Acute disseminated encephalomyelitis (ADEM) is an immunologically mediated inflammatory disease of the central nervous system that typically occurs after a viral infection or recent vaccination, and is most commonly seen in the pediatric population. In 2007 the International Pediatric Multiple Sclerosis Study Group proposed a consensus definition for ADEM for application in research and clinical settings. This article gives an overview of ADEM in children, focusing on differences that have emerged since the consensus definition was established. Although the focus is on neuroimaging in these patients, a synopsis of the clinical features, immunopathogenesis, treatment, and prognosis of ADEM is provided.
Key points
- •
Acute disseminated encephalomyelitis (ADEM) typically occurs after a viral infection or recent vaccination.
- •
ADEM can represent a diagnostic challenge for clinicians, as many disorders (inflammatory and noninflammatory) have a similar clinical and radiologic presentation.
- •
The differential diagnosis for multifocal hyperintense lesions on neuroimaging includes an exhaustive list of potential mimickers, namely infectious, inflammatory, rheumatologic, metabolic, nutritional, and degenerative entities.
Introduction
Acute disseminated encephalomyelitis (ADEM) is an immunologically mediated inflammatory disease of the central nervous system (CNS) resulting in multifocal demyelinating lesions affecting the gray and white matter of the brain and spinal cord. ADEM is characteristically a monophasic illness that is commonly associated with an antigenic challenge (febrile illness or vaccination), which is believed to function as a trigger to the inflammatory response underlying the disease. It is most commonly seen in the pediatric population, but can occur at any age. Symptoms are highly dependent on the area of the CNS affected, but are polyfocal in nature. Common symptoms include hemiparesis, cranial nerve palsy, seizures, cerebellar ataxia, and hypotonia. The diagnosis of ADEM depends on the history, physical examination, and supplemental neuroimaging.
Despite the long-standing recognition of ADEM as a specific entity, no consensus definition of ADEM had been reached until recently. Historically, different definitions of ADEM have been used in published cases of pediatric and adult patients, which varied as to whether events required (1) monofocal or multifocal clinical features, (2) a change in mental status, and (3) a documentation of previous infection or immunization. To avoid further misdiagnosis and to develop a uniform classification, the International Pediatric Multiple Sclerosis (MS) Study Group proposed a consensus definition for ADEM for application in both research and clinical settings ( Box 1 ). One of the most significant changes proposed by this definition was the mandatory inclusion of encephalopathy as a clinical symptom in patients presenting with ADEM. Before the development of the consensus definition, although encephalopathy was included in the clinical description it was not considered an essential criterion for the diagnosis. Thus, many of the previous studies investigating the clinical and radiologic features of pediatric ADEM were performed on patients who may no longer meet the consensus criteria, and may have led to the classification of other neurologic disorders as ADEM ( Table 1 ). It may be that there is an inherent difference in the patients who present with multifocal symptoms and encephalopathy as opposed to those without encephalopathy; therefore, this distinction is imperative. Because of the lack of uniform description and clear clinical and neuroimaging diagnostic criteria in ADEM, caution must be exercised when applying previous clinical and radiologic descriptions of patients with this disorder.
- •
No history of prior demyelinating event
- •
First clinical event with presumed inflammatory or demyelinating cause
- •
Acute or subacute onset
- •
Affects multifocal areas of central nervous system
- •
Must be polysymptomatic
- •
Must include encephalopathy (ie, behavioral change or altered level of consciousness)
- •
Neuroimaging shows focal/multifocal lesion(s) predominantly affecting white matter
- •
No neuroimaging evidence of previous destructive white matter changes
- •
Event should be followed by clinical/radiologic improvements (although may be residual deficits)
- •
No other etiology can explain the event
- •
New or fluctuating symptoms, signs, or magnetic resonance imaging findings occurring within 3 months are considered part of the acute event
| Authors, Ref. Year | N | New Criteria a (%) | White Matter (%) | Gray Matter (%) | Brainstem (%) | Cerebellum (%) | Enhancing b (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Deep | Juxt | Peri | CC | Cort | BG | Thal | ||||||
| Hynson et al, 2001 | 31 | 68 | 90 | 29 | 29 | 61 | 39 | 32 | 42 | — | 8/28 | |
| Tenembaum et al, 2002 | 84 | 69 | — | — | — | — | — | — | 12 | — | — | 8/27 |
| Murthy et al, 2002 | 18 | 44 | 100 8.0 c (1–30) | — | 60 2.5 c (0–16) | 7 0.1 c (0–1) | 80 4.9 c (0–28) | 20 0.3 c (0–2) | 27 0.3 c (0–2) | 47 1.1 c (0–8) | 13 0.1 c (0–1) | 4/15 |
| Anlar et al, 2003 | 33 | 48 | 42 | 12 | 12 | — | 45 | — | 45 | 42 | 2/31 | |
| Richer et al, 2005 | 10 | 40 | 90 | — | 30 | 80 | — | 60 | 50 | 3/9 | ||
| Madan et al, 2005 | 7 | 43 | 86 | — | — | — | — | 43 | 14 | 14 | 14 | — |
| Singhi et al, 2006 | 52 | 56 | 54 | — | 19 | 13 | — | 17 | 30 | 17 | 26 | — |
| Mikaeloff et al, 2007 | 108 | 100 | — | 61 | 42 (>2) | — | 18 | 58 | 63 | 14/85 | ||
| Atzori et al, 2009 | 20 | 65 | — | 60 | 20 | 5 | 15 | 50 | 60 | 10/14 | ||
| Alper et al, 2009 | 24 | 42 | 68 | 21 | 18 | 9 | — | 43 | 41 | 50 | — | |
| Callen et al, 2009 | 20 | 100 | 80 | 90 | 65 | 80 | 80 | 70 | 70 | 70 | 5/11 | |
| Visudtibhan et al, 2010 | 16 | 100 | 75 | 19 | 75 | — | 19 | 50 | 50 | 25 | — | |
| Pavone et al, 2010 | 17 | 100 | — | — | — | — | — | — | 23 | — | — | 3/17 |
a Consensus criteria of International Pediatric Multiple Sclerosis Society.
b Number of patients showing enhancing lesions (numerator) over the number of study patients who received contrast (denominator).
c Mean lesion count in category and minimum and maximum lesion counts (in parentheses).
This review is intended to give an overview of ADEM in the pediatric population, focusing on differences that have emerged since the consensus definition was established. Although the focus is on neuroimaging in these patients, a synopsis of the clinical features, immunopathogenesis, treatment, and prognosis of ADEM is provided.
Epidemiology and clinical presentation
Considering that the diagnostic criteria for ADEM were not elucidated before 2007, the annual incidence rate and prevalence within the population is not precisely known. In addition, no analyses of worldwide distribution of ADEM have been completed; therefore, the reported prevalence and incidence taken within a single area may not be generalizable to the population as a whole. Before 2007, the prevalence of ADEM within the pediatric population was estimated at 0.8 to 1.1 per 100,000 in those younger than 10 years. A study by Leake and colleagues evaluated the incidence of ADEM in San Diego County, USA. The investigators estimated this to be 0.4 per 100,000 per year in those younger than 20 years. More recent studies completed after the definition of ADEM had been established have suggested that these rates may actually be higher. Visudtibhan and colleagues reported the prevalence of children with definite ADEM in Bangkok, Thailand to be 4.1 per 100,000. Another study from Fukuoka Prefecture, Japan reported the annual incidence to be 0.64 per 100,000. The overall frequency of ADEM in Canadian children with acquired demyelinating disorders had been estimated at 22%.
Although ADEM may present at any age, it is most frequently described in the pediatric population. The mean age of onset in the pediatric population is reported to be 7.4 ± 1.3 years of age and the median age of onset is 8 years, according to a recent meta-analysis. However, 12 of the 13 studies included in the analysis were performed before the revised ADEM definition. More recent studies using the new criteria for ADEM have shown a similar mean age of onset, ranging from 5.7 to 7.6 years. An equal sex distribution was previously suggested, but more current studies show a slight male preponderance (1.4–2.3:1) in patients presenting with ADEM at. A seasonal distribution of ADEM has been described, with an increased number of cases during the winter and spring months.
ADEM typically occurs after a viral infection or recent vaccination. The frequency with which a preceding febrile illness is noted has widely varied in previous literature, ranging from 46% to 100%. This variation is likely attributable to the lack of uniform diagnostic criteria, with some investigators using the term ADEM only when a preceding febrile illness has been documented, as well as different latency periods from febrile illness to ADEM being deemed clinically significant. A recent meta-analysis showed that a preceding triggering event occurred in 69% of 492 patients diagnosed with ADEM. The most common preceding trigger is a nonspecific upper respiratory tract infection. The duration between antigenic challenge and the first signs and symptoms of ADEM has been cited as ranging from 1 to 28 days with a mean of 6 to 12 days. Pavone and colleagues showed different latency periods depending on the triggering factor, with the shortest latency being seen for upper respiratory tract infections or gastroenteritis (2–4 days). Although a link between viral illness and ADEM is likely, it should be noted that there is a high frequency of viral episodes in childhood, and medical history is often positive regardless of whether a causal correlation exists.
The neurologic features of ADEM are often seen following a short prodromal phase consisting of fever, malaise, headache, nausea, or vomiting. Patients subsequently develop neurologic symptoms subacutely, within a mean period of 4.5 to 7.5 days (range: 1–45 days). Occasionally there is rapid progression of symptoms and signs to coma and/or decerebrate posturing. The clinical presentation of ADEM is widely variable, with the type and severity clinical features being determined by the distribution of lesions within the CNS. Although studies have previously reported an encephalopathy in 21% to 74% of patients (with only 55% having altered mental status in a recent meta-analysis ), application of the new diagnostic criteria mandates that encephalopathy (either behavioral change or altered mental status) be present in 100% of cases. It should thus be noted that previous studies looking at the presenting signs and symptoms encompass patients who, today, would not meet diagnostic criteria for ADEM. With that in mind, the neurologic features that have previously been noted in ADEM include: unilateral or bilateral pyramidal signs (60%–95%); cranial nerve palsies (22%–89%); hemiparesis (76%–79%); ataxia (18%–65%); hypotonia (34%–47%); seizures (10%–47%); visual loss due to optic neuritis (7%–23%); and speech impairment (5%–21%). Peripheral nervous system involvement has been reported in the adult cohort (with frequencies up to 43.6% of patients), but is considered rare in childhood ADEM patients.
Only 2 studies have looked at presenting signs and symptoms of patients with ADEM according to the new consensus definitions. Mikaeloff and colleagues reported that 79% presented with long tract dysfunction, 48% presented with brainstem dysfunction, 32% presented with seizures, and 6% presented with optic neuritis. Pavone and colleagues found the following signs and symptoms: ataxia (47%), hypotonia (41%), seizures (29%), thalamic syndrome (23%), hemiparesis (23%), cranial nerve palsy (18%), headache (18%), fever (12%), and ptosis (6%).
Respiratory failure secondary to brainstem involvement or severely impaired consciousness has been reported in 11% to 16% of patients in previous studies. In a study where all children met the consensus definition of ADEM, the numbers of patients with respiratory failure were strikingly similar (11%). A recent study investigating the necessity of intensive care unit (ICU) admissions for patients with ADEM found that 25% of patients with ADEM required an ICU admission, with an incidence of 0.5 per million children per year. Rates of ICU admission may be higher than previously expected, considering the mandatory inclusion of altered mental status. In the study by Pavone and colleagues, ICU admissions were necessary in 41% of patients with ADEM at some point during their clinical course.
Immunopathogenesis
The precise mechanisms implicated in ADEM are not well known, and the relationship between the pathogenesis of ADEM and MS continues to remain a matter of controversy. There is a general consensus that ADEM is an immune-mediated disorder resulting from an autoimmune reaction to myelin. An autoimmune pathogenesis is supported by the pathologic similarities between ADEM and the experimental allergic encephalomyelitis (EAE) model, which is a demyelinating disease that may be induced in a variety of animal species after immunization with myelin proteins or peptides.
There are 2 basic mechanisms proposed to cause ADEM, both of which rely on the exposure of the immune system to an antigenic challenge (ie, viral, bacterial, or exposure to degradation products via immunization):
- 1.
Molecular mimicry theory. This theory relies on the idea that myelin antigens (for example, myelin basic protein [MBP], proteolipid protein, and myelin oligodendrocyte protein) could share a structural similarity with antigenic determinants on the infecting pathogen. The infected host mounts an immune response producing antiviral antibodies that are thought to cross-react with myelin antigens that share a similar structure, inadvertently producing an autoimmune response. Myelin proteins have shown resemblance to several viral sequences, and cross-reactivity of immune cells has been demonstrated in several studies. T cells to human herpesvirus-6 (HHV-6), coronavirus, influenza virus, and Epstein-Barr virus (EBV) have been shown to cross-react with MBP antigens. Furthermore, enhanced MBP reactive T-cell responses have been demonstrated in patients with postinfectious ADEM, and enhanced anti-MBP antibodies have been shown in patients with postvaccinial ADEM following vaccination with Semple rabies vaccine.
- 2.
Inflammatory cascade theory. Nervous system tissue is thought to be damaged secondary to viral infection, resulting in the leakage of myelin-based antigens into the systemic circulation through an impaired blood-brain barrier. These antigens promote a T-cell response after processing in the lymphatic organs, which in turn causes secondary damage to the nervous system tissue through an inflammatory response. This theory is supported by the Theiler murine encephalomyelitis virus (TMEV)-induced demyelinating disease model, which is a biphasic disease of the CNS whereby direct infection of neurotropic TMEV picornavirus results in an initial CNS injury followed by a secondary autoimmune response. However, this model has been criticized for its superficial resemblance to ADEM, as ADEM is not thought to be due to a direct viral infection of the CNS. It is more likely that the nervous system tissue damage proposed by this theory is indirect, through the release of multiple cytokines and chemokines in response to the initial infection. The role of chemokines, particularly interleukin-6, tumor necrosis factor α, and matrix metalloproteinase 9, have been hypothesized to play a role in ADEM, and the spectrum of chemokines found to be elevated in ADEM may differ from that in MS.
As expected by the currently proposed mechanisms discussed here, ADEM is frequently preceded by an infection or recent vaccination. However, in most cases investigations fail to identify the precise infectious agent responsible. Viruses that have been implicated in promoting the immune response responsible for ADEM include herpes simplex virus, human immunodeficiency virus, HHV-6, mumps, measles, rubella, varicella, influenza, enterovirus, hepatitis A, coxsackie, EBV, and cytomegalovirus. It has been proposed that the risk of ADEM is highest with measles and rubella, with the risk after infection with these viruses being 1:1000 and 1:20,000, respectively. Other infectious agents that have been linked to ADEM include group A β-hemolytic streptococcal infection, pertussis, Mycoplasma pneumonia , Borrelia burgdorferi , Legionella , Rickettsiae, and Plasmodium falciparum and Plasmodium vivax malaria.
Approximately 5% to 12% of patients with ADEM have a history of vaccination within the month before presentation. The only vaccination that has been epidemiologically and pathologically proven to be associated with ADEM is the Semple form of the rabies vaccine. Other vaccinations that have been reported to have a temporal association with the onset of ADEM include hepatitis B, pertussis, smallpox, diphtheria, measles, mumps, rubella, human papilloma virus, pneumococcus, varicella, influenza, Japanese B encephalitis, polio, and meningococcal A and C. The rates at which these vaccinations are reported to cause ADEM are: 1 to 2 per million for measles ; 1 in 3000 to 1 in 7000 for Semple rabies ; 1 in 25,000 for duck embryo rabies ; less than 1 in 75,0000 for nonneural human diploid cell rabies ; 0.2 per 100,000 for inactivated mouse brain–derived Japanese B encephalitis ; 3 in 665,000 for smallpox ; and 0.9 per 100,000 for diphtheria, pertussis, and tetanus. On rare occasions ADEM has been also reported following organ transplantation.
Recently, studies have investigated the role of genetics in predisposing patients to ADEM. Multiple human leukocyte antigen (HLA) alleles have been found to occur at a higher frequency in patients with ADEM, including HLA-DRB1*1501, HLA-DRB5*0101, HLA-DRBQ*1503, HLA-DQA1*0102, HLA-DQB1*0602, HLA-DRB1*01, HLA-DRB*03, and HLA-DPA1*0301. The exact frequency of expression of these alleles and their future clinical utility in ADEM is currently unknown.
Laboratory findings
Laboratory findings are useful for ADEM, mainly to rule out other causes for the patient’s presenting symptoms. Despite ADEM patients commonly reporting a recent infection before their neurologic presentation, only 17% have evidence of a recent infection on serology.
Cerebrospinal fluid (CSF) is normal in up to 61.5% of patients with ADEM. If patients are found to have abnormal CSF parameters, they are usually minor and nonspecific. A lymphocyte pleocytosis (usually between 50 and 180 cells/mm 2 ) and elevated protein (commonly 0.5–1.0 g/dL) can be seen. Rarely a mild increase in glucose has been noted. Oligoclonal bands, which are commonly positive in patients with MS, are less frequently observed in patients with ADEM (seen in 0%–29%). Elevations in immunoglobulins have been reported in up to 13% of patients.
An electroencephalogram (EEG) may be completed as part of the workup of a patient with ADEM following a presentation with seizures or to rule out nonconvulsive status epilepticus as a cause of the accompanying encephalopathy. Although 78% of patients with ADEM have a diffusely slow background (consistent with encephalopathy), focal slowing (10%) or focal epileptiform discharges (2%) may be seen. EEG is seldom useful in establishing diagnosis.
Evoked potentials (including visual evoked potentials, brainstem auditory evoked potentials, and somatosensory evoked potentials) may be normal, depending on the location of brain lesions. Abnormal visual evoked potentials have been reported in up to 12% of patients.
Neuroimaging
Computed Tomography of the Brain
There are few studies that comment on computed tomography (CT) findings in patients with ADEM, namely because these patients are more commonly imaged with more sensitive imaging modalities, in addition to the concern of radiation exposure in the pediatric population. Most studies indicate that CT is unrevealing when completed early in the disease and that this imaging modality is insensitive for smaller demyelinating lesions. The most commonly reported abnormalities are discrete hypodense areas within cerebral white matter and juxtacortical areas. However, some investigators have reported high rates of CT-scan abnormalities in patients with ADEM. Tenembaum and colleagues reported abnormal findings in 78% of patients after a mean interval of 6.5 days from symptom onset. In an article by Pavone and colleagues, where all patients met the new consensus criteria for ADEM, abnormal CT scans were reported in 86% of their patients when performed after a mean interval of 2.5 days from initial neurologic presentation.
Magnetic Resonance Imaging of the Brain and Spine
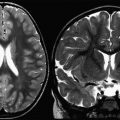
Magnetic resonance (MR) imaging of the brain is the most important paraclinical tool available to aid in the diagnosis of ADEM and to distinguish the clinical presentation from other inflammatory and noninflammatory neurologic diseases. Since the advent of MR imaging, many studies have evaluated the radiologic appearance of ADEM in children and adults. From the listed studies, the typical MR imaging findings described in ADEM are widespread, bilateral, asymmetric patchy areas of homogeneous or slightly inhomogeneous increased signal intensity on T2-weighted imaging within the white matter, deep gray nuclei, and spinal cord. Within the white matter, juxtacortical and deep white matter is involved more frequently than is periventricular white matter, which is an important contrast to patients with MS. In addition, lesions involving the corpus callosum, which are considered typical in MS, are rarely seen in ADEM. Infratentorial lesions are common, including the brainstem and cerebellar white matter. With respect to lesion size and morphology, variation is seen, ranging from small, round lesions to large, amorphous, and irregular lesions. Unenhanced T1-weighted images reveal that lesions are typically inconspicuous unless the lesions are large, in which case a faint hypodensity is seen within the affected areas. These lesions typically appear simultaneously with clinical presentation. However, delayed appearance of abnormalities up to 1 month after clinical onset has been described, so a normal MR image within the first days after symptom onset suggestive of ADEM does not exclude the diagnosis. Contrast enhancement in ADEM is variable and has been reported in 30% to 100% of patients with ADEM, in nonspecific patterns (nodular, diffuse, gyral, complete, or incomplete ring).
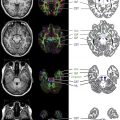
In the past 10 years, only 4 studies have described the MR imaging appearance of cohorts of children who uniformly meet the current consensus criteria for ADEM. A summary of the neuroimaging findings of selected studies completed within the last 10 years is presented in Table 1 . Fig. 1 displays the potential location sites for demyelination as described below.
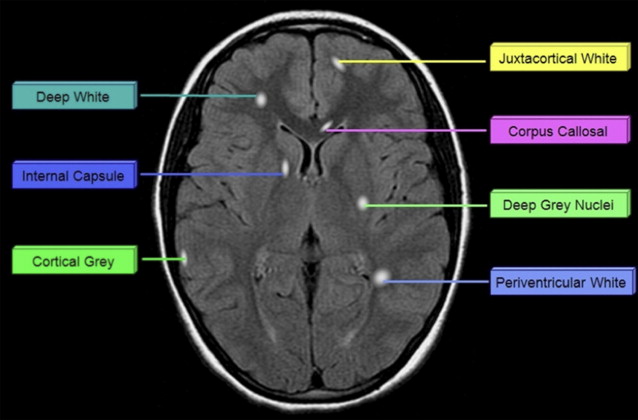
Mikaeloff and colleagues qualitatively described the radiologic appearance of 108 children with monophasic ADEM. In their cohort, juxtacortical white matter, deep gray matter, and infratentorial structures were affected with approximately 60% frequency. Lesions were less commonly seen in periventricular white matter, cortical gray matter (18%), and spinal cord (12%). Deep white matter lesions and cerebellar lesions were not specifically described, thus their frequency of involvement cannot be commented on. Almost half of the children in their cohort had at least 9 lesions, but comprehensive lesion counts were not reported. Bilateral lesions were described in 81% of their patient cohort. “Large lesions” (>2 cm) were present in 72% of cases, but there was no comment made about other lesion sizes. Nearly half of their patients met at least 3 Barkoff criteria. Of the patients who received gadolinium, only 16% displayed enhancing lesions. However, there was no comment about the pattern of enhancement.
A smaller study was more recently published by Vistudtibhan and colleagues. Although 16 patients met the initial criteria for ADEM, 3 of these patients subsequently fulfilled criteria for relapsing-remitting MS. Based on the reported imaging characteristics described for the remaining 13 patients with monophasic ADEM, the brain region most commonly involved was the subcortical/periventricular region (88%). A distinction was not made between subcortical lesions and periventricular lesions, which would have been useful considering that the literature seems to favor periventricular lesions as being more characteristic of MS in comparison with ADEM. Similar to the results from Mikaeloff and colleagues, brainstem and deep gray nuclei were also commonly affected, with a frequency of 69% and 50%, respectively. Lesions involving cortex and juxtacortex were seen less frequently (31%); however, there was no distinction made between these 2 areas. The cerebellum was involved in approximately one-third of the patients. Spinal cord involvement was much higher than that reported by Mikaeloff and colleagues, with a frequency of approximately 60%. There was no comment made about lesion size, nor the frequency of gadolinium enhancement.
Data from the authors’ group was published in 2009. The characteristics of lesions in 20 children with monophasic ADEM were quantitatively assessed. When the data are viewed qualitatively (ie, displaying the number of patients having at least 1 lesion in any given location), lesions appear to be relatively common in all regions of the brain. However, when the mean lesion counts are evaluated for each region ( Table 2 ), a difference in the lesion distribution is apparent. Similar to the findings by Mikaeloff and colleagues and Vistudtibhan and colleagues, lesions were more commonly seen in the deep white matter and juxtacortical white matter than in the periventricular white matter. In contrast to the previous studies by both groups, lesions were commonly found to impinge on the cortical ribbon. Another consistent result between all 3 studies was the frequent involvement of the deep gray nuclei. Although the mean lesion count was only 2.5, only 30% of the patients had no deep gray involvement. Infratentorial regions were commonly involved, as reported in the other studies, but the mean lesion counts in these regions were low. With respect to lesion size, small (<1 cm axial, <1.5 cm longitudinal) and medium (1–2 cm axial, 1.5–2.5 cm longitudinal) lesions were found in all patients, but 70% also had large (>2 cm axial, >2.5 cm longitudinal) lesions. This amount is nearly identical to the number of “large lesions” seen by Mikaeloff and colleagues. Finally, only 11 of the authors’ patients received gadolinium, 5 of whom (45%) displayed enhancement.