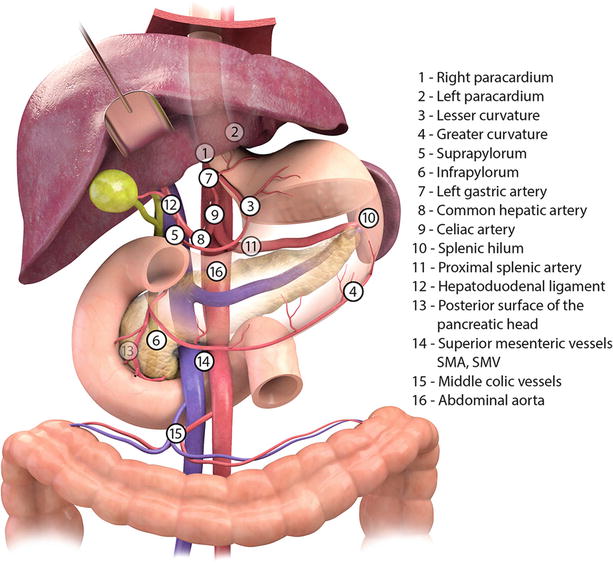
Fig. 9.1
Stomach gross anatomy
Blood supply from the celiac axis and superior mesenteric artery with multiple collaterals perfuse the stomach with the lesser curvature receiving blood from the right and left gastric arteries and the greater curvature from the right and left gastroepiploic arteries (Fig. 9.2). Venous drainage is to the splenic and superior mesenteric veins (Fig. 9.3). Lymphatics follow the arteries to the celiac axis lymph nodes [1].
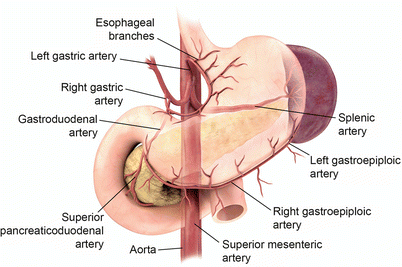
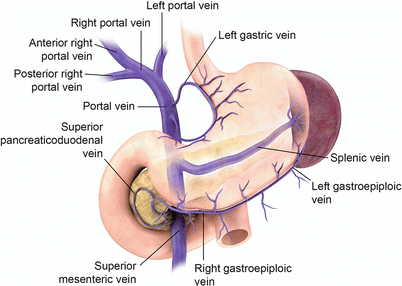

Fig. 9.2
Stomach arterial anatomy

Fig. 9.3
Stomach venous anatomy
Internally, the gastric wall appears corrugated due to redundant mucosal folds, known as rugae. Like other segments of the gastrointestinal tract, the gastric wall is divided into five layers: mucosa (internally), muscularis mucosa, submucosa, muscularis propria, and serosa (externally). The muscularis propria is in turn divided into an inner oblique, middle circular, and out longitudinal muscular layers (Fig. 9.4).
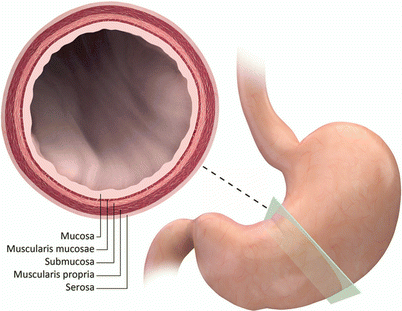

Fig. 9.4
Gastric wall layers
Externally, several ligamentous attachments to other organs may also serve as medium for the spread of disease. These include the gastrohepatic ligament (or lesser omentum) attaching the lesser curvature to the medial hepatic border; the gastrocolic ligament, which continues as the anterior leaf of the greater omentum after attaching the greater curvature to the transverse colon; and the gastrosplenic ligament connecting the stomach and the spleen (Fig. 9.5).
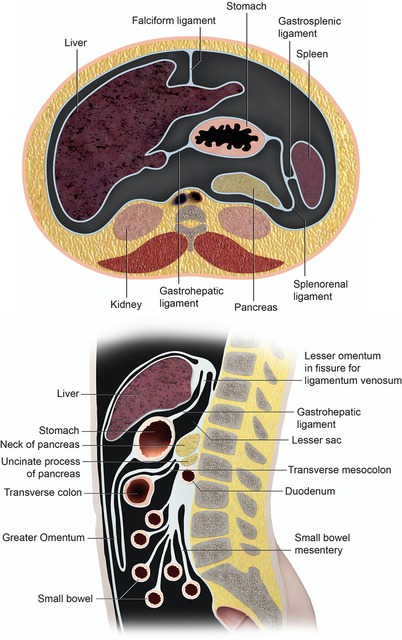

Fig. 9.5
Stomach cross-sectional anatomy
Stomach Pathology
Table 9.1
Table 9.1
Stomach pathology
Congenital | Inflammatory/infectious | Benign gastric tumors | Malignant neoplasms | Acquired structural anomalies | Gastric wall disruption | Vascular disease | Extrinsic processes |
|---|---|---|---|---|---|---|---|
Gastric diverticulum | H. pylori gastritis | Hyperplastic polyps | Gastric adenocarcinoma | Sliding hiatal hernia | Vomit-induced tear | Gastric ischemia | Pancreatic cyst |
Hypertrophic pyloric stenosis | Secondary gastritis: NSAIDS, EtOH, coffee, stress, XRT | Adenomatous polyps: familial adenomatous polyposis, Gardner’s and Turcot’s syndrome | Lymphoma | Paraesophageal hernia | Steroid use | Duplications cysts | |
Gastric atresia | Crohn’s disease | Hamartomatous polyps: Peutz-Jeghers, juvenile polyps, Cowden’s and Cronkhite-Canada syndrome | GIST (malignant) | Volvulus: organoaxial and mesenteroaxial | Traumatic tear | Organomegaly | |
Antral web | Tuberculosis | Lipoma | Carcinoid | Gastric fistula | Gastric wall hematoma | Masses and cysts from other organs | |
Congenital diaphragmatic hernia | Ménétier disease | GIST (benign) and leiomyoma | Metastases | Gastric outlet obstruction | |||
Microgastria | Zollinge-Ellison syndrome | Benign neural, neuroendocrine, and carcinoid tumors | Gastric atony | ||||
Dextrogastria: situs inversus, isolated | Caustic injury | Ectopic pancreas | |||||
Gastric duplication | Adjacent inflammation (i.e., pancreatitis) | ||||||
Emphysematous gastritis |
Cross-Sectional Modalities and Techniques
For many years, endoscopy and fluoroscopy have been the mainstay of gastric imaging. These are outside the scope of this book, as cross-sectional imaging is discussed, herein. Furthermore, cross-sectional exams have for many years been an important part of the workup for gastric pathology. As the technology continues to advance, cross-sectional imaging has become an essential, if not the primary mode of evaluating the stomach.
Computed Tomography
When gastric disease is suspected, a dedicated computed tomography (CT) protocol of the stomach can be used. Yet, gastric pathology is more commonly identified incidentally during CT examination performed for other reasons. CT also commonly serves as an adjunct to endoscopy and fluoroscopy [2]. In addition, CT is the modality of choice for gastric neoplasm staging and postoperative follow-up.
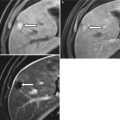
The stomach is best evaluated after a 6–8 h fasting. Approximately 1,000–1,500 cc of neutral (water density) contrast material, such as Volumen, may be administered stepwise, with 75 % of the volume 15 min prior to scan and 25 % just before scanning. Conversely, negative contrast can be obtained by administering gas-producing crystals. Positive oral contrast is less useful, as it may mask enhancing lesions or vessels with similar post-contrast attenuation values (Fig. 9.6). Adequate distention of the stomach is the key in detecting and differentiating various gastric conditions. Some authors advocate administration of hypotonics, such as scopolamine to obtain good luminal distension [3, 4].
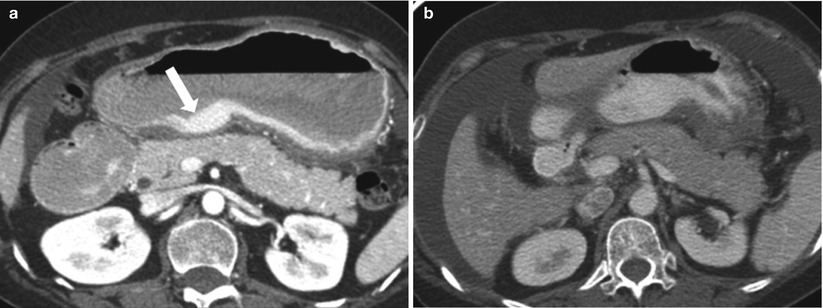

Fig. 9.6
Contrast-enhanced CT using negative (a) and positive (b) enteric contrast. Abnormal enhancing thickening of the posterior gastric wall (arrow on a) is not evident with the use of positive enteric contrast. This was pathologically proven as gastric adenocarcinoma
Intravenous contrast-enhanced CT is performed during the late arterial/early venous phase [3, 5]. Not all the layers of the gastric wall are clearly visible by CT, but the enhancing mucosa, hypoenhancing submucosa, and moderately enhancing muscularis propria and serosa may be discerned [6]. Detection of typically hypovascular hepatic metastases from gastric adenocarcinoma is best accomplished during the portal venous phase, whereas an early arterial phase may aid in local staging [3, 7]. Multiplanar reformation (MPR) can aid in the diagnosis, and thin-section slice scan can also be obtained for virtual gastroscopy (VG) after processing the imaging data with endoluminal and volume rendering software [3, 5, 8, 9].
Magnetic Resonance Imaging
Magnetic resonance imaging (MRI) is less commonly used for evaluation of the stomach but can be a powerful tool, if properly conducted. As in CT, adequate distension may again be achieved with 1 l of water or other negative enteric contrast and administration of a hypotonic agent. Additionally, the use of fast sequences, such as single-shot spin-echo and steady-state free precession, will decrease motion artifacts. Similarly, post-intravenous contrast, volumetric/3D gradient-echo images can be utilized. Despite superior soft tissue contrast and lack of ionizing radiation, these exams require more time and expertise [10, 11].
Positron Emission Tomography
The use of positron emission tomography (PET) in the evaluation of gastric carcinoma is not as well established as with other neoplasms. As with other cancers, evaluation of gastric cancer patients with whole-body with fluorodeoxyglucose (FDG) PET may be a useful adjunct in the detection of distant nodal and solid organ metastases. Response to chemotherapy may also be evaluated with FDG-PET [7]. Locally, the normal stomach demonstrates moderate FDG uptake, which is greater proximally. Focal areas of uptake, intense uptake diffusely or distal greater than proximal uptake, suggest possible inflammatory or neoplastic pathology. Focal increased uptake may also be seen at the GEJ in the setting of reflux or hiatal hernia [12].
Ultrasound
Due to the presence of gas, evaluation of the stomach can be quite challenging, and dedicated gastric ultrasound (US) is reserved for selected cases, such as hypertrophic pyloric stenosis. Like the rest of the gastrointestinal tract, the laminar anatomy of the gastric wall can be delineated by US, especially when endoscopic US (EUS) is performed. Most commonly, an echogenic inner mucosa and submucosa layer are seen, surrounded by a hypoechoic muscularis propria. Techniques such as ingestion of water and compression can aid in diagnosis [13, 14].
Imaging Patterns
Algorithm 9.1 Imaging patterns of stomach pathology
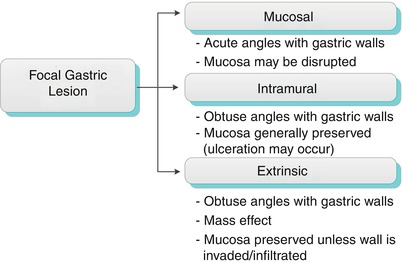
Focal Gastric Lesion
Characterization of gastric wall lesions as mucosal, intramural, and extrinsic can help narrow the differential considerations. A polypoid lesion producing acute angles with the gastric wall and an interrupted, irregular, or indistinct mucosal surface suggest a mucosal or epithelial lesion. More obtuse angles and preservation of the mucosal pattern point to a subepithelial lesion. This includes intramural lesions, typically arising from the submucosal layer, as well as extrinsic lesions outside the gastric wall. Some intramural lesions may also grow outside the gastric wall, thus exhibiting and exogastric component. Extrinsic lesions may mimic intramural lesions, as they have similar intraluminal appearance, but they also exhibit mass effect on the gastric wall [6]. The lesion is usually seen on cross-sectional studies without difficulty (Fig. 9.7).

Algorithm 9.2 Imaging findings of focal gastric lesions

Fig. 9.7
Focal gastric lesions may be polypoid with acute angles to the gastric wall, as with mucosal or epithelial lesions (a). Subepithelial lesions produce more obtuse angles with the gastric wall and preserve the mucosal pattern, as may be seen with lesions of submucosal origin (b). If growth extends beyond the gastric wall outwardly, an exogastric component is said to be present (c). Extrinsic lesions have similar intraluminal appearance to intramural lesions, but being outside the stomach, they may also exhibit mass effect on the gastric wall (d)
Mucosal Lesions
Benign Mucosal Tumors
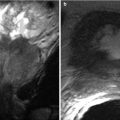
Mucosal lesions in the stomach include benign polypoid lesions, such as post-inflammatory, adenomatous, and hamartomatous polyps (Fig. 9.8). Post-inflammatory polyps are most common. They are typically small (<1 cm), of smooth and sessile morphology, and multiple in number. They have no malignant potential but may be associated with atrophic gastritis and gastric cancer. Adenomatous polyps are usually larger (>2 cm) and solitary, unless associated with familial polyposis syndromes, in which case colonic polyps are also seen. These include familial adenomatous polyposis, as well as Gardner’s syndrome (associated with osteomas and extraintestinal soft tissue tumors) and Turcot’s syndrome (associated with cerebral gliomas). Morphologically, adenomatous polyps are lobulated with a sessile or pedunculated base. Adenomas also have malignant potential [3, 14]. Gastric hamartomas are seen in the setting of Peutz-Jeghers, juvenile polyps, Cowden’s syndrome, and Cronkhite-Canada syndrome. Despite the lack of malignant potential in hamartomas, Peutz-Jeghers syndromes and Cowden’s syndrome are associated with extraintestinal malignancies, while juvenile polyps are associated with gastrointestinal cancer [15].
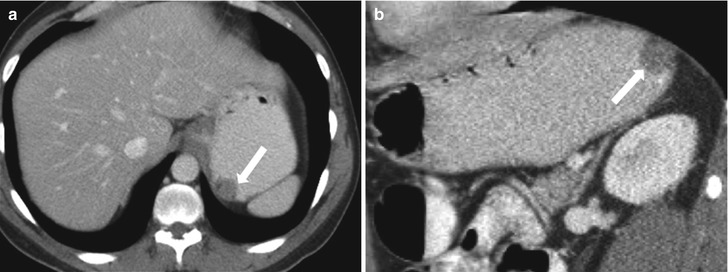

Fig. 9.8
Contrast-enhanced axial (a) and coronal (b) CT with positive enteric contrast demonstrates a polypoid nodule of the gastric fundus (arrows). Note the acute angle between the lesion and the gastric wall, indicating a mucosal location of this lesion
Gastric Adenocarcinoma
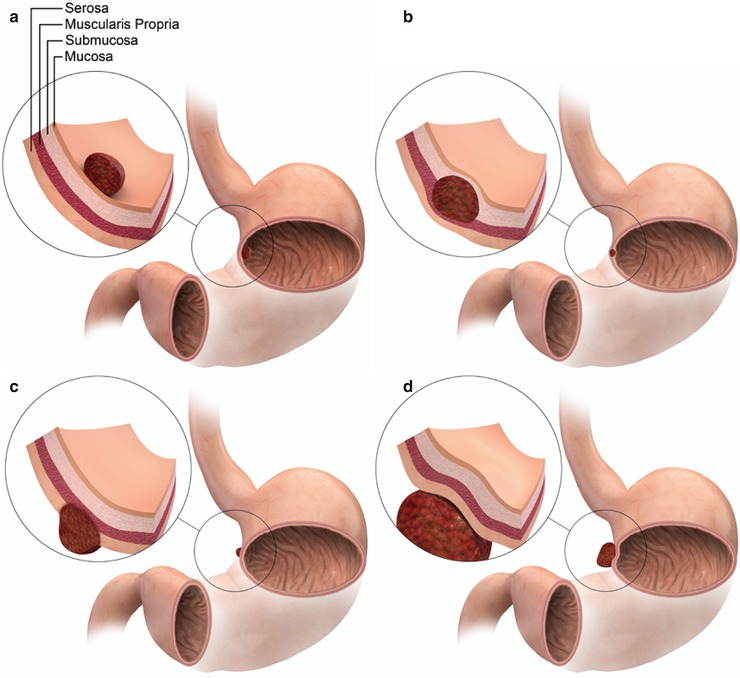
Early gastric adenocarcinoma usually presents as a mucosal lesion with limited invasion. These cancers are classified by according to morphology (Fig. 9.9): type I lesions are polypoid lesions extending >5 mm beyond the normal gastric wall, whereas Type IIa lesions protrude <5 mm. Type IIb are flat lesions appearing as mucosal irregularities. Type IIc lesions are depressed but confined to the mucosa, while type III lesions are frankly ulcerated lesions disrupting the muscularis mucosa but confined to the submucosa [3]. Early diagnosis of subtle lesions, especially type II and III lesions, may require more sophisticated tools, like VG [8].
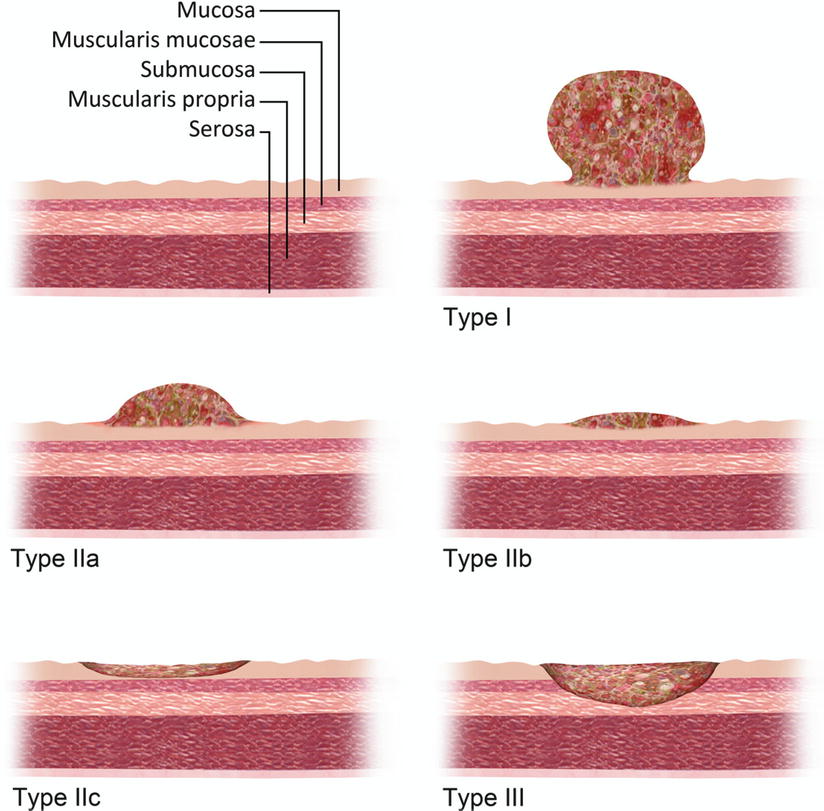

Fig. 9.9
Early gastric cancer: type I lesions are polypoid and extend >5 mm beyond the normal gastric wall. Type IIa lesions protrude <5 mm. Type IIb are flat lesions. Type IIc lesions are depressed but confined to the mucosa. Type III lesions are frankly ulcerated lesions disrupting the muscularis mucosa but confined to the submucosa [3]
Gastric cancer may continue to grow in mass-like fashion (Fig. 9.10), or further involvement may be in the form of segmental of diffuse gastric wall involvement [5]. Focal, eccentric, or prominent wall thickening (>1 cm) may suggest a malignant lesion, but no combination of these is both sensitive and specific for cancer [16]. More advanced gastric cancers penetrate the muscularis propria and beyond. Accurate assessment of local disease is key to effective therapy. Thus, CT criteria for local staging closely parallel the American Joint Committee on Cancer’s TNM staging (Fig. 9.11). Most important is detection of serosal invasion, which is a poor prognostic indicator. Hazy, linear, or reticular densities at the perigastric fat interface suggest such involvement (Fig. 9.12) [17]. Aside from the common presentation of a mucosal lesion or gastric mass, gastric adenocarcinoma may commonly present as a malignant ulcer or wall thickening, possibly with linitis plastica (Tables 9.2 and 9.3).
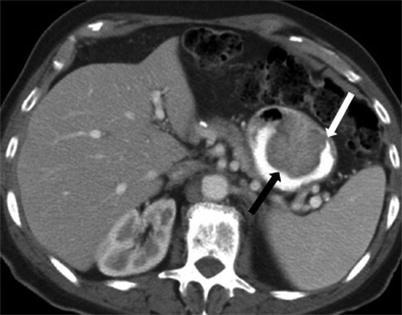
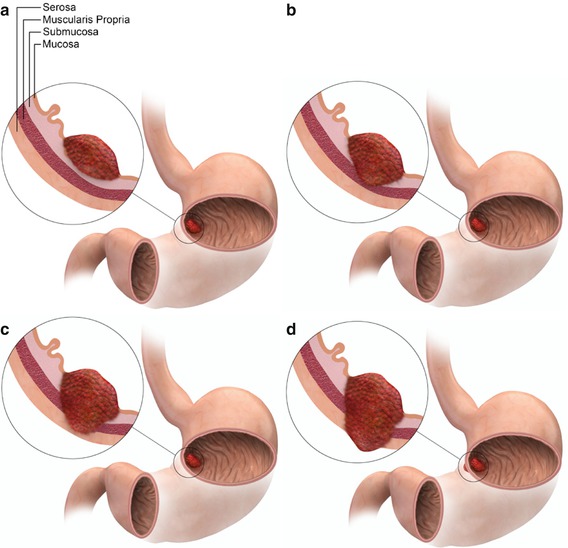
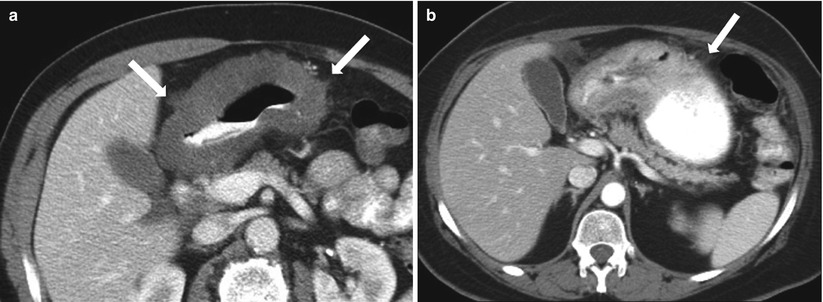

Fig. 9.10
Contrast-enhanced axial CT demonstrates a polypoid mass in this patient with adenocarcinoma (black arrow). Note acute angles of the lesion with the gastric wall (white arrow)
CT feature | CT stage |
|---|---|
Normal gastric wall | T0 |
Nontransmural enhancement inner/middle wall enhancement only | T1 |
Transmural enhancement with smooth outer wall | T2 |
Transmural enhancement with irregular wall or reticular/linear perigastric opacities | T3 |
Extension to adjacent organs | T4 |
T – primary tumor | TX: primary tumor cannot be evaluated |
T0 (T plus zero): no evidence of a primary tumor | |
Tis: carcinoma in situ | |
T1: tumor limited to submucosa | |
T2: tumor invades greater than half the gastric wall thickness | |
T3: tumor involves entire gastric wall without extension beyond serosa | |
T4: tumor extends beyond serosa to organs surrounding the stomach | |
N – regional lymph nodes | NX: regional lymph nodes cannot be evaluated |
N0: no regional lymph node spread | |
N1: spread to perigastric lymph nodes within 3 cm of primary tumor | |
N2: spread to regional tumors more than 3 cm from primary tumor | |
N3: involvement of para-aortic hepatoduodenal or more distant regional lymph nodes | |
M – metastasis | MX: distant metastasis cannot be evaluated |
M0: no distant metastasis | |
M1: distant metastasis |

Fig. 9.11
T-staging of gastric adenocarcinoma; Tumors confined to the gastric wall submucosa are categorized as T1 (a). T2 tumors involve greater than half of the gastric wall (b), whereas T3 tumors cover the thickness of the gastric wall without extension beyond the serosa (c). Once there is extension beyond the serosa to adjacent organs, the tumor is T4 (d)

Fig. 9.12
Contrast enhanced CT images of diffuse (a) and segmental (b) gastric adenocarcinoma with perigastric densities suspicious for serosal invasion (arrows)
Perigastric and extraperigastric lymphadenopathy may be considered when lymph node short-axis reaches >6 mm and >8 mm, respectively, or when lymph nodes have eccentric or absent fatty hila, increased or heterogeneous enhancement, or round shape (longitudinal-transverse diameter ratio <1.5).
Extraperigastric lymphadenopathy (stations 12p-16) (Fig. 9.13) is considered distant metastasis, but the liver is the most common site of hematogenous spread. Less commonly, gastric adenocarcinoma metastasizes to the lungs, adrenal glands, kidneys, bones, and brain [3, 7]. Clues to peritoneal metastasis include ascites, peritoneal soft tissue lesions, peritoneal thickening, and hyperenhancement (Fig. 9.14) [3].
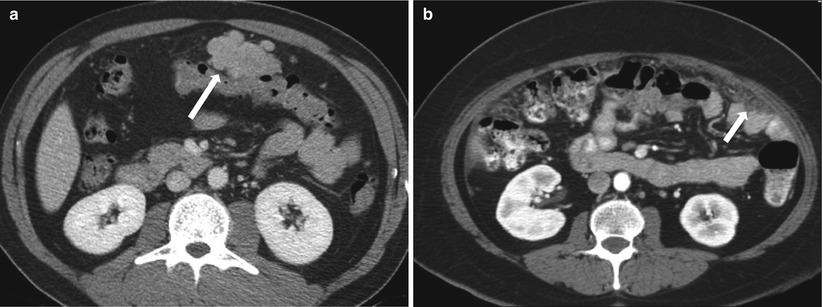

Fig. 9.14
Contrast enhanced axial CT images demonstrate soft tissue nodules (a) and mesenteric haziness (b) indicative peritoneal seeding (arrows) in two different patients with gastric carcinoma
Spread via the peritoneum can also lead to “drop metastases” involving small bowel, sigmoid colon, and rectum, as well as ovaries. Ovarian metastases typically from a gastrointestinal source, but also from other primary tumors, such as the breast or lung, are known as Krukenberg tumors (Fig. 9.15). Krukenberg tumors may be seen as unilateral or bilateral mixed cystic and solid ovarian lesions with diameters ranging from 1 to 20 cm [2]. Metastases to uterus can rarely occur.
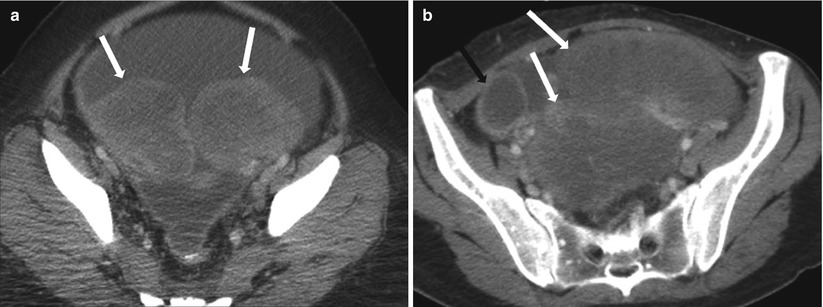

Fig. 9.15
Contrast enhanced axial CT images demonstrate side-by-side (a) and anterior-to-posterior (b) Krukenberg tumors in two different patients (white arrows). Another right adnexal cystic metastatic implant is seen in (b, black arrow)
Detection of extraperigastric lymph nodes and distant solid organ metastases may be enhanced with the use of FDG-PET (Fig. 9.16). Small, but hypermetabolic lesions may be better detected with FDG-PET. Distant metastases outside the abdominopelvic coverage of CT may be detected by whole-body FDG-PET. Diffuse or multifocal abdominopelvic FDG uptake may suggest peritoneal metastases [7].
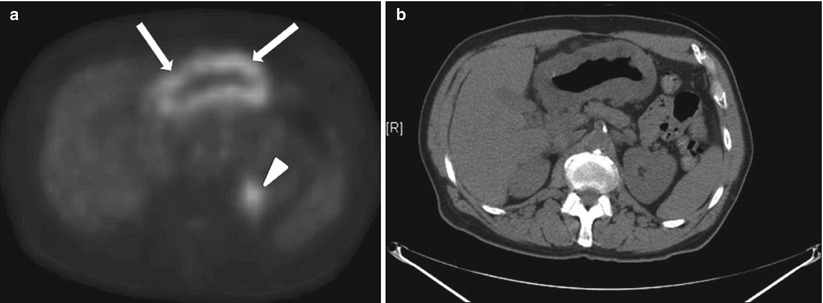

Fig. 9.16
FDG-PET (a) and corresponding CT (b) of diffuse gastric cancer with increased FDG uptake in the gastric wall (white arrows). No evidence of lymphadenopathy is present on this patient. Focus of increased FDG uptake on the left retroperitoneum corresponds to excretion of FDG by the left kidney (white arrowhead)
Other Polypoid or Nodular Malignant Tumors
Predominantly submucosal processes such as gastric lymphoma and gastrointestinal stromal tumors can uncommonly have a nodular or polypoid appearance [18, 19]. Similarly, hematogenous metastases like malignant melanoma and Kaposi’s sarcoma typically grow intramurally but may have a nodular appearance that mimics mucosal lesions (Fig. 9.17). These two entities may be distinguished by their hyperenhancement, however [6].
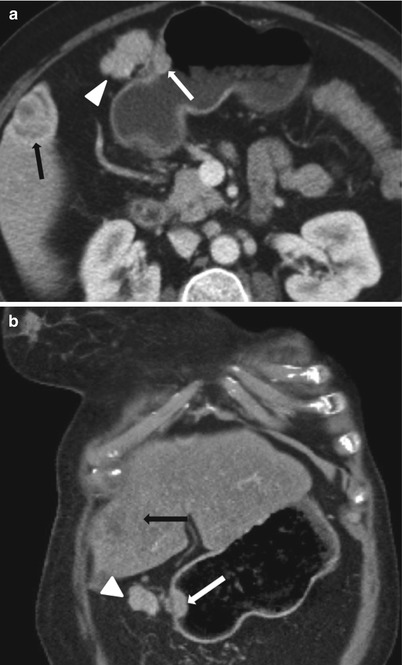
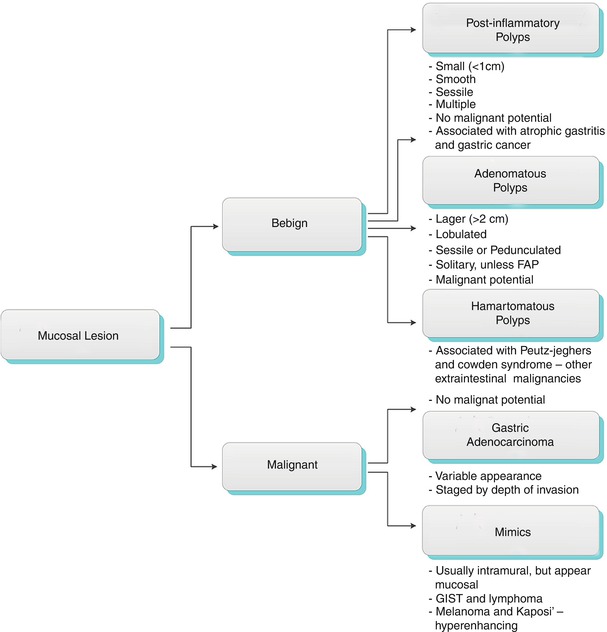

Fig. 9.17
Contrast-enhanced axial (a) and coronal (b) CT images demonstrate peritoneal lesion (white arrowhead), nodular gastric lesion (white arrows), as well as extragastric hepatic lesion (black arrows) in a patient with metastatic breast cancer

Algorithm 9.3 Imaging findings of mucosal gastric lesions
Intramural (Subepithelial/Submucosal) Lesions
Benign Submucosal Tumors
Benign submucosal tumors typically present as small, smooth, hyperenhancing intramural soft tissue lesions (Figs. 9.18 and 9.19). These include mesenchymal tumors, most commonly gastrointestinal stromal tumors (GIST). GIST expresses KIT (CD117), a tyrosine kinase receptor, which is what distinguishes it from other mesenchymal tumors. The diagnosis typically requires histopathologic evaluation to assess for malignant GIST. Hemangiomas, leiomyomas, neural tumors, such as schwannomas or neuromas, and neuroendocrine tumors, such as carcinoids, are less common benign entities [5, 6, 19]. Pancreatic rests may have similar characteristics. These may be indistinguishable on CT or MRI.
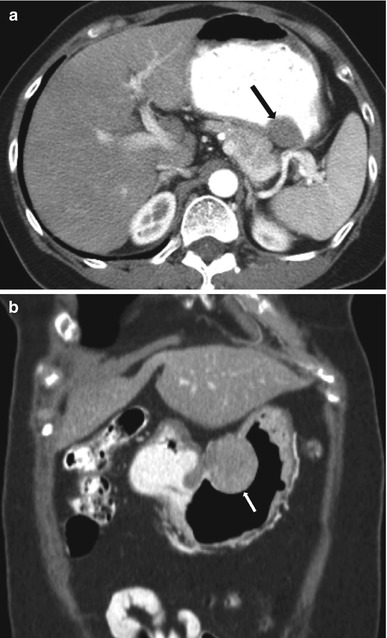
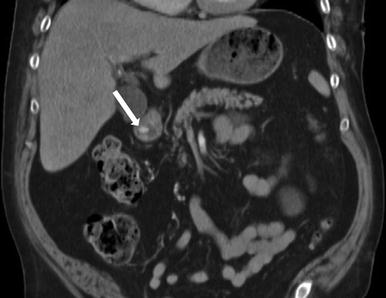

Fig. 9.18
Contrast-enhanced axial CT (a) with positive enteric contrast demonstrates a focal benign GIST (black arrow) with smooth borders and more obtuse angles. A similar appearance is appreciated on coronal images (b) of a different patient with GIST at the lesser curvature of the stomach (white arrow)

Fig. 9.19
Contrast-enhanced coronal CT images demonstrate a hyperenhancing subepithelial lesion (arrow) compatible with carcinoid
Lipomas have characteristic features on CT and MRI with a typical fat density on CT (Fig. 9.20), T1 hyperintensity on nonfat-suppressed pulse sequences, and hypointensity following fat suppression [6].
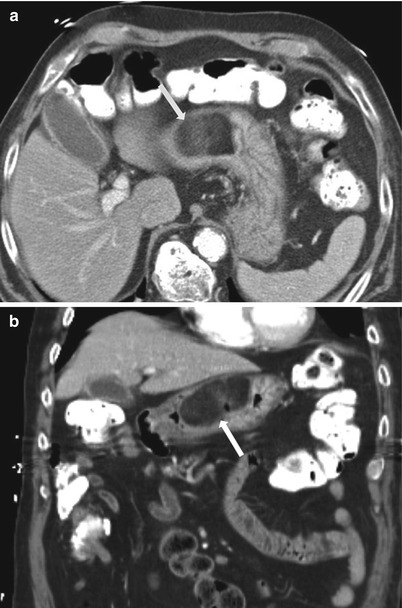

Fig. 9.20
Contrast-enhanced axial (a) and coronal (b) CT images demonstrate intraluminal gastric lipoma with characteristic fat density on CT (white arrows)
Hyperdensity on non-contrast CT may also suggest an intramural hematoma, particularly in the setting of trauma or bleeding diathesis.
Malignant Submucosal Tumors
Lymphoma
Lymphoma infiltrates the submucosal and deep mucosal layer of the gastric wall. Among its various presentations, gastric lymphoma can manifest as a focal intramural mass. Multiple, sometimes confluent nodules may be seen with low-grade MALT lymphomas [14]. More commonly, lymphoma thickens a large segment of the gastric wall, if not the entire stomach, producing segmental or diffuse thickening.
GIST
The diagnosis of malignant GIST requires histopathologic analysis, but certain characteristics suggest malignancy, which develops in 10–30 % of these lesions. These include exogastric growth, diameter >5 cm, central necrosis, and extension to other organs [3, 5].
Commonly, GIST may exhibit central necrosis, cystic degeneration, or hemorrhage, as well as ulceration (Fig. 9.21). There may be peripheral enhancement of viable tumor, while the centrally necrotic or cystic spaces will show low attenuation on CT or low T1 and high T2 signal intensity (SI) on MR. Hemorrhage has variable CT and MR characteristics, depending on the age of the blood products. Another differentiating feature is a lack of lymphadenopathy [19]. Direct invasion of adjacent structures and low-attenuation, necrotic metastases to the liver or lung may also be observed. Because of their KIT expression, these tumors are highly receptive to treatment with KIT tyrosine kinase inhibitors, such as imatinib.
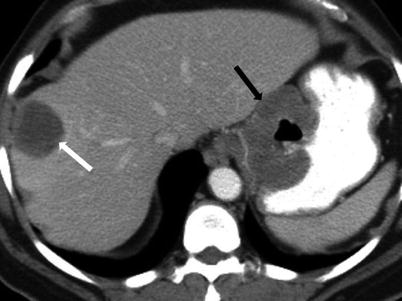

Fig. 9.21
Contrast-enhanced axial CT image demonstrates typical characteristics of a malignant GIST, including a large, exogastric mass with ulceration (black arrow), which may relate to central necrosis and erosion. Also note metastatic hepatic lesion (white arrow)
Gastric Carcinoid
An important differential consideration of submucosal mass is a gastric carcinoid. Three types of gastric carcinoids are described; types 1 and 2 are typically small (<3 cm), associated with enterochromaffin-like cell hyperplasia and are more likely to be benign. The latter may be multifocal and associated with Zollinger-Ellison syndrome in the setting of multiple endocrine neoplasia, type 1. Thus, the diagnosis may be suggested by the presence of multiple nodules in association with gastric wall thickening. While type 2 carcinoids may spread to regional lymph nodes, they are unlikely to spread distally, cause carcinoid syndrome, or be fatal. Conversely, type 3 tumors are large, may ulcerate, and are likely to have distant metastases, with carcinoid syndrome being possible if hepatic spread is present [6, 19].
As previously discussed, hematogenous metastases, such as breast, melanoma, and Kaposi’s sarcoma, also typically develop in the submucosal layer, but they can appear as mucosal or intramural lesions on imaging [6].
Secondary and Adult Idiopathic Hypertrophic Pyloric Stenosis
Hypertrophic pyloric stenosis (HPS) is known as a congenital lesion that presents in infancy [20]. In adults, idiopathic HPS may also present as focal wall thickening. More commonly, adult pyloric stenosis results secondary to other causes of wall thickening, such as peptic ulcer disease. Regardless of the cause, this condition may mimic gastric cancer on CT [5, 21].
Extrinsic Space-Occupying Lesions
Various space-occupying lesions can produce mass effect on the stomach. These are usually readily apparent on cross-sectional imaging and may have a clear fat plane with the stomach with an intact gastric wall, although some may erode the gastric wall [6]. Extrinsic lesions include hepatomegaly and splenomegaly, as well as liver, pancreatic, splenic, adrenal, and renal focal mass lesions. Omental metastases and pancreatic and colon cancer may invade the gastric wall.
Other extrinsic lesions are cystic in nature and include pancreatic pseudocysts, duplication cysts, and abscesses (Fig. 9.22). A dilated gastric diverticulum can simulate an extrinsic cystic mass.
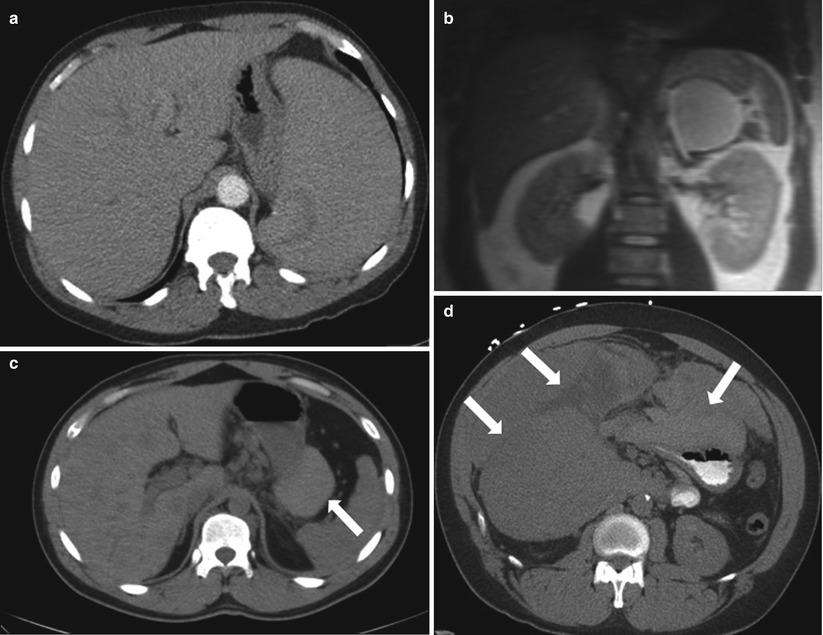
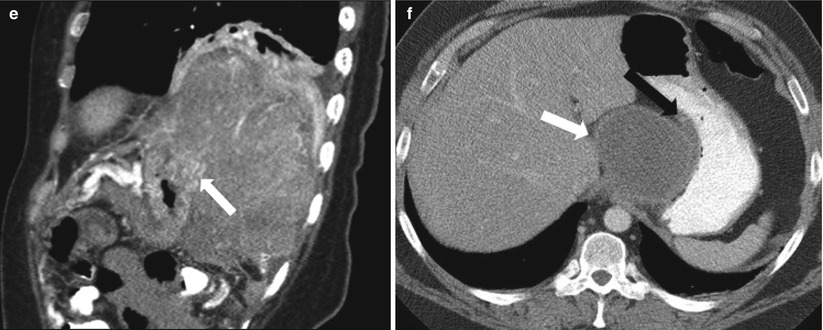


Fig. 9.22
Contrast-enhanced axial CT image (a) demonstrates gastric compression due to hepatosplenomegaly. Coronal ssFSE image (b) shows inferior mass effect on the stomach from a left adrenal cyst. Unenhanced axial CT images (c, d) demonstrate perigastric hematoma (white arrows). The latter (d) is secondary to rupture of a large, exophytic HCC seen arising from segment 6 of the liver. Contrast-enhanced sagittal CT (e) image demonstrates mass effect and frank invasion (white arrow) of the stomach by a large renal cell carcinoma. Contrast-enhanced CT (f) demonstrates a gastric duplication cyst (white arrow) displacing and distorting the stomach with obtuse angles typical of an extrinsic lesion (black arrows)
Pseudomass
Adequate distension and close inspection are also needed to exclude pseudolesions from gastric folds or focal wall thickening (Fig. 9.23). A gastric bezoar is another space-occupying lesion but easily discernible on CT, where a rounded mass with flocculent air is usually depicted [22].
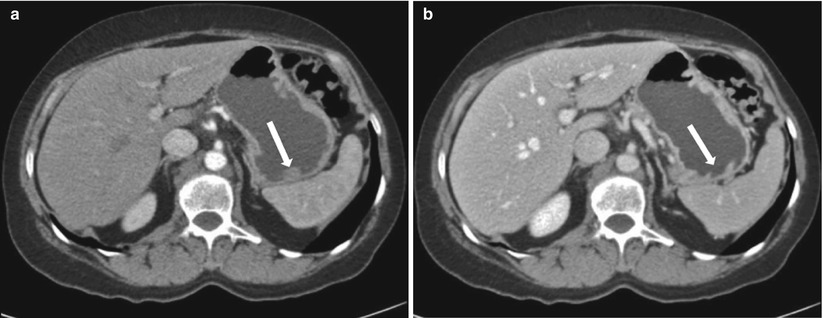
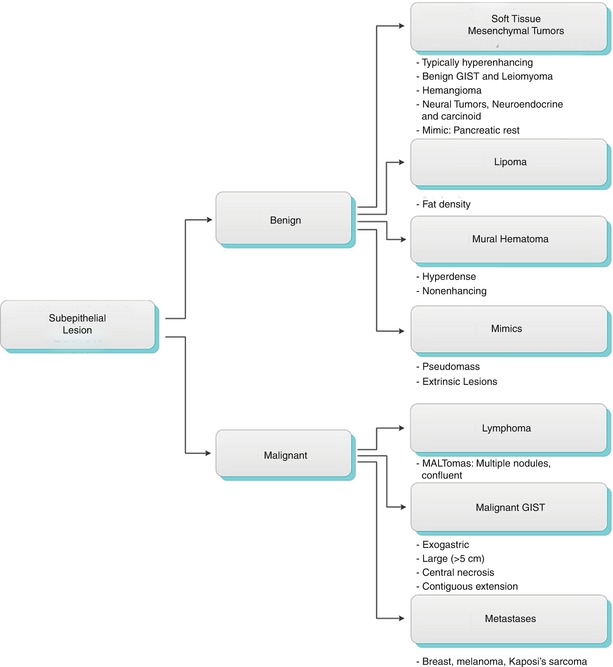

Fig. 9.23
Contrast-enhanced axial CT images in the portal venous (a) and delayed (b) phases of contrast enhancement. Apparent nodular lesion in the gastric fundus seen on (a, white arrow) does not persist on (b). This apparent lesion is due to thickening of an undistended gastric fold

Algorithm 9.4 Imaging findings of subepithelial gastric lesions
Segmental and Diffuse Gastric Wall and Fold Thickening
Involvement of the gastric wall may be diffuse, rather than focal. This may involve the entire stomach or just a segment. Whether segmental or complete, the finding of a diffusely thickened gastric wall can have an extensive differential. As with more focal wall thickening, adequate distension is key to avoiding false positives. Overdistension can also efface the gastric rugae. A wall thickness of <5 mm between rugae can be considered normal.
Gastritis and Inflammatory Diseases
Thickening of gastric folds is most commonly due to edema in the setting of gastritis by any cause. Most commonly associated with Helicobacter pylori infection, mucosal inflammation may also result from aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), alcohol or coffee ingestion, as well as stress, chemotherapy, and radiation exposure, among others. Most commonly located in the antrum, gastritis can be diffuse or proximal [3, 5].
Mucosal hyperemia may be evidenced by smooth early mucosal hyperenhancement. On CT, decreased attenuation to near-water density may accompany thickened folds due to gastritis folds (Fig. 9.24). The alternating hyperenhancing mucosa with hypoattenuating edematous mucosa has been described as a “water halo sign” [23] or “target sign” and typically suggests inflammatory disease. A potential mimic is mucinous adenocarcinoma or edematous neoplasm, which can also produce low-attenuation folds [2, 24].
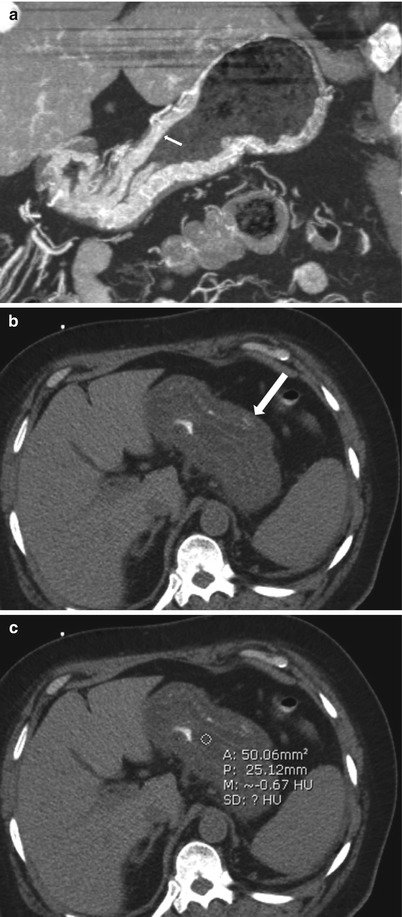

Fig. 9.24
Contrast-enhanced axial CT image (a) demonstrates mucosal hyperenhancement (white arrow) in a patient with H. pylori gastritis. Unenhanced axial CT images of a different patient without (b) and with (c) density measurements demonstrate thickened gastric folds (white arrow, a) of fluid density (circle, −0.67 HU), consistent with edema seen in gastritis
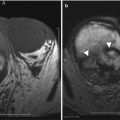
Uncommonly, gastritis may be secondary to Zollinger-Ellison syndrome, where gastrin-producing tumors are the culprit for gastric acid hypersecretion and inflammatory changes of the stomach and duodenum. Pancreatic islet cell tumors and post-bulbar ulcers may be seen in these cases (Fig. 9.25).
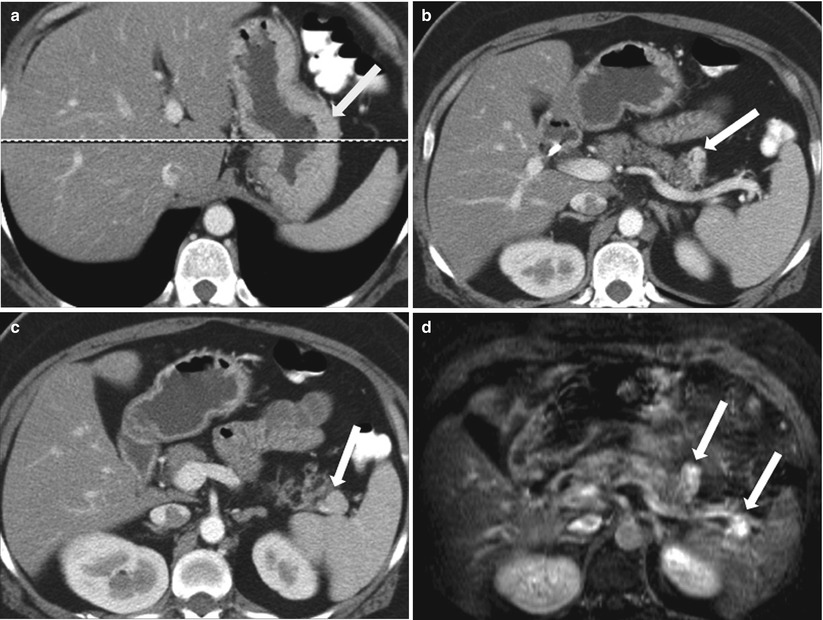

Fig. 9.25
Contrast-enhanced axial CT images (a–c) and fat-suppressed T2 axial images of a patient with a history of MEN I syndrome and elevated gastrin levels. Gastric wall thickening consistent with gastritis (a, white arrow) seen along with hyperenhancing pancreatic lesion (b, c, white arrows), with hyperintense signal intensity on T2 weighted (d, white arrows), suggestive of gastrinoma
Other less common inflammatory conditions, such as tuberculosis, Crohn’s disease, eosinophilic gastritis, Ménétrier disease, and caustic injury, may cause similar findings, with the latter two producing marked amounts of wall edema (Fig. 9.26). Both, the degree of thickening and the lobulated folds of Ménétrier disease, may mimic a neoplastic process.
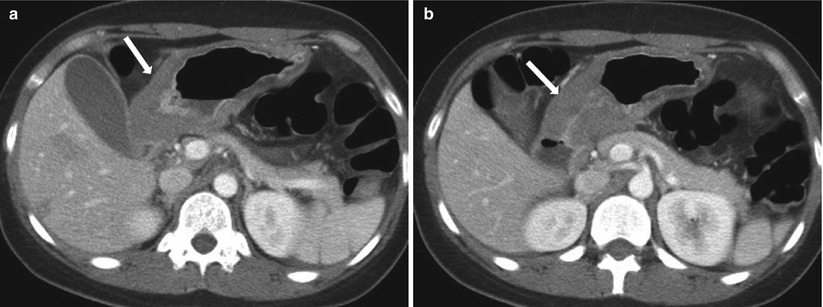





Fig. 9.26
Axial contrast enhanced axial CT images at the level of the gallbladder (a) and just inferior to it (b) demonstrate marked gastric wall thickening with mucosal hyperenhancement (white arrows) in this patient with eosinophilic gastritis
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree