The Gallbladder and Biliary System
Teresa M. Bieker
|
OBJECTIVES
Illustrate surface, relational, and internal anatomy of the normal gallbladder and biliary system.
Discuss the embryologic development, common anatomic variants, and congenital anomalies of the gallbladder and biliary tree.
Describe the physiology of the gallbladder and biliary tree and include the laboratory values associated with normal and abnormal function.
Explain the sonographic evaluation of the gallbladder and biliary tree to include patient preparation, protocol, and demonstrate completing the examination procedure.
Describe the embryologic development, clinical signs and symptoms, and sonographic appearance for each of the following congenital anomalies: septate gallbladder, interposition of the gallbladder, biliary atresia, and choledochal cyst.
Identify gallbladder pathology in terms of etiology, clinical signs and symptoms, and sonographic appearance for acquired diseases to include biliary sludge, cholelithiasis, acute cholecystitis, acute acalculous cholecystitis, complicated cholecystitis, chronic cholecystitis, wall thickening, cholestasis, neoplasms, hyperplastic cholecystoses, and miscellaneous pathology.
Identify biliary system pathology in terms of etiology, clinical signs and symptoms, and sonographic appearance for acquired diseases to include postcholecystectomy, bile duct obstruction, cholangitis, other pathology, and AIDS cholecystopathy.
Differentiate between the advantages and disadvantages of utilizing other gallbladder and biliary system imaging procedures to include radiography, nuclear medicine, computed tomography, and magnetic resonance.
KEY TERMS
acquired diseases
alanine aminotransferase (ALT)
aspartate aminotransferase (AST)
bilirubin
cholecystokinin (CCK)
congenital anomalies
extrahepatic biliary system
gallbladder
GLOSSARY
cholangitis inflammation of the bile ducts
cholecystectomy surgical removal of the gallbladder
cholecystitis acute or chronic inflammation of the gallbladder
cholecystokinin a hormone secreted into the blood by the small intestine that stimulates gallbladder contraction
choledocholithiasis calculi within the bile duct
cholelithiasis the formation or presence of calculi or bile stones within the gallbladder
common bile duct the duct that carries bile from the cystic and hepatic ducts to the duodenum
cystic duct the duct of the gallbladder that joins with hepatic duct to form the common bile duct
gallbladder a pear-shaped sac that lies on the undersurface of the liver; the gallbladder holds bile from the liver until released through the cystic duct
junctional fold a fold within the gallbladder neck or body
phrygian cap a fold within the gallbladder fundus
pneumobilia air within the bile ducts
sludge solid, semisolid, or thickened bile within the gallbladder or bile ducts
sonographic Murphy sign pain over the gallbladder when the ultrasound transducer is used to compress the right upper quadrant
Sonography plays a key role in the evaluation of suspected gallbladder and biliary disease. Because the quality of sonographic examinations is strongly operator-dependent, it is crucial to understand the anatomy, physiology, pathology, techniques, and pitfalls of scanning these structures. The quality of education and the experience of a sonographer are directly related to accuracy of findings.1 This is especially true of the gallbladder and biliary tree because scanning may require patience, skill, and appropriate technique. Sonography is considered the modality of choice to evaluate gallbladder and ductal pathology.
ANATOMY
The normal distended gallbladder is a pear- or teardrop-shaped sac measuring approximately 8 cm long and 4 to 5 cm in anteroposterior (AP) and transverse diameter. The normal wall measures less than 3 mm in thickness.1,2 The gallbladder is located in the main lobar fissure between the right and left hepatic lobes (Fig. 8-1). It lies under the visceral surface of the liver, lateral to the second part of the duodenum, and anterior to the right kidney and transverse colon3 (Fig. 8-2A, B).
The gallbladder is divided into a neck, body, and fundus (Figs. 8-2B and 8-3A, B). The narrowest portion is the neck, which lies to the right of the porta hepatis. The body is the central or main portion. The fundus varies considerably in position. Normally, the fundus is the most inferolateral portion of the gallbladder and it extends caudally and anteriorly below the inferior margin of the right hepatic lobe; however, it can extend as low as the right lower quadrant (RLQ) or as far left as the left anterior axillary line.3
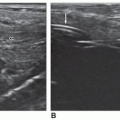
 FIGURE 8-1 Longitudinal image of the normal gallbladder (GB). Note the anatomic landmarks: the main lobar fissure (arrow, MLF) and the right portal vein (RPV). |
Histologically, the gallbladder consists of an inner epithelial mucosa with folds, a muscular layer, a subserous layer, and an outer serosal surface. Mucous glands are found only in the gallbladder neck. Aberrant vestigial bile ducts of the liver may enter the adventitia (outermost covering) of the gallbladder and serve as a pathway for infection from the liver.4
The cystic duct arises from the superior aspect of the gallbladder neck and enters into the common bile duct (CBD) (Fig. 8-4).2,3 It is 2 to 6 cm in length and its lumen contains a series of mucosal folds, the spiral valves of Heister, which prevent collapse or overdistention during sudden changes in position.3
The intrahepatic bile ducts run in juxtaposition to the portal veins and hepatic arteries. Together, these three structures form portal triads. The portal triads are surrounded by connective tissue and radiate through the lobes and segments of the liver. This fibrous connective tissue lines the portal vein walls, creating an echogenic appearance on sonography. The intrahepatic ducts join to form the right and left main hepatic ducts. The right and left main ducts unite at the porta hepatis to form the common hepatic duct (CHD). The cystic duct joins the CHD, forming the CBD. The CBD courses inferiorly within the hepatoduodenal ligament and anterior to the portal vein to the first portion of the duodenum and the head of the pancreas. In some cases, the CBD is surrounded by pancreatic tissue (Fig. 8-5A-D). The duct ends at the ampulla of Vater, which is difficult to visualize sonographically. Ducts can vary in their course, length, and site of anastomosis. For example, the CBD can be straight, curved, or angled.2,3 There can also be accessory hepatic ducts.3,4 Anatomically, the proximal duct is located at the liver, whereas the distal duct is located at the bowel. Central refers to the porta, and peripheral is the branching within the liver.2
On sonography, the duct is measured inner wall to inner wall. A normal intraluminal measurement for the intrahepatic duct is 2 mm, or no more than 40% of the portal vein.2 Measurements for the CBD and CHD are controversial; however, the CHD typically does not exceed 6 mm, and the CBD should measure less than 7 to 8 mm (Fig. 8-6A-D).5,6 There is also controversy over whether duct size increases with age or after a cholecystectomy. Normal diameters of up to 10 mm have been reported in asymptomatic populations.2
SECTIONAL VIEWS
Anatomic structures are sonographically identified by location and relationships with other structures. The schematic views with corresponding sonographic sectional images of the gallbladder and extrahepatic biliary tree demonstrate this relationship (Figs. 8-7A-C, 8-8A, B, 8-9A-C, and 8-10A, B).
PHYSIOLOGY
The biliary system transports bile, which is produced continually by hepatic parenchymal cells, to the duodenum, where it aids in digestion. Bile contains bile pigments (chiefly bilirubin), bile acids, cholesterol, lecithin, mucin, and other organic and inorganic substances. Bile helps to emulsify and promote the absorption of fats, and it also facilitates the actions of lipase, a pancreatic enzyme. The gallbladder concentrates and stores bile until needed and regulates biliary pressure.3,4
When food, especially fats, enters the small intestine, cholecystokinin (CCK) is secreted by the proximal small intestine, causing the gallbladder to contract and the sphincter of Oddi to relax. Bile is then released into the cystic duct, flows through the CBD, and enters the duodenum.4 Gallbladder contraction can also be induced by commercially available “fatty meals” or by intravenous (IV) injections of CCK, although these methods are not commonly utilized. Gallbladder emptying may be diminished in some patients with gallstones. Residual gallbladder volume is known to increase during pregnancy. Sonography can monitor such gallbladder kinetics by measuring the gallbladder volume in various fasting and postprandial states.7
Several laboratory tests can be helpful in evaluating pathophysiology of the biliary tract. An increased WBC indicates infection. Aspartate aminotransferase (AST) and alanine aminotransferase (ALT) are enzymes produced by tissues of high metabolic activity, including the liver. Both values, but particularly the latter, can be mildly to moderately elevated in biliary obstruction. Lactic dehydrogenase (LDH), an enzyme, can be mildly elevated in obstructive jaundice. Alkaline phosphatase, another liver enzyme, markedly increases in obstructive jaundice. Bilirubin results from the breakdown of hemoglobin in red blood cells. Direct, or conjugated, bilirubin level tends to be elevated in obstructive (surgical) jaundice, whereas the indirect, or unconjugated, bilirubin level rises in hepatocellular disease and hemolytic anemias.3,8 Although helpful, the results of liver function tests can be nonspecific and must be considered with the clinical presentation and the findings of diagnostic imaging. This can help identify trends; therefore, it is important to evaluate the laboratory results over time to determine if function is improving or deteriorating.
SONOGRAPHIC EXAMINATION, PREPARATION, PROTOCOL, AND PROCEDURE
Ideally, patients should not have anything by mouth for 6 to 8 hours prior to an examination of the gallbladder and biliary tree. Clear liquids are accepted. Fasting distends the gallbladder and reduces bowel gas for optimal visualization. The diagnosis of various gallbladder and ductal pathologies can be made with a partially contracted, nonfasting gallbladder in emergent situations, when the patient is not fasting.
The chief complaint and pertinent medical or surgical history should be verified with the patient. This includes the type, frequency, and duration of symptoms; location of pain; factors that aggravate or alleviate symptoms; prior similar episodes; and previous surgery or medical illnesses. Additional information, such as previous imaging studies, laboratory work, or clinic notes, is also helpful. The patient should also be evaluated physically. Conditions such as jaundice and/or surgical scars should be noted.
The normal gallbladder has thin echogenic walls, an anechoic lumen, and posterior enhancement. The bile duct lumen should also appear anechoic; therefore, proper technique is important to avoid the presence of artifact filling these structures. The gallbladder is located in the main lobar fissure to the right of the ligamentum teres, anterior to the right kidney and lateral to the pancreatic head. Although its position can vary, the neck has a constant relationship to the region of the porta hepatis (Fig. 8-3A). The CBD is usually identified anterior to the portal vein and hepatic artery at the porta hepatis and should be followed throughout its course to the pancreatic head. The CHD and the right and left intraductal branches should also be evaluated. Dilated intrahepatic biliary ducts can be identified along the intrahepatic portal vein branches. The gallbladder and ducts are carefully evaluated for size, wall thickness, contents, course,
and caliber. The presence or absence of pathology in the gallbladder, porta hepatis, and intrahepatic and extrahepatic biliary system should be documented.
and caliber. The presence or absence of pathology in the gallbladder, porta hepatis, and intrahepatic and extrahepatic biliary system should be documented.
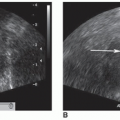
Meticulous real-time examination of the gallbladder and bile ducts should be performed in all scan planes. A 3.5-MHz probe or higher-frequency probe should be utilized. In thinner patients, a 7.5-to-9-MHz probe can be used for optimal resolution. Proper setting of the overall gain, the time gain compensation, compression, spatial compounding, and dynamic range should be optimized for adequate and accurate visualization of the gallbladder. Using harmonics is also helpful in reducing artifacts within the gallbladder as well as identifying small stones (Fig. 8-11A, B).5,9 The focal zone should be adjusted for each area of interest. The focal zone is the narrowest segment of the beam, and suspected calculi (or other pathology) should lie within this zone to demonstrate shadowing. Even then, many small calculi may not shadow. Changing the transducer frequency or angle may be necessary to bring the gallbladder into the focal zone. Also, removing compound imaging may help to visualize a shadow posterior to a small stone (Fig. 8-12).
The patient should be examined in two positions—typically supine and left lateral decubitus (LLD) or posterior oblique. A right lateral decubitus (RLD), erect (sitting or standing), or even prone positions may be necessary. While in the decubitus positions, small changes in the patient’s angle, from 45 to 90 degrees, may improve visibility. The erect positions demonstrate gravity dependence. A prone position can show the mobility of stones and allows the liver to fall anteriorly, thereby providing an acoustic window and displacing bowel. Because the prone scanning position may be awkward, the technique may be varied by turning the patient prone for 10 to 15 seconds and then quickly returning the patient to an LLD position and rescanning the area.
Depending on the patient’s body habitus and ability to cooperate, breathing techniques may also be beneficial. Suspended, full inspiration is often best, but sometimes, varying degrees of inspiration or expiration may also help. Breathing techniques are useful in moving organs inferiorly for improved subcostal access.
Another technique is to vary the scanning approaches. With the patient in an LLD position, scan should be performed subcostally with the transducer angled slightly toward the patient’s right shoulder to elongate the portal vein and the bile duct at the porta hepatis. If this technique is not optimal, intercostal scanning may be necessary. Structures should be evaluated by scanning through many different windows to achieve the best angle and resolution. Gentle transducer pressure is useful often for pushing bowel away from the field of view.
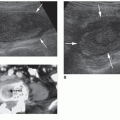
 FIGURE 8-8 The sonogram (A) and illustration (B) demonstrate a longitudinal image through the gallbladder (GB) body and fundus. The portal vein is just posterior to the gallbladder. |
 FIGURE 8-10 The sonogram (A) and illustration (B) demonstrate a longitudinal scanning plane through the common bile duct (CBD). CHD, common hepatic duct; HA, hepatic artery; PV, portal vein. |
Owing to absorption, gallstones should produce a clean shadow. If echogenic foci are seen within the gallbladder and do not shadow, several techniques should be attempted. First, the gain distal to the foci should be reduced. Second, the frequency of the transducer should be increased. Third, the scanning angle should be changed to decrease the distance between the ultrasound beam and the stone. When a focal zone is used, the focus should be placed at or just below the stone. However, new technologies allow for and create multi focal images. Removing compound imaging may also be helpful to visualize a shadow posterior to a small stone.
While scanning, it is also important to determine if there is a sonographic Murphy sign. To evaluate, transducer pressure should be applied directly over the gallbladder. When positive, the patient will have focal pin-point tenderness. Care must be taken to ensure pressure is placed
directly over the gallbladder and not the epigastrium or the liver. If the patient has received pain medication or is unresponsive, the sonographic Murphy sign will not be accurate.5
directly over the gallbladder and not the epigastrium or the liver. If the patient has received pain medication or is unresponsive, the sonographic Murphy sign will not be accurate.5
 FIGURE 8-11 Sonographic images of the gallbladder (A, left) without harmonics and (B, right) with harmonics on the same patient. Note the decrease in artifact within the gallbladder lumen. |
 FIGURE 8-12 Sonographic images of the gallbladder (A, left) with compound imaging and (B, right) without compound imaging. Note the posterior shadow once compound imaging was renewed. |
It is also valuable to interrogate the gallbladder and biliary structures with color Doppler imaging. This may be helpful in evaluating hyperemia in inflammatory conditions, in distinguishing solid masses from avascular pathology, and in differentiating intrahepatic and extrahepatic bile ducts from blood vessels.2
Meticulous scanning technique is crucial and can decrease or eliminate the need for other diagnostic tests.
CONGENITAL ANOMALIES AND NORMAL VARIANTS
There are many common gallbladder variations, including different shapes (e.g., hourglass), positions, folds, and/or septations (Fig. 8-13A, B). The gallbladder may occasionally contain a small infundibulum at the neck, Hartman pouch, where stones can collect. The phrygian cap is a common variant that forms when the fundus kinks or folds back on the body (Fig. 8-14). The gallbladder can be excessively mobile, ectopic (on the left, midline, and transversely), or low in the RLQ. It can also be located partially or totally embedded in the liver parenchyma, completely surrounded by peritoneum, in the abdominal wall or falciform ligament, contained in the retroperitoneum, or above the liver.2, 3 and 4
Embryonic development of the liver, gallbladder, and biliary duct system arises from the hepatic diverticulum of the foregut in the fourth week of gestation. This diverticulum divides into two parts: a larger cranial part, which gives rise to the liver, and a smaller caudal part, which develops into the gallbladder and cystic duct. At the beginning of the fifth week, the hepatic ducts, extrahepatic duct system, gallbladder, cystic duct, and pancreatic duct are demarcated as a solid cord of cells. Ductal lumina begin development during the sixth week in the common duct and slowly progress distally. The lumen extends into the cystic duct by the 7th week, but the gallbladder remains solid until the 12th week. Therefore, most gallbladder anomalies probably occur between the 4th and 12th week.10,11
Agenesis of the gallbladder is rare. Often incidental, but dilated ducts and choledocholithiasis can be seen with agenesis of the gallbladder. Duplication of the gallbladder can be diagnosed prenatally and often involves duplication of the cystic duct.2 Anomalies of the gallbladder alone do not generally give rise to any characteristic symptoms. Although some of the defects predispose to bile stasis and attacks of cholecystitis, the attacks themselves have usual aspects. The symptoms only call attention to the anomaly.10
Septate Gallbladder
A gallbladder septum may result from a congenital mucosal diaphragm, adenomyomatosis, or a combination of the two. Although a gallbladder septum may be an incidental finding during an otherwise normal examination, stasis of bile in the distal segment predisposes to calculus formation.12 A single septum appears as a thin linear echo separating the gallbladder into compartments. Simple junctional folds may mimic a septum.
The multiseptated gallbladder is one of the rarest congenital gallbladder malformations. This anomaly may be associated with biliary colic or cholelithiasis or may be entirely asymptomatic without associated cholelithiasis.13
A multiseptated gallbladder can have variable sonographic appearances. There can be fine linear septa or a honeycomb pattern of clustered septations resulting in multiple communicating cyst-like compartments. Septa may cluster in the neck and body region of the gallbladder. Differential diagnoses include desquamated gallbladder mucosa (an unusual finding in acute cholecystitis) and hyperplastic cholecystoses (such as polypoid cholesterolosis or adenomyomatosis). Desquamated gallbladder mucosa appears as multiple, haphazardly arranged, linear, nonshadowing densities within the gallbladder lumen, which does not consistently arise from the gallbladder wall because they do in multiseptated gallbladder. Polypoid cholesterolosis may more often resemble multiseptated gallbladder, although the nonshadowing polypoid densities are more bulbous, and there is no bridging of the lumen by septa as in the multiseptated gallbladder. In adenomyomatosis, Rokitansky-Aschoff sinuses could be confused with the honeycomb pattern, but cyst-like Rokitansky-Aschoff sinuses are smaller and are actually within the thickened gallbladder wall; there is no bridging of the lumen itself to form cyst-like compartments.13,14
Interposition of the Gallbladder
Childhood jaundice is usual. Although interposition of the gallbladder (the absence of the CHD and cystic duct) is a rare anomaly, its diagnosis is important because it is surgically correctable. Normally, the right and left main hepatic ducts join to form the CHD, which are entered by the cystic duct to form the CBD. In interposition, the main hepatic ducts drain, separately or together, directly into the gallbladder. The gallbladder then drains directly into the CBD, although variants can also occur (Fig. 8-15). The cause of interposition of the gallbladder is unknown.11
A patient with interposition of the gallbladder presents with jaundice, which may be intermittent, abdominal
pain, and sometimes an enlarged gallbladder. Sonography may show enlarged intrahepatic ducts with a normal CBD, mimicking Caroli disease, or the ducts may appear to enter a cystic mass in the porta hepatis, mimicking a choledochal cyst. Although sonography may be difficult to interpret in this situation, it is still a good initial step, indicating that jaundice is caused by an anatomic biliary abnormality.11
pain, and sometimes an enlarged gallbladder. Sonography may show enlarged intrahepatic ducts with a normal CBD, mimicking Caroli disease, or the ducts may appear to enter a cystic mass in the porta hepatis, mimicking a choledochal cyst. Although sonography may be difficult to interpret in this situation, it is still a good initial step, indicating that jaundice is caused by an anatomic biliary abnormality.11
Biliary Atresia
Biliary atresia is the most common type of obstructive biliary disease in infants and young children.15 Destruction of the extrahepatic biliary system occurs because of inflammation and sclerosing cholangiopathy.16 Progressive obliteration of the extrahepatic ducts and, in many instances, the gallbladder takes place. This obliteration extends into the proximal intrahepatic duct system, which usually remains patent in the first few weeks of life. The severity varies with the duration of involvement. Fibrosis and obliteration of the biliary tree progress distal to proximal.17
More than 50% of neonates have transient jaundice characterized by mild elevation of serum bilirubin, which resolves spontaneously. Persistent or sudden-onset jaundice after the 1st and 2nd week of life may indicate a more serious abnormality, most commonly, biliary atresia or neonatal hepatitis. Less common causes include choledochal cysts, inspissated bile syndrome, enzyme deficiencies, metabolic abnormalities, hemolysis, hyperalimentation, and other congenital biliary anomalies.11,17
 FIGURE 8-15 Schematic diagram of bile flow pattern (arrows) in interposition of the gallbladder. The sonographic appearance reveals dilated intrahepatic ducts adjacent to a normal or enlarged gallbladder with no dilatation of the common bile duct (CBD). Differential diagnosis includes choledochal cyst, gallbladder hydrops, and Caroli disease.27 HD, hepatic duct. |
Biliary atresia is twice as common in males, whereas neonatal hepatitis is four times more common in females. It is important to distinguish biliary atresia from neonatal hepatitis because atresia may be treated surgically with a liver transplant or the Kasai procedure. The outcome is better with early surgical intervention. If surgical correction is not possible, death usually occurs within months; however, a few children survive several years.4,17 Complications of untreated biliary atresia are cirrhosis, cholangitis, portal hypertension, malabsorption, and failure of biliary drainage.16,17
In normal neonates, the CHD is generally visible sonographically and measures no more than 1 mm. Intrahepatic duct dilatation combined with inability to visualize the CHD is suggestive of biliary atresia.17 If only the cystic duct is obstructed, a hydropic gallbladder will develop.4 Detection of both intrahepatic and extrahepatic dilatations excludes atresia and indicates obstruction (choledochal cyst, inspissated bile, and biliary calculi).17
Choledochal Cysts
There are five types of choledochal cysts, the most common being Type I, a fusiform dilatation of the CBD. Along with Type IV, Type I, has an abnormally long channel between the bile duct and pancreatic duct. The less common Type II is seen as true diverticula extending off the CBD. Type III is a duodenal choledochocele and Type IV is characterized by multiple cystic dilatations of the intrahepatic and extrahepatic ducts. Type V, Caroli disease (communicating cavernous ectasia), is a nonobstructive, saccular dilatation of communicating intrahepatic ducts.2 Different causes of choledochal cysts have been cited, including congenital weakness of the duct wall, which results in the formation of a cystic structure, and angulation of the CBD, causing partial obstruction leading to dilatation and cyst formation.4
Clinically, the signs and symptoms include intermittent jaundice associated with colicky pain, failure to thrive, and sometimes a palpable subhepatic mass displacing the stomach and the duodenum.4,18 Choledochal cysts are three to four times more common in females than in males, and often present early in life. Surgical management is recommended because of the increased incidence of malignant transformation that may occur later in life.2
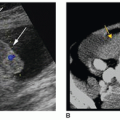
Sonographically, choledochal cysts appear as a localized cystic mass separate from the gallbladder in the region of the porta hepatis or intrahepatically depending on the type (Fig. 8-16). To avoid mistaking a fluid-filled bowel loop for a choledochal cyst, the examiner should verify peristalsis.
ACQUIRED DISEASES
Biliary Sludge
Sludge represents precipitates formed in the bile. It consists of a collection of calcium bilirubinate, mucus, and lesser amounts of cholesterol crystals within viscous bile that contains high concentrations of mucus and other proteins.19
 FIGURE 8-16 Longitudinal image in postcholecystectomy patient. A choledochal cyst is seen at the porta hepatis (arrows). PV, portal vein. |
The pathogenesis, clinical significance, and ultimate prognosis of sludge remain uncertain. Sludge alone can produce biliary symptoms such as the classic pain of gallstones, and it can also be associated with other complications. Therefore, sludge associated with biliary pain can be a significant finding. The presence of sludge implies the formation of a precipitate and should not be regarded as normal. Sludge is sometimes a precursor to gallstone disease.2,19
Sludge may be caused by conditions such as prolonged fasting, total parenteral nutrition (TPN), bile stasis, pregnancy, rapid weight loss and recent surgery and in critically ill patients.2,19
Sonographically, sludge produces a homogeneous, low-amplitude, nonshadowing echo pattern that tends to layer dependently (Figs. 8-17A-D and 8-18A, B). True sludge often forms a fluid-fluid level that remains constant in longitudinal and transverse images. Sludge slowly moves with changes in patient position. Sludge can disappear and
reappear over time. Scattered brighter echoes within sludge may represent larger cholesterol crystals. If sludge completely fills the gallbladder (total bile sludging or hepatization of bile), it may be difficult to distinguish the echo-filled gallbladder from adjacent liver parenchyma (Fig. 8-19). Sludge may also lead to gallstones.2,19
reappear over time. Scattered brighter echoes within sludge may represent larger cholesterol crystals. If sludge completely fills the gallbladder (total bile sludging or hepatization of bile), it may be difficult to distinguish the echo-filled gallbladder from adjacent liver parenchyma (Fig. 8-19). Sludge may also lead to gallstones.2,19
Tumefactive sludge from long-standing biliary obstruction frequently does not layer but often resembles a polypoid mass that can mimic a gallbladder neoplasm (Fig. 8-20A, B). Color Doppler sonography can be useful in determining tumefactive sludge from a neoplasm.20 Occasionally, mobile, round, echogenic, nonshadowing masses known as sludge balls are seen within the gallbladder.19 Also, sludge may be found in conjunction with gallstones (Fig. 8-21).
 FIGURE 8-19 Hepatization of bile. Sludge in the gallbladder has the same echo texture as liver. GB, gallbladder; LIV, liver. |
Using excessively high-gain settings fills the gallbladder with artifactual echoes, giving a false appearance of sludge. This artifactual pattern has a snowflake appearance, whereas true sludge has a defined, low-level pattern. Increased echogenicity of surrounding organs is another clue of too much gain. It is also important to distinguish sludge from the “false debris” echo pattern of slice thickness artifacts.
Cholelithiasis (Gallstones)
Gallstones can be large or small, single or multiple, symptomatic or silent. Gallstones are common worldwide with 2% to 10% of the population affected, with North America being near the 10% mark. The prevalence of gallstones is higher in females than in males.2 Gallstones are occasionally seen in fetuses and children (Fig. 8-22A, B).15,21
The majority of stones contain a mixture of cholesterol, bilirubin, and calcium. Approximately 75% of the gallstones in the United States are primarily cholesterol, with black or brown pigment stones accounting for 25% to 30% (Fig. 8-23). Many factors are associated with gallstone formation. Supersaturation of bile with cholesterol, abnormal gallbladder emptying, and altered absorption contributes to cholesterol stone formation.22,23
The most common risk factors for cholelithiasis are female gender, obesity, age, pregnancy, and diabetes.2 Other associated risks include ethnicity, genetics, diet, TPN, cirrhosis, rapid weight loss, ileal disorders (Crohn), and various medications.21
The majority of patients with gallstones are asymptomatic, with most found during routine abdominal scanning. Patients with symptoms, however, generally present with right upper quadrant (RUQ) pain that is steady, occurs after meals, or radiates to the upper back, shoulder, or epigastric area. The patient may also have nausea or vomiting.24 Pertinent laboratory values may include an elevated alkaline phosphatase and mildly elevated AST and ALT levels, when the cystic duct or CBD is obstructed.8
Prognosis and treatment of cholelithiasis can vary depending on the frequency and severity of the attacks as well as the size of the stones. Small calculi tend to be more troublesome because they may exit the gallbladder and cause ductal obstruction.24 Cholelithiasis may take a benign course, and a low-fat diet may be sufficient treatment in some cases. Persistent symptoms may require surgical intervention, either laparoscopic or open cholecystectomy, to provide definitive treatment of gallstones.21 Percutaneous cholecystostomy may assume a limited role in elderly patients and patients at high risk. Surgical cholecystostomy to remove stones has been performed, but cholecystectomy is often necessary subsequently because of the high recurrence rate.25 Sonography is the modality of choice for monitoring stones in these patients.2
The classic sonographic appearance of a gallstone is a mobile, gravity-dependent, echogenic foci within the gallbladder lumen that casts a posterior acoustic shadow (Fig. 8-24A-F).5 If the proper technique and transducer are used, virtually all calculi over 5 mm can be accurately diagnosed. If less than 2 to 3 mm, stones can be more difficult to visualize. However, small stones are typically multiple and described as “gravel,” thereby making detection easier (Fig. 8-25A, B).19 Small gallstones that are located within the cystic duct or in the neck of the gallbladder can be more difficult to visualize.5 The majority of gallstones produce “clean” shadows with distinct margins because they are highly reflective. The visualization of a clean shadow is dependent on gain settings, transducer position and frequency, harmonics, focusing, and compound imaging.2 Cholesterol stones or polyps can demonstrate reverberations and comet tail artifacts because of the rigidity and physical characteristics of cholesterol. Other causes of echogenic foci with reverberation and comet tail artifacts within the RUQ include air in the biliary tree, surgical clips in the gallbladder bed, gas in an intrahepatic abscess, drainage catheters, emphysematous cholecystitis, lead pellets, focal hepatic calcifications, and scars.
As the patient changes position, gallstones should roll to the most dependent portion of the gallbladder (Fig. 8-26A, B). Stones that do not demonstrate mobility may be polyps, stones impacted in the gallbladder neck, or stones adherent to the gallbladder wall (Fig. 8-27A, B).
There are many possible technical, anatomic, and diagnostic pitfalls of cholelithiasis (Table 8-1). Again, meticulous scanning in multiple planes using a variety of techniques, patient positions, and transducer frequencies is crucial for accurate diagnosis and proper patient management.
With careful technique, the wall-echo-shadow (WES) triad or double-arc shadow sign may be seen (Fig. 8-28A, B). The first arc or curved echogenic line represents the thickened gallbladder wall. The second arc is from the surface of the stone followed by posterior acoustic shadowing.1,2 With chronic disease, the gallbladder may be so contracted that it is difficult to visualize sonographically. Air-filled bowel loops in the RUQ may create shadowing, which can be mistaken for a contracted gallbladder with stones. The differential diagnosis for chronic cholecystitis is adenomyomatosis and gallbladder carcinoma.26
Acute Cholecystitis
In up to 95% of cases, acute cholecystitis or inflammation of the gallbladder results from impacted stones within the neck of the gallbladder or the cystic duct.5,19,20,27 It is the most common inflammatory condition of the gallbladder.27 Inflammation may result in necrosis, ulceration, swelling, and edema.5 Various degrees of bacterial infection occur with acute cholecystitis, leading to potential complications.8
Clinically, the signs and symptoms of cholecystitis are indistinguishable and somewhat nonspecific. Patients present with RUQ pain, positive Murphy sign, nausea, vomiting, distention, fever, a palpable RUQ mass, and/or jaundice.8,20,28 These symptoms can be confused with acute pancreatitis, perforated peptic ulcer, liver abscess, or acute alcoholic hepatitis.8 Laboratory results can also be nonspecific, possibly showing serum liver transaminase, leukocytosis, hyperbilirubinemia, or elevated alkaline phosphatase.29 Approximately 20% of patients with cholelithiasis will develop acute cholecystitis; however, only 20% to 35% of patients with RUQ pain will have acute cholecystitis.5,20
On sonogram, a positive sonographic Murphy sign, wall thickening, and gallstones are signs of acute cholecystitis. Pericholecystic fluid and, at times, a hydropic gallbladder can also be seen.5,20 Together, the findings of cholelithiasis and a positive sonographic Murphy sign are highly suggestive of acute cholelithiasis. Gallbladder wall thickening and pericholecystic fluid are considered secondary signs.27 Color or power Doppler imaging may also be helpful in diagnosing acute cholecystitis by detecting hyperemia and an enlarged cystic artery (Fig. 8-29A-F).2
One thought is to manage the patient medically because 60% of acute cases resolve spontaneously, and surgery should be saved until the acute attack has subsided. The preferred approach is to perform a cholecystectomy within the first several days of the onset of symptoms because early surgical intervention results in fewer complications and lower costs.20,30 In patients with severe acute cholecystitis who are poor surgical candidates, are very ill, or are elderly, sonographically guided aspiration and percutaneous drainage of the gallbladder or antibiotic therapy is an alternative.29 The pathophysiologic events of acute cholecystitis represent a dynamic process. Time is needed for these changes to occur. As a result, the clinical onset may precede the appearance of sonographic signs by as much as 12 to 24 hours.31
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree