommended to be injected at the rate of 3–5 cc’s per second, and images are acquired in the portal venous phase (approximately 60 s after the contrast administration), using 2.5 mm slice thickness on a multislice CT scanner.
Magnetic Resonance Imaging (MRI)
Because of its better tissue characterization, multiplanar capabilities and excellent anatomical delineation, contrast-enhanced MRI helps in differentiating benign from malignant ovarian masses in most cases [10]. MRI is frequently used as a problem solving modality to diagnose indeterminate ovarian and adnexal lesions.
Female pelvis studies are performed using a phased-array surface coil. One milliliter of anti peristaltic agent, such as glucagon, may be administered intramuscularly or intravenously prior to the examination, to limit motion artifacts secondary to bowel peristalsis.
The female pelvis protocol includes a coronal T2-weighted single-shot fast spin echo sequence with wide field of view. Subsequently sagittal and axial images through the pelvis using T2-weighted pulse sequence are acquired. The T1-weighted images are acquired using breath-hold spoiled gradient-echo sequence. Additionally, postintravenous gadolinium contrast-enhanced images are acquired using fat-suppressed T1-weighted spoiled gradient-echo sequence.
Positron Emission Tomography (PET)/CT
Positron emission tomography (PET)/CT is primarily used for treatment planning and follow-up and is not recommended for the primary detection of cancer. Increased metabolic activity has been demonstrated in endometriosis, fibroids, and normal follicles in premenopausal women, thereby limiting PET/CT’s role in cancer staging in this age group [11, 12]. However, increased metabolic uptake in postmenopausal women is always abnormal.
Normal Imaging Appearance of Ovaries
On sonography, normal ovaries in premenopausal women are well-defined, round or oval structures in the adnexae, with isoechoic ovarian stroma in the center and follicles along the periphery that vary in size on the basis of maturation. In premenopausal women, T2-weighted MR images reveal zonal anatomy of the ovaries, demonstrating a hypointense cortex and an intermediate- to high-signal-intensity medulla, which is attributed to loose hypervascular connective tissue [13, 14]. In postmenopausal women, the ovarian stroma (medulla) has decreased T2 signal intensity because of the paucity of follicles and decreased vascular connective tissue [14, 15]. Normal follicles measure <3 cm, are of low signal intensity on T1-weighted images and high signal intensity on T2-weighted images, and have thin enhancing walls on postcontrast T1-weighted MR [15].
Corpus luteum cysts are involuting dominant functional cysts that tend to have irregular walls. When hemorrhage occurs in the corpus luteum, the cysts have intermediate to high signal intensity on T1- and T2-weighted images, depending on the maturity of blood products. The corpus luteal and hemorrhagic cyst may demonstrate mild wall enhancement on post gadolinium contrast images [14, 16].
Algorithmic Approach of Ovarian Masses (Algorithm 28.1)
An algorithmic approach can be followed in order to make a practical differential diagnosis of ovarian masses. This can first start by confirming the ovarian location to differentiate the lesions from any extra-ovarian pathologies. Once the ovarian location is confirmed, morphological features can be used to reach to a specific diagnosis. These morphological features can categorize ovarian lesions as simple cystic, complex cystic, cystic with solid component (mixed cystic/solid) and solid.
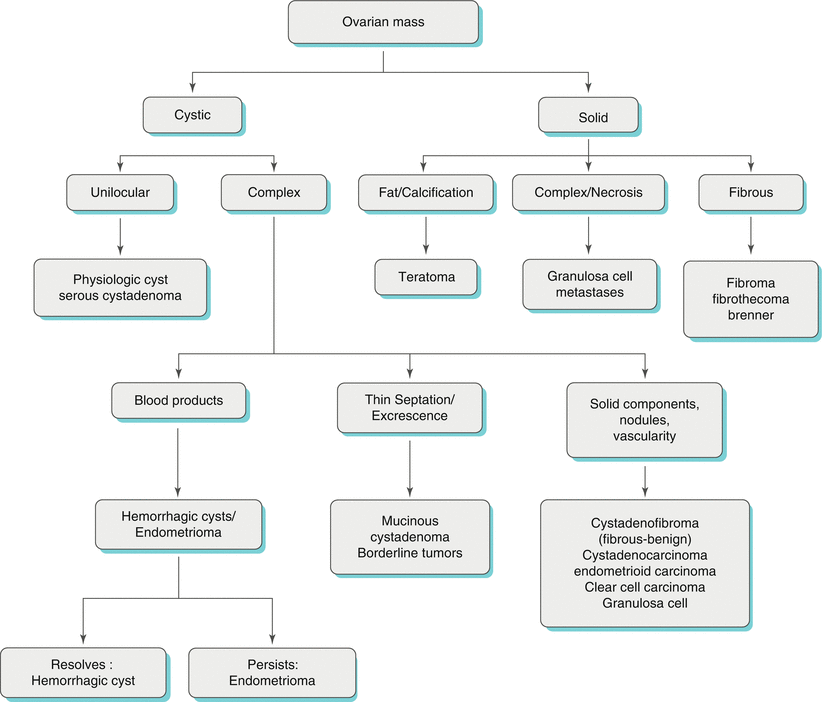

Algorithm 28.1 Imaging differential of ovarian mass
Simple Cystic Adnexal Lesions (Algorithm 28.2)
Adnexal lesions can be entirely cystic without any complex features. These can be categorized as ovarian and extra-ovarian according to location. Ovarian simple cystic lesions include functional cyst and serous cystadenoma. Extra-ovarian simple cystic lesions include paraovarian cyst and hydrosalpinx. Multiple simple ovarian cysts can be seen in polcystic ovaries and ovarian hyperstimulation syndrome.
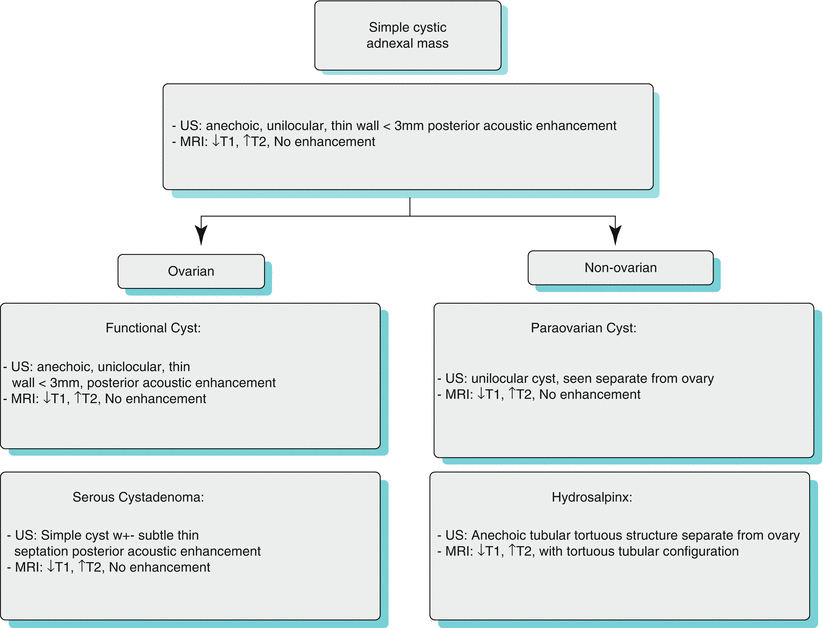

Algorithm 28.2 Imaging differential of simple cystic adnexal masses
Functional and Benign Cysts
By definition, simple cysts are anechoic unilocular and thin walled (<3 mm) on sonography, with posterior acoustic enhancement (Fig. 28.1) [2]. On CT, these appear as well-circumscribed fluid attenuation lesions with no postcontrast enhancement or solid component (Fig. 28.1). On MRI, simple cyst demonstrate low signal intensity on T1-weighted images and high signal intensity on T2-weighted images similar to simple serous fluid (Fig. 28.1). These are commonly found in premenopausal women and are physiologic or functional in nature. Per the consensus statement of the Society of Radiologist in Ultrasound in premenopausal women, simple cysts <5 cm are benign with no further follow-up needed, whereas those 5–7 cm should be followed up yearly. For cysts >7 cm, further characterization with MRI may be helpful [17, 18]. Simple <1 cm cysts are commonly seen in postmenopausal women and are considered clinically unimportant [18]. Simple cyst >1 cm and <7 cm should be followed up annually to document interval changes in size and morphologic characteristics. For cysts >7 cm, further imaging with MRI or surgical evaluation is recommended [18]. Algorithm 28.2 summarizes imaging differential consideration of simple cystic adnexal masses.
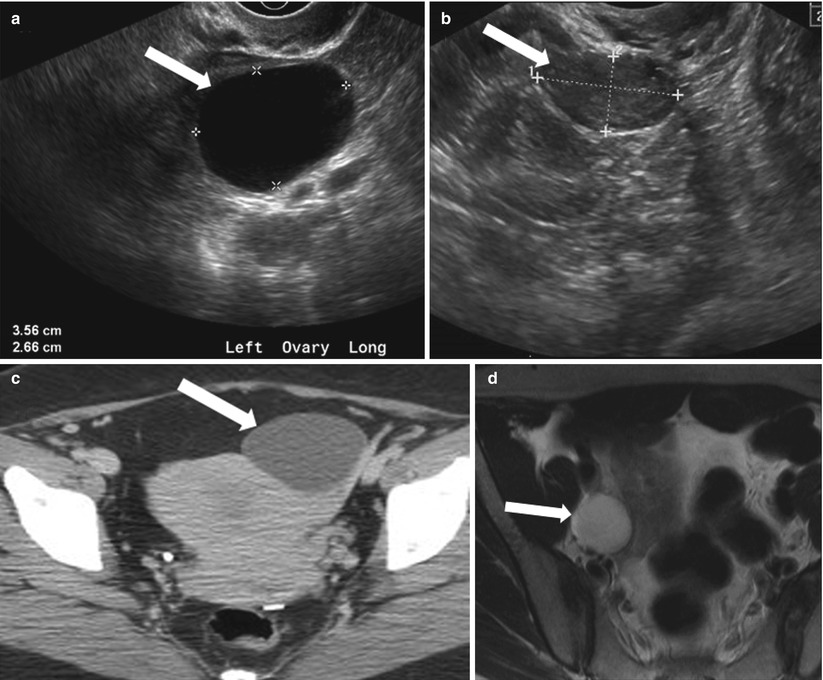
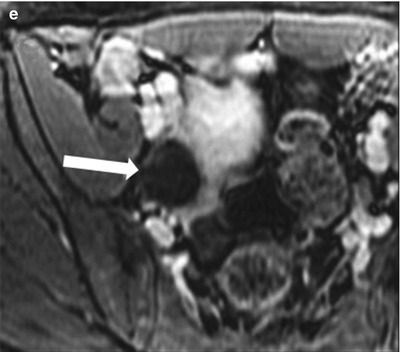


Fig. 28.1
Functional cyst: (a) TVUS showing a thin-walled cyst (arrow) with mild posterior acoustic enhancement, suggesting simple functional cyst. (b) Follow-up ultrasound in 2 months interval demonstrates complete interval resolution. (c) Axial contrast-enhanced CT through pelvis showing a well-defined fluid attenuation cyst in the left adnexa (arrow). (d) Axial fat saturated T2-WI through the pelvis shows a fluid attenuation cyst in the left ovary (arrow) without internal debris or postcontrast enhancement (e)
Polycystic Ovarian Syndrome (PCOS)
It is one of the most common endocrinopathies in women of reproductive age [19], although not all patients with sonographic features of PCO have clinical PCOS, which is characterized by menstrual irregularities, hirsutism, obesity, and sclerotic ovaries. TVUS is currently the gold standard for diagnosing PCO. The classic sonographic feature is an enlarged ovary with ten or more fairly uniform peripherally arranged cysts of 2–8 mm diameter, with an echogenic central stroma [20]. Although, MRI is rarely used in the primary evaluation of PCO as it does not provide any additional information, the best sequence is T2-weighted images in long and short axes of the uterus, demonstrating peripherally arranged, uniformly sized, high-signal-intensity cysts with hypointense central stroma (Fig. 28.2).
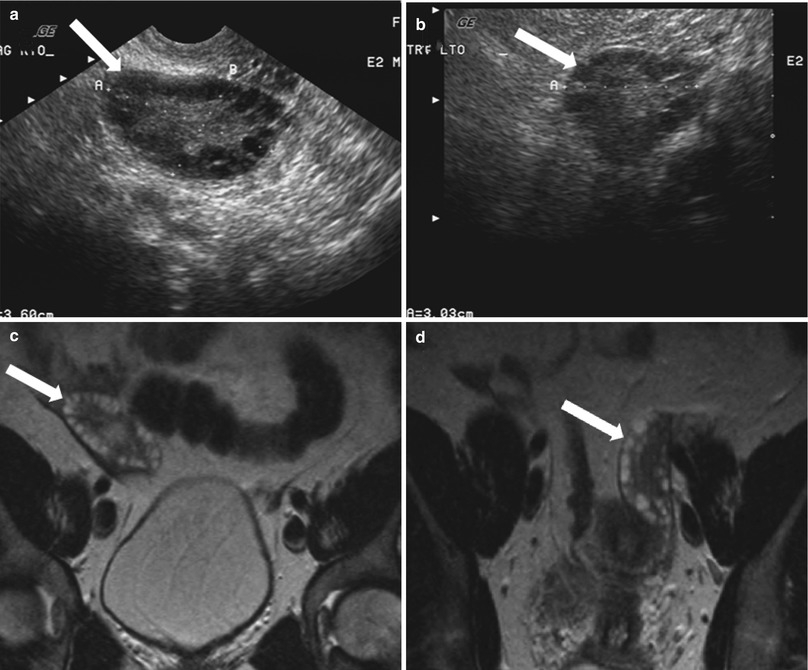

Fig. 28.2
Polycystic ovaries: (a, b) TVUS through the pelvis shows mildly enlarged bilateral ovaries (arrows) demonstrating uniform peripherally arranged follicles. Patient had clinical and laboratory findings suggestive of PCOS. (c, d) Coronal T2-WI through the pelvis shows peripherally arranged small hyperintense follicles (arrows) in both the ovaries
Ovarian Hyperstimulation Syndrome (OHSS)
It is a rare complication seen in patient treated for infertility with ovulation induction drugs. Patients may present with abdominal discomfort, nausea and vomiting [21]. Because of exudation of fluid in the extravascular space in the severe form, it may be associated with pleural effusions, intravascular volume depletion, and consequently hemoconcentration and oliguria [22]. Sonography and MRI demonstrate bilateral enlarged ovaries with multiple large peripherally located cysts, producing a “spoke wheel” appearance (Fig. 28.3) [23]. Patients with OHSS have an increased risk of ovarian torsion.
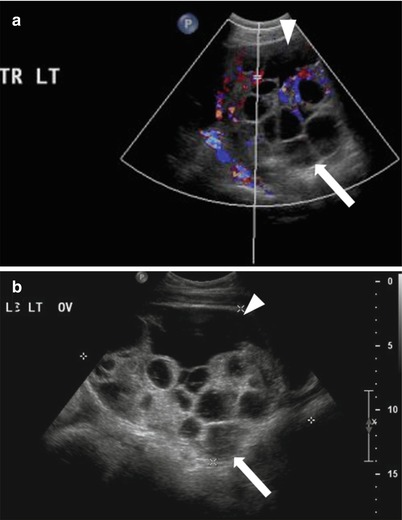

Fig. 28.3
Ovarian hyperstimulation syndrome: TVUS with (a) and without Doppler (b) in a patient undergoing infertility treatment shows enlarged left ovary with multiple prominent and enlarged cysts (follicles) (arrow) and increased vascularity of the stroma, also noted in mild ascites (arrowhead)
Ovarian Serous Cystadenoma
These are common benign epithelial tumors in premenopausal women. On ultrasonography, ovarian cystadenoma are anechoic, thin walled cyst, usually unilocular although rarely may demonstrate thin internal septations. Serous cystadenomas are usually bilateral and smaller than mucinous cystadenoma. They demonstrate homogeneous high signal intensity on T2-weighted images and low signal intensity on T1-weighted images, with no significant postcontrast enhancement of the septae (Fig. 28.4) [24].
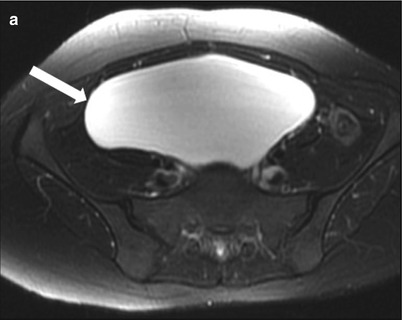
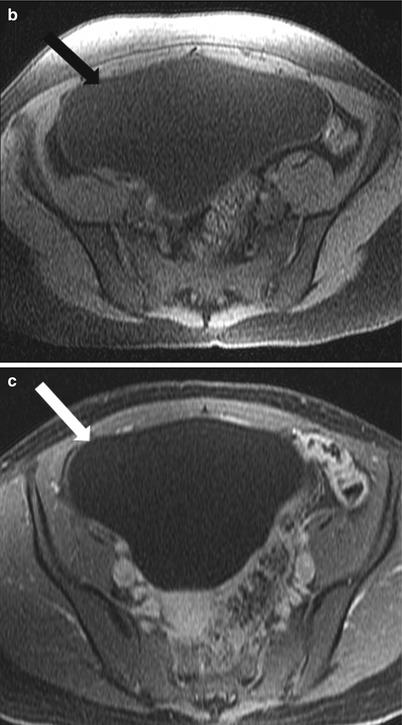


Fig. 28.4
Serous cystadenoma: (a) Axial fat saturated T2-WI through the pelvis shows a well-defined cystic mass (arrow) without internal septation or debris; the mass is hypointense on noncontrast T1-WI (b) and shows no postcontrast enhancement (c)
Paraovarian or Paratubal Cysts
These are located in the mesosalpinx, separate from the ovaries, hence the name. They have been described to be arising from mesothelial, paramesonephric (Müllerian), and mesonephric (Wolffian) structures [4]. The paraovarian cyst demonstrate imaging features similar to simple cyst on ultrasonography and MRI (Fig. 28.5).
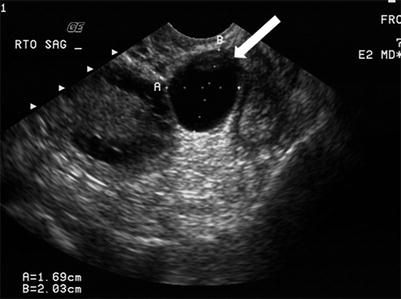

Fig. 28.5
Paraovarian cyst: transvaginal grayscale ultrasound demonstrates a well-circumscribed small anechoic paraovarian cyst (arrow) seen separate from the adjacent ovary (O)
Hydrosalpinx (See Below with Hematosalpinx and Pyosalpinx)
Algorithm 28.3 demonstrate imaging differentials of complex cystic ovarian lesion masses.
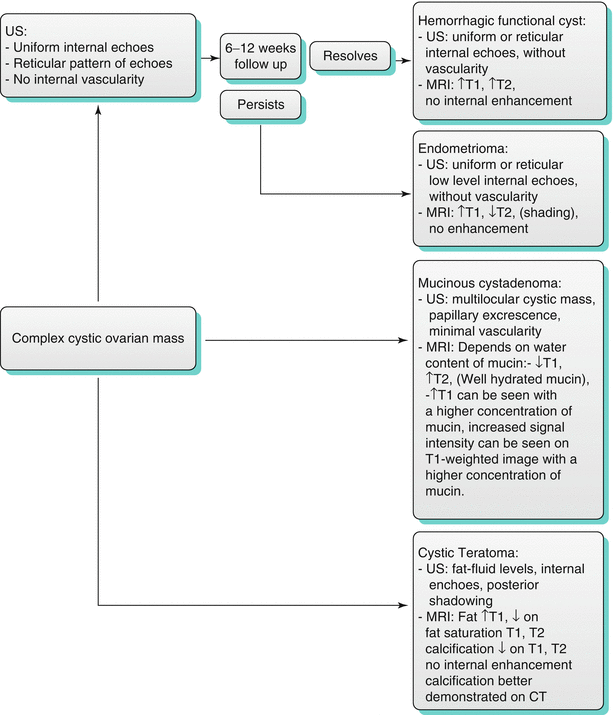
Algorithm 28.3 Imaging differentials of complex cystic ovarian lesion masses
Hemorrhagic Cysts
These are usually functional cysts undergoing hemorrhage or corpus luteum cysts. In premenopausal women, cysts with uniform internal echoes, reticulations, or septations may represent hemorrhagic functional cysts or endometrioma. A follow-up sonogram should be performed in 6–12 weeks in these cases [18]. Functional hemorrhagic cysts demonstrate complete interval resolution, whereas endometrioma persists or even slightly increases in size at intervals. Hemorrhagic corpus luteum cysts have a characteristic appearance of blood products, manifested by relatively high signal intensity on T1-weighted images and intermediate to high signal intensity on T2-weighted image (Figs. 28.6 and 28.7); this may lead to difficulty in differentiating them from endometrioma if no prior images are available [14, 25, 26].
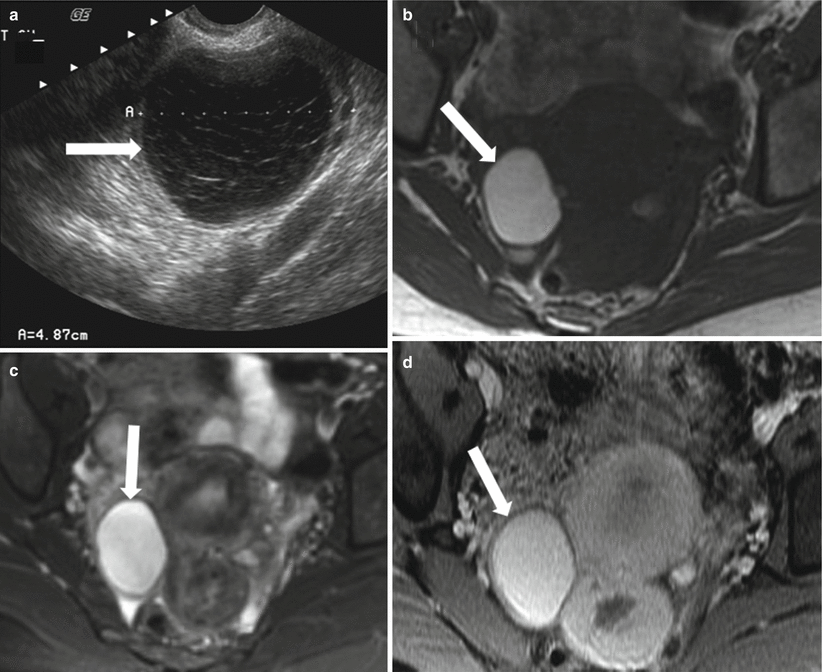
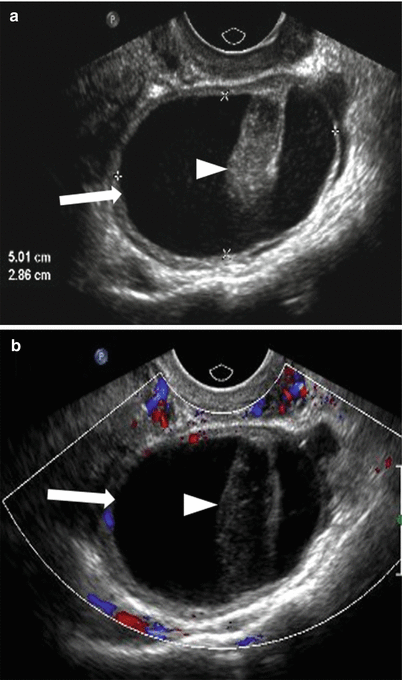

Fig. 28.6
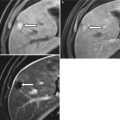
Hemorrhagic ovarian cyst: (a) TVUS shows a well-defined right ovarian lesion (arrow) with internal echoes and reticulations suggesting hemorrhagic cyst. (b) Axial T1-WI shows the lesion to be well defined (arrow) and remains high signal intensity on the fat-saturated T2-WI (c) and fat-saturated postcontrast T1-WI (d) compatible with acute/subacute hemorrhagic cyst

Fig. 28.7
Hemorrhagic ovarian cyst: (a) Grayscale and (b) color doppler TVUS shows a complex ovarian cyst (arrow) with mildly thickened wall and non-vascular echogenic clot (arrowhead) which may be mistaken for solid lesion
Endometrioma
Endometriosis is the ectopic implantation of endometrial tissue outside the uterus, with the ovaries being the commonest site. Sonographically, endometrioma are complex cystic masses with uniform low-level internal echoes and may contain hyperechoic foci secondary to blood clots in the periphery [2]. MRI usually reveals high signal intensity on T1-weighted images (light bulb) and intermediate to low signal intensity on T2-weighted images (known as T2 shading), which is attributed to hemoglobin degradation products in various stages (deoxyhemoglobin, methemoglobin, and iron products) and decreased free water content (Fig. 28.8) [27, 28]. High signal intensity on T1-weighted images can be seen secondary to fat or blood, and a persistently high signal on fat-saturated T1-weighted imaging confirms the absence of fat in the lesion. Other MRI findings may include high signal intensity on T1- and T2-weighted images and adhesion or tethering to adjacent structures. The presence of multiple high-signal-intensity foci on T1-weighted images in both ovaries also favors the diagnosis of endometriosis.
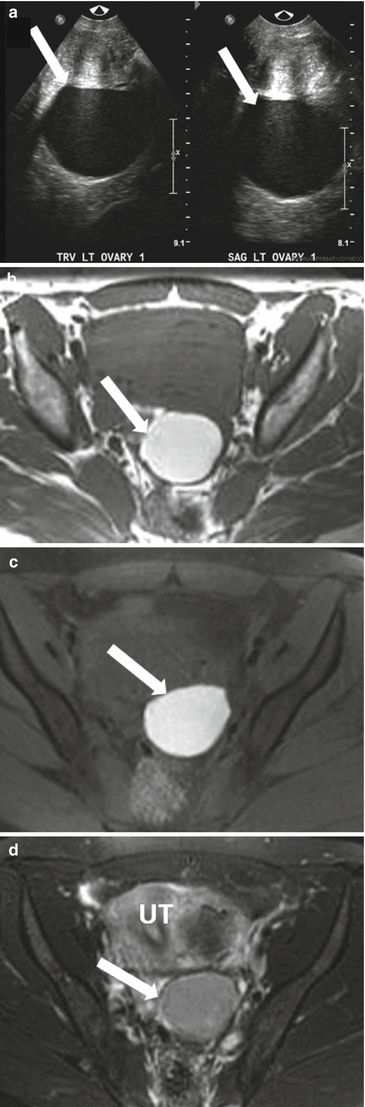

Fig. 28.8
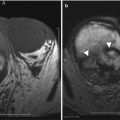
Endometrioma: (a) TVUS shows well-defined hypoechoic lesion in the left ovary with uniform low-level internal echoes which was unchanged in appearance from the prior ultrasound (not shown), raising the possibility of endometrioma. The lesion (arrow) shows high signal intensity on T1-weighted image (b) and remains high signal on fat saturated T1-WI (c). The lesion shows uniform low signal intensity on fat saturated T2-WI (T2 shading) (d) confirmed as endometrioma on surgery. UT uterus
Ovarian Torsion
Ovarian torsion results from partial or complete twisting of the vascular pedicle, causing obstruction to the vascular supply of the ovary. It is mostly seen in premenopausal women (can occur in post-menopausal women with large ovarian mass) and usually presents with acute-onset abdominal or adnexal pain. Enlarged ovaries from cysts or masses (>5 cm) remain the predisposing factor, the commonest cause being cystic teratoma [29, 30]. On sonography, ovarian torsion manifests as complex enlarged adnexal mass showing peripherally arranged follicles secondary to displacement by the edematous ovarian stroma [31]. Color Doppler sonographic evaluation is important, although the features depend on the degree of vascular compromise and may range from decreased to completely absent vascular flow (Fig. 28.9) [32]. The presence of central venous flow has been described with spontaneous partial detorsion and are shown to have better outcomes [33]. CT and MRI are rarely used for diagnosis and may reveal nonspecific features such as enlarged adnexal masses, engorged adnexal vessels, and ascites.
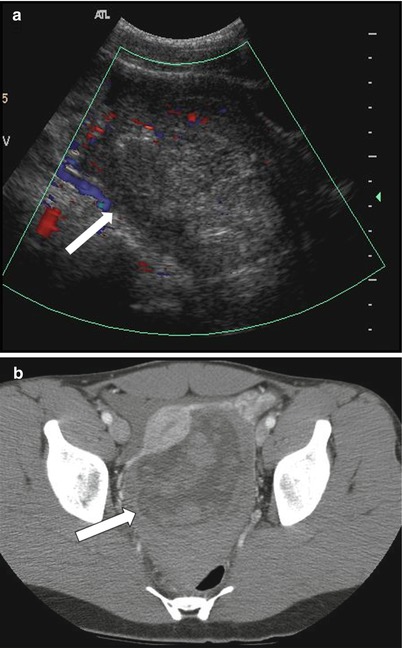

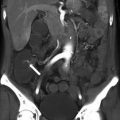
Fig 28.9
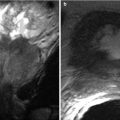
Ovarian torsion in a young female with acute-onset right pelvic pain: (a) Color Doppler transabdominal ultrasound shows enlarged heterogeneous right ovary with minimal internal vascularity (arrow). (b) Axial contrast-enhanced CT shows the large heterogeneous mass in the pelvis (arrow). This was confirmed as a benign serous cyst, and ovarian parenchyma with marked hemorrhage and edema, consistent with torsion
Mucinous Cystadenomas
These are benign epithelial tumors of the ovaries that present as multilocular cysts (honeycomb-like locules) with thin regular walls and septa with no endocystic or exocystic vegetation or significant postcontrast enhancement. The MRI signal intensity of mucinous cystadenoma depends on the hydration of the mucin: with the watery (hydrated) mucin presenting as low signal intensity of T1-weighted images and high signal intensity on T2-weighted images (Fig. 28.10), while dehydrated thicker mucin results in slightly higher T1- and lower T2-signal intensity [2].
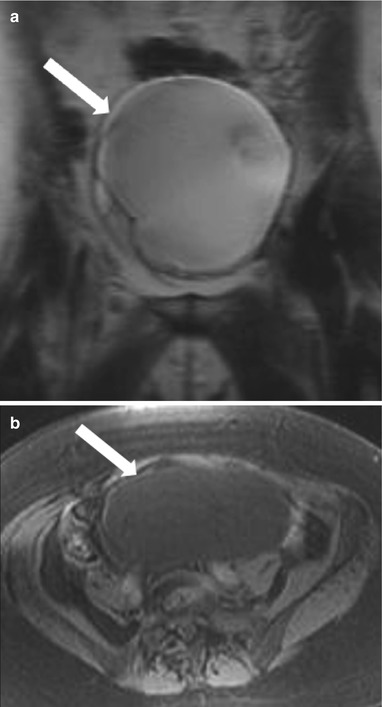
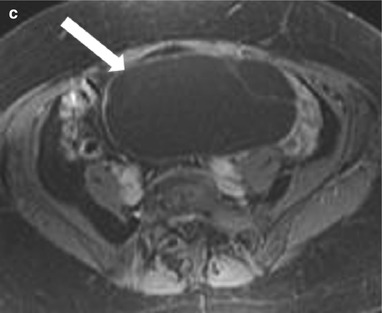


Fig. 28.10
Ovarian mucinous cystadenoma: (a) Coronal T2-WI shows a high-signal-intensity well-defined large cystic lesion (arrow) in the pelvis with thin internal septae but no soft tissue or solid component. The cyst is of low signal intensity on fat saturated noncontrast T1-WI (b) and shows no enhancing component on postcontrast T1-WI (c)
Mature Cystic Teratomas
These are common ovarian tumors that are composed of tissue from all three embryogenic germ cell layers and thus are composed of sebaceous material, hair follicles, osseus structure/tooth, and muscles. On sonography, their appearance varies from purely cystic lesions with peripheral nodules to partially echogenic masses with posterior shadowing from adipose material, calcifications, and hair. The hair ball in the teratoma may present as multiple thin echogenic bands [34]. CT demonstrates characteristic fat attenuation, with or without calcification in the wall [35, 36]. On MRI, the sebaceous components have high signal intensity on T1-weighted imaging, similar to that of retroperitoneal fat, and lose signal on fat-suppressed T1-weighted sequences, suggesting macroscopic fat and excluding hemorrhage. Low signal intensity on T1- and T2-weighted images may be secondary to soft tissue protuberances from Rokitansky nodules and calcifications (teeth) (Fig. 28.11). Complications include torsion in large size teratomas and rupture, causing leakage of the sebaceous component into the peritoneum and granulomatous peritonitis.
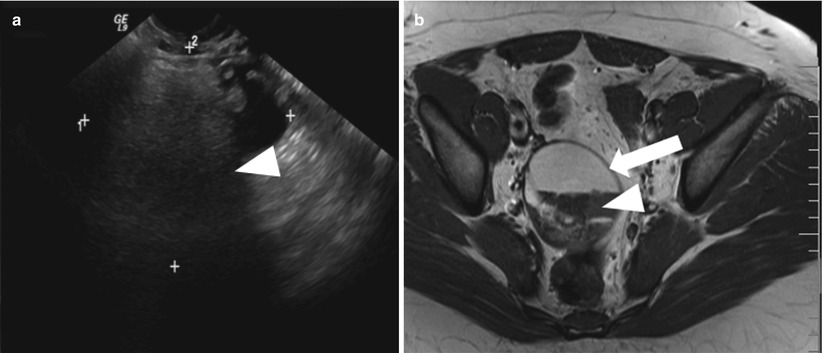
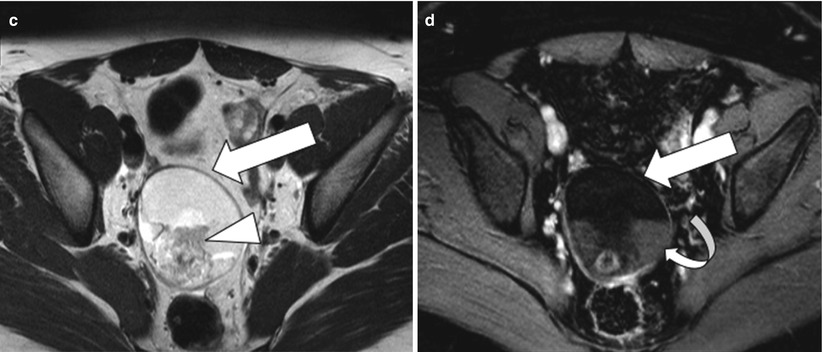


Fig. 28.11
Mature cystic teratoma: (a) TVUS shows a complex right adnexal mass with posterior acoustic shadowing (arrowhead). (b) T2-weighted image and (c) nonfat saturated T1-weighted image demonstrate nondependent high signal intensity in the mass (arrow) which could represent fat or blood and a hypointense foci (arrowhead) suggesting calcification. (d) Fat-saturated postcontrast T1-weighted image shows loss of signal in the nondependent area confirming fat and persistent hypointensity in the dependent foci consistent with calcification with non-enhancing soft tissue (curved arrow) suggesting Rokitansky nodule
Immature Cystic Teratoma
These are malignant counterpart of teratoma and constitute less than 1 % of all teratoma, commonly encountered in the first two decades of life [37, 38]. These tumors contain immature tissue from all three germ cell. These tumors tend to grow fast and are usually very large at time of presentation and can present with perforation and rupture of the capsule [3]. On imaging, these tumors are seen as large complex masses with cystic and solid components with scattered calcifications (Fig. 28.12).
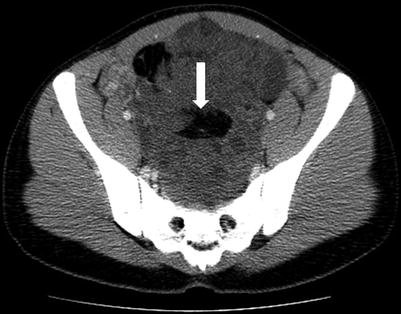

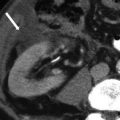
Fig. 28.12
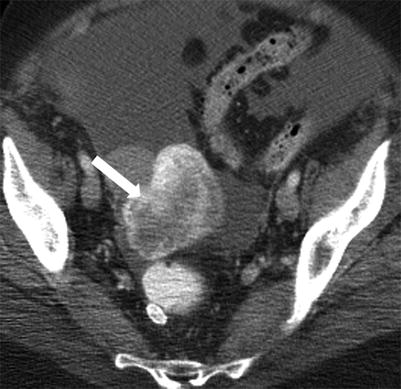
Immature teratoma: axial contrast-enhanced CT in a young female shows the large heterogeneous solid mass in the pelvis with central fat attenuation foci (arrow), confirmed on surgery to be immature teratoma
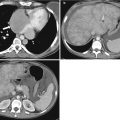
Struma Ovarii (SO)
SO or also called ovarian goiter is a rare ovarian tumor included in the subtypes of teratoma. It constitutes 2.7 % of ovarian teratoma and is described to occur in third to seventh decade [37]. Majority of SO are benign with only 5 % being malignant [39]. Clinically, they may present as nonspecific abdominal symptoms. Symptoms of hyperthyroidism have been described. Malignant forms are associated with ascites and pseudo-Meigs syndrome. There is an increase in CA-125 and serum thyroid hormone levels. CT appearance of SO may include solid, cystic, or mixed characteristics. A feature that has been described on CT to be associated with SO is high density cavities, which have been attributed to thyroglobulin and thyroid hormones (Fig. 28.13) [40]. On MR, SO shows complex fluid signal with low or high signal on T1-WI and on T2-WI [39]