|
Identify the sonographic role in the evaluation of the adrenal glands.
Describe the embryologic development of the adrenal glands.
Discuss the anatomy and physiology of the adrenal cortex and medulla.
List the hormones secreted by the adrenal cortex and medulla.
Identify conditions caused by hyposecretion and hypersecretion of adrenal hormones.
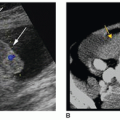
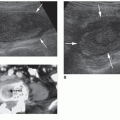
Identify the normal sonographic appearance of the adrenal glands.
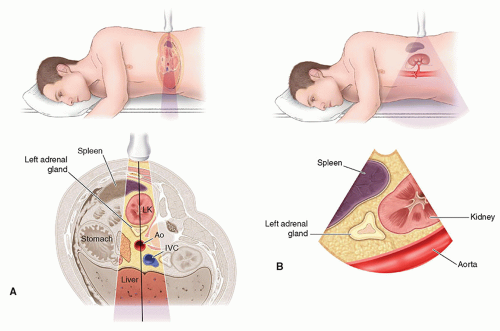
Describe adrenal gland scanning technique, patient positions, and scanning pitfalls.
Discuss the differential diagnosis for solid adrenal masses.
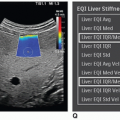
Discuss alternative imaging modalities used to evaluate the adrenal glands.
outer parenchyma of the adrenal gland that makes up 90% of the organ’s weight and secretes corticoids including cortisol and aldosterone
inner portion of the adrenal gland that secretes the catecholamines epinephrine and norepinephrine
hormone secreted by the pituitary gland that causes the adrenal gland to produce and release corticosteroids
an ultrasound transducer on a thin flexible endoscope is inserted in the mouth or anus to visualize the walls of the upper or lower digestive tract and surrounding organs
a group of autosomal dominant disorders characterized by benign and malignant tumors of the endocrine glands
of the normal gland as well as adrenal pathology. As a sonographer, it is important to develop critical-thinking and problem-solving skills for sorting out the origin and extent of abdominal masses, including those of suprarenal and retroperitoneal origin. Related techniques are discussed. In addition, EUS, IOUS, computed tomography (CT), magnetic resonance imaging (MRI), and associated nuclear medicine imaging procedures are presented as related to adrenal imaging. Owing to technical improvements in all the diagnostic imaging modalities and the increased use of these modalities, more clinically silent or unexpected masses are being detected. The general term for an unexpected mass detected during an imaging procedure being performed for unrelated disease is “incidentaloma.” This term may be applied to any unexpected mass occurring anywhere in the body. Adrenal incidentaloma (AI) has become a common term in the medical literature and represents a diagnostic challenge that includes discovery of masses, which range from benign, nonfunctional lesions to malignant tumors such as pheochromocytomas, adrenocortical carcinomas, or metastases from other primary cancers.8 Sonographers have a role both in the initial detection of AIs and in gathering diagnostic information regarding newly discovered AIs.
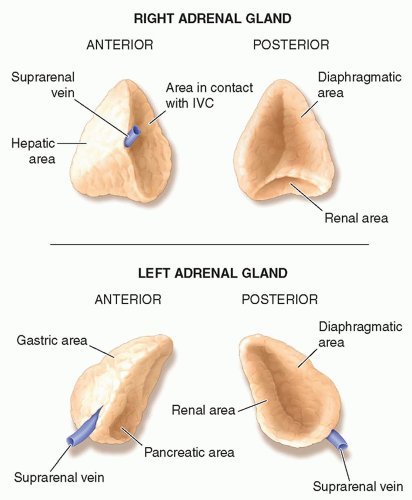
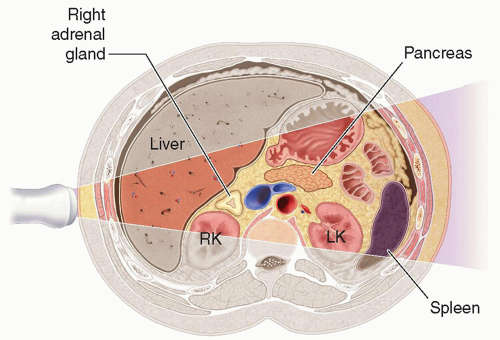
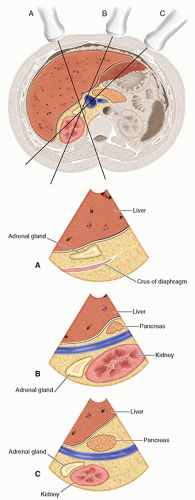
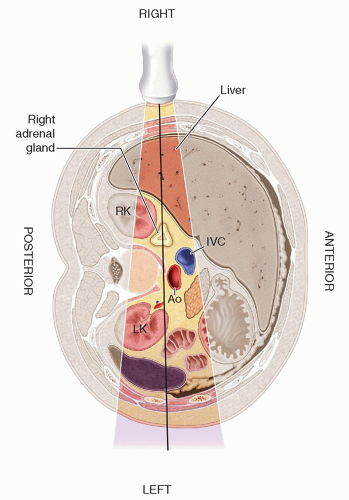
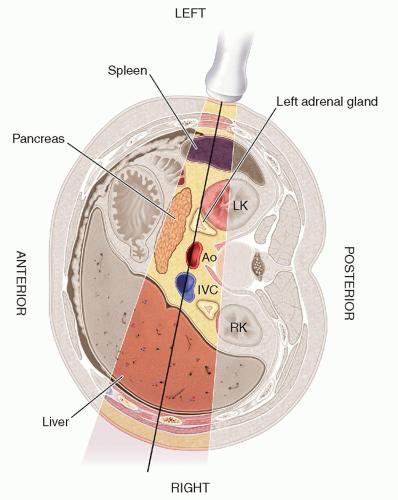
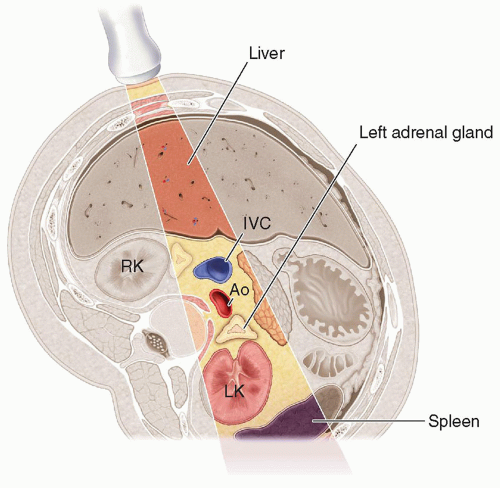
the liver and lateral to the crus of the diaphragm.9,10,13,15,16 The right adrenal gland has been described as sitting like a triangular cap on the anterior, medial, and superior aspects of the superior pole of the right kidney.
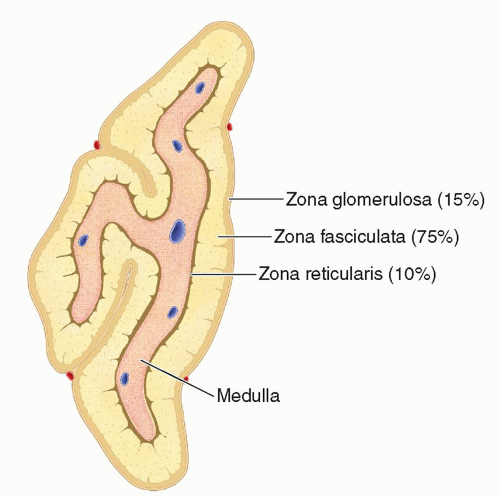 FIGURE 15-1 Adrenal gland. The anatomic sectional illustration of the adrenal gland demonstrates the medulla surrounded by three differentiated zones of the cortex. |
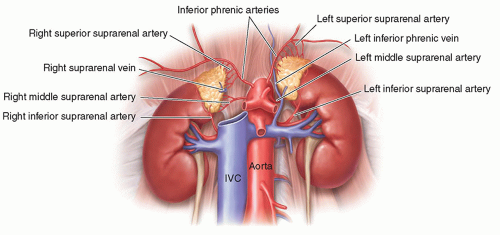
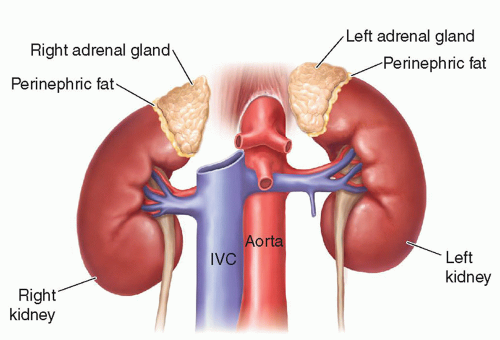 FIGURE 15-2 Relational anatomy. The illustration demonstrates the anteromedial relationship of the adrenal glands to the kidneys. IVC, inferior vena cava. |
| ||||||||||||||||||||
|
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree