8 The Vestibulocochlear Nerve, with an Emphasis on the Normal and Diseased Internal Auditory Canal and Cerebellopontine Angle
The vestibulocochlear nerve is the eighth cranial nerve (CN VIII) and is responsible for the transmission of electrical impulses from cochlear hair cells of the inner ear to the cochlear nuclear complex in the brainstem. The vestibulocochlear nerve passes through the internal auditory canal (IAC) and cerebellopontine angle (CPA), forming the extraaxial component of the central auditory pathway. Dysfunction in this pathway results in sensorineural hearing loss (SNHL). The most common abnormality when imaging a child with SNHL is a congenital inner ear malformation,1 whereas the most common cause of unilateral or asymmetric SNHL in an adult is a schwannoma of the vestibulocochlear nerve.
When presented with a request to image a patient with a history of SNHL, dedicated imaging is required of the cochlea, IAC, and the CPA. If additional cranial neuropathies are present in association with the SNHL, such as trigeminal neuralgia or facial nerve palsy, then a large CPA mass, a leptomeningeal process, or a brainstem abnormality such as multiple sclerosis must be considered. SNHL in association with cerebellar-type neurological findings such as ataxia and weakness suggests the presence of a posterior fossa and/or brainstem abnormality. Bilateral SNHL or complex auditory dysfunction, such as auditory agnosia, suggests a process that is more centrally located along the auditory pathway.
This chapter begins with a discussion of the anatomy of the retrocochlear auditory pathway from the cochlear modiolus to the primary auditory cortex of the superior temporal gyrus. Magnetic resonance imaging (MRI) plays an important role in the evaluation of patients with SNHL, and a discussion of the currently available techniques for high-resolution imaging of the inner ear, IAC, and CPA follows. The congenital and acquired pathologic conditions that may be found along the central acoustic pathway from the IAC to the auditory cortex are then discussed, with an emphasis on the most commonly found lesion causing adult SNHL, the vestibular schwannoma. Finally, lesions of the petrous apex are discussed, both as potential causes of hearing loss and as they may be unexpectedly discovered at the time of temporal bone imaging for SNHL or conductive hearing loss (CHL).
Normal Anatomy of the Auditory Pathway
The cell bodies of the cochlear nerve are located in the modiolus, the spongy bone at the center of the cochlea. These spiral ganglia are bipolar, with one axon arising from the cochlear organ of Corti that receives auditory information and one axon that transmits this information to the brainstem. The cochlear nerve is formed from those axonal fibers that exit the modiolus of the cochlea through a small bony opening called the cochlear canal or cochlear aperture.2–4 This cochlear nerve enters the anteroinferior quadrant of the fundus of the IAC and courses medially toward the CPA and brainstem auditory nuclei (Fig. 8.1). Because the cochlear nerve is a sensory nerve, the centripetal pathway toward the brainstem is considered the anterograde direction. The bony contours of the cochlear aperture and the IAC are readily appreciable on temporal bone CT imaging when acquired at ≤ 1 mm thick slices using a bone algorithm (Fig. 8.2). The internal auditory canal with its lateral “dead-end” fundus and medial internal auditory meatus, or porus acusticus, is roughly cylindrical in shape. There are variations between patients of the vertical height and transverse diameter, with the average height (from tomographic studies) measured at 4 mm (range 2 to 8 mm). More important, the IAC is essentially symmetric in patients with a difference in dimensions of 1 mm in 99% of patients and 1 to 2 mm in 1% of patients.5 The IAC is directed at 45 degrees to the long axis of the petrous bone, which is at 45 degrees to the true sagittal plane of the skull.
At the fundus, or lateral aspect of the IAC, the three components of the vestibulocochlear nerve are distinct separate nerves: the cochlear nerve lies in the anteroinferior aspect of the IAC, the inferior vestibular nerve posterior to this, and the superior vestibular nerve in the superior aspect of the fundus, posterior to the facial nerve (Fig. 8.3 and Fig. 8.4). In 90% of normal cases, evaluation of the nerves at the fundus of the IAC shows the cochlear nerve to be larger than the superior or inferior vestibular nerves, and of similar size or larger than the facial nerve in nearly two thirds of normal cases.6 At the medial aspect of the IAC the superior and inferior vestibular nerves and the cochlear nerve join to form the vestibulocochlear nerve. There is significant variability between individuals in the exact site of union of these three nerves (cochlear, superior, and inferior vestibular); however, the location is relatively symmetric within individuals.6,7 At the level of the porus acusticus, CN VIII is usually found as one nerve bundle with an elongated, almost rectangular, cross-sectional shape (Fig. 8.5). Oblique sagittal T2-weighted magnetic resonance images (T2WIs) performed perpendicular to the course of the IAC allow appreciation of the anatomical arrangement of the facial and cochleovestibular nerves, as they are hypointense and are surrounded by the marked hyperintensity of the cerebrospinal fluid (CSF) that fills the IAC.6,7
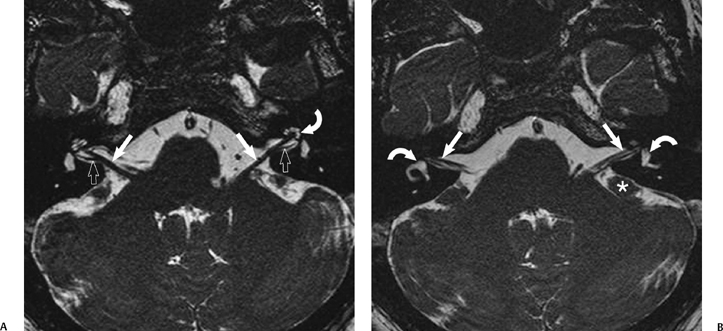
Fig. 8.1 Internal auditory canal (IAC) anatomy. Heavily T2-weighted FIESTA sequence through the IACs (A) shows the normal anatomy with the thick vestibulocochlear nerve (VCN) in the CPA and IAC (arrows), reaching the modiolus of the fluid-filled cochlea (curved arrow). Branching of the VCN can be seen in the axial plane (open arrows). (B) More superiorly, the facial nerve is seen in the CPA and IAC (arrows). The fluid-filled vestibule and canals are also clearly seen bilaterally (curved arrows). The flocculus of the cerebellum is a rounded “mass-like” structure in the CPA (*).
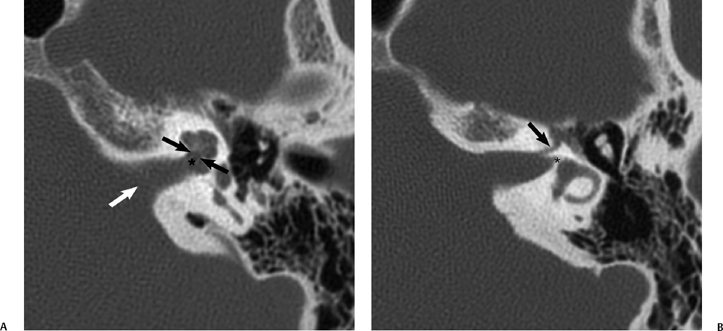
Fig. 8.2 Internal auditory canal (IAC) anatomy by computed tomography (CT). (A) Axial CT through the mid-IAC demonstrates the porus acusticus (white arrow), the fundus (*), and the cochlear aperture (black arrows). (B) Axial CT through the superior aspect shows the labyrinthine segment of the facial nerve (arrow) and the small canal for the nerve to the horizontal semicircular canal (*).
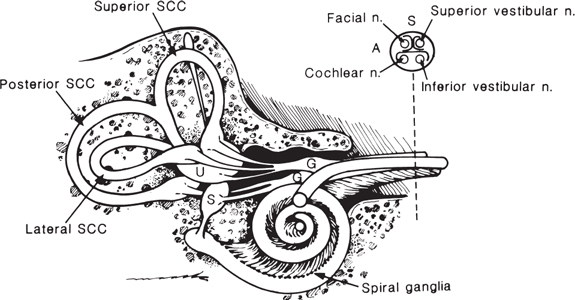
Fig. 8.3 Drawing of membranous labyrinth and distal internal auditory canal (IAC) shows the relationship of the cochlea to other labyrinth structures. Inset shows the position of the cochlear nerve in the anteroinferior quadrant of the IAC. It is separated from the facial nerve by the crista falciformis in the fundus of the IAC. (SCC, semicircular canal; U, utricle; S, saccule; G, vestibular ganglion; A, anterior; S, superior.)
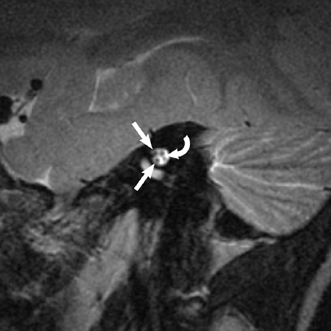
Fig. 8.4 Normal nerves at the internal auditory canal (IAC) fundus. Oblique sagittal T2-weighted magnetic resonance image demonstrates the four nerves at the fundus of the IAC: the facial nerve superiorly and anteriorly, the cochlear nerve anteroinferiorly, and the superior and inferior vestibular nerves posteriorly (curved arrow).
On computed tomography (CT) evaluation of the IAC, the nerve elements cannot be identified, though their location can be inferred using bony landmarks. The superior half of the IAC fundus (containing the facial nerve with the nervus intermedius and the superior vestibular nerve) is separated from the inferior IAC (containing the cochlear and inferior vestibular nerves) by the crista falciformis (falciform crest). This is a horizontal bony bar that is evident on coronal CT imaging, and frequently also seen as an area of signal void on T2WIs , which lies at or above the midpoint of vertical canal height (Fig. 8.6).5 A vertical crest called Bill’s bar separates the superior fundus above the crista falciformis into anterior and posterior portions containing the facial nerve and superior vestibular nerve, respectively. Bill’s bar is described as consisting of a thin layer of arachnoid tissue that may have an osseous medial component, but it itself is not discernible at high-resolution CT (HRCT) or MRI.8–10 The superior vestibular nerve has several branches that supply the superior and horizontal semicircular canals and the saccule. The inferior vestibular nerve supplies the utricle and has a separate branch to the ampulla of the posterior semicircular canal. This posterior ampullary nerve (or “singular nerve”) traverses the singular canal, which can be identified on axial temporal bone CT arising from the mid to distal portion of the IAC (Fig. 8.7). The singular canal is an important surgical landmark identified for a retrosigmoid approach for resection of a vestibular schwannoma. It should not be mistaken for a fracture on CT.
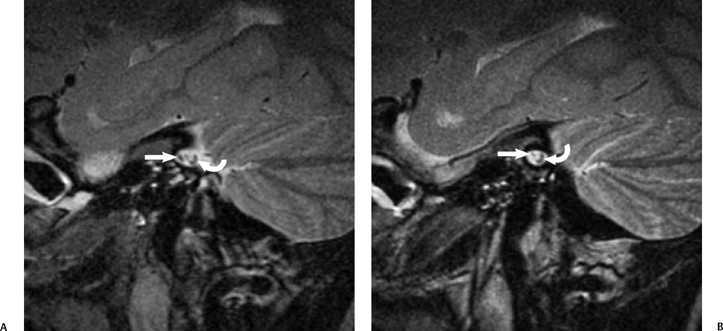
Fig. 8.5 Nerves in the Internal auditory canal (IAC). Sagittal T2-weighted magnetic resonance image through the IAC. (A) At the porus, the facial nerve is anterior (arrow), with the vestibulocochlear nerve (VCN) posterior (curved arrow) as an elongated thick bundle. (B) As it is imaged more laterally in the mid-IAC, the VCN (curved arrow) starts to elongate more, before becoming three nerves, as seen in Fig. 8.4 at the more lateral aspect of the IAC.
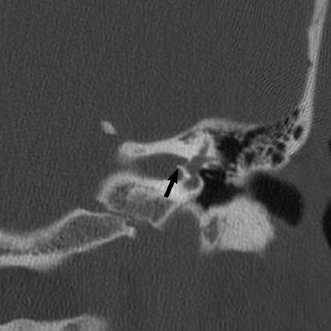
Fig. 8.6 Crista falciformis. Coronal computed tomography image shows the horizontal crista falciformis (arrow) that divides the internal auditory canal into superior and inferior halves.
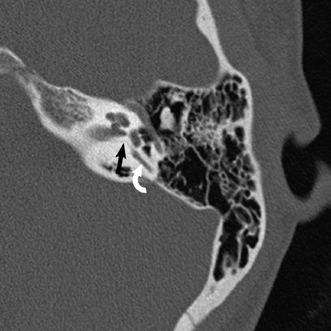
Fig. 8.7 Singular canal. Axial computed tomography scan at the level of the modiolus shows this tiny canal (arrow) from the posterosuperior aspect of the internal auditory canal to the ampulla of the posterior semicircular canal (curved arrow)
As the vestibulocochlear nerve traverses the CPA, it is posterior to CN VII, and both of these nerves are intimately related to the anteroinferior cerebellar artery (AICA). This vessel is a branch of the basilar artery, which is ventral and inferior to CN VII and CN VIII. On axial T2WIs, the AICA is seen as a flow void. In two thirds of normal cases this artery loops into the IAC or projects to the porus acusticus (Fig. 8.8).11 Up to four internal auditory arteries arise from the AICA to supply the inner ear.12
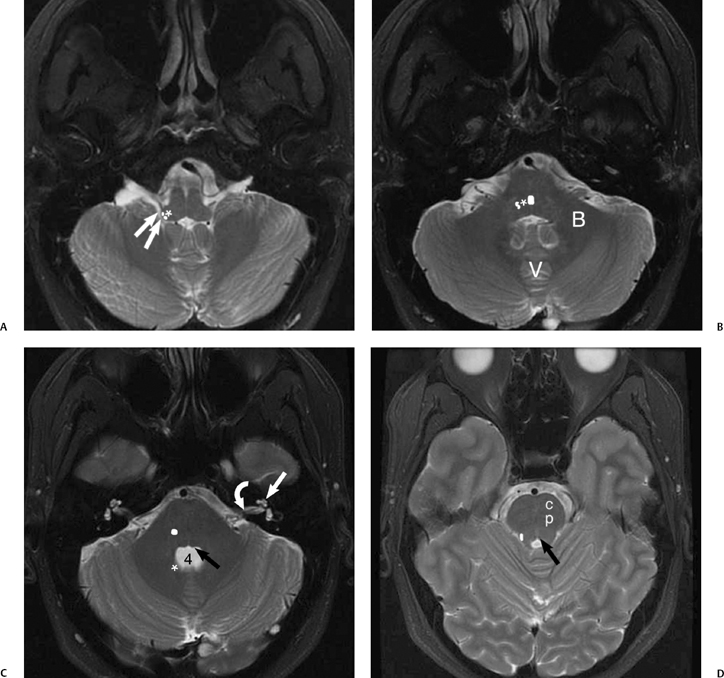
The vestibulocochlear nerve enters the brainstem at the root entry zone of the upper lateral medulla immediately superficial to the inferior cerebellar peduncle.13,14 The cochlear component of CN VIII synapses with two nuclei known as the cochlear nuclear complex. The dorsal nucleus receives high-frequency signals from the lower turns of the cochlea, and the ventral cochlear nucleus receives lower frequencies from the upper cochlear turns.13 The cochlear nuclear complex produces a bulge of the medulla into the lateral recess of the fourth ventricle and the foramen of Luschka (Fig. 8.9). Additional useful landmarks for identifying the site of the cochlear nuclear complex on imaging studies are the cerebellar flocculus, which lies posterolateral to the nerve complex, and the intervening choroid plexus, which may protrude through the foramen of Luschka from the fourth ventricle (Fig. 8.10). Four vestibular nuclei are also located in the brainstem medial to the cochlear nuclei.15
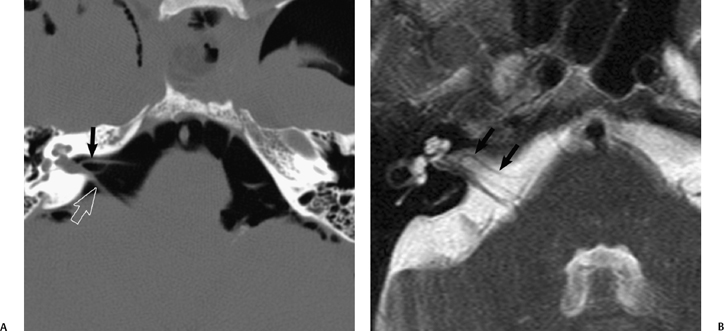
Fig. 8.8 Anteroinferior cerebellar artery (AICA) loop. (A) Axial unenhanced computed tomography image performed after craniotomy shows air in the cerebellopontine angles and air outlining a right AICA (arrow) as it loops into the internal auditory canal (IAC) adjacent to CN VII and CN VIII (open arrow). (B) Axial heavily T2-weighted constructive interference in steady-state (CISS) sequence in a different patient similarly demonstrates a vascular AICA loop (arrows) in the IAC.
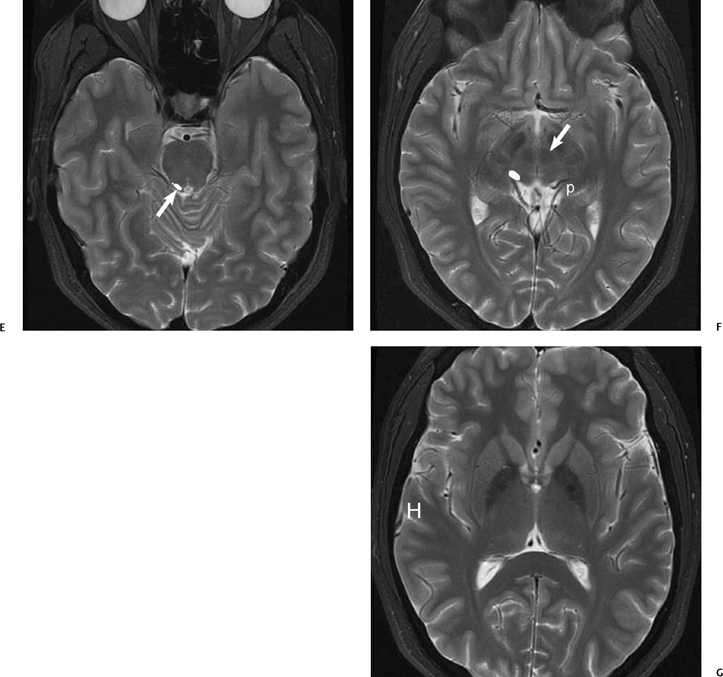
Some second-order neurons arising from the dorsal and ventral nuclei of the cochlear nuclear complex cross in the trapezoid body of the medulla, while some synapse in the superior olivary nucleus of the medulla. These fibers then ascend to the contralateral inferior colliculus of the midbrain. Other fibers do not cross, but ascend on the ipsilateral side to the inferior colliculus. These ascending crossed and uncrossed fibers form the lateral lemniscus of the pons (Fig. 8.11). There is further crossing of fibers in the commissure of the inferior colliculi. Third-order neurons from the inferior colliculus ascend to the medial geniculate body of the thalamus, where fibers from fourth-order cell bodies form the auditory radiation to the transverse temporal gyrus of Heschl. Heschl’s gyrus is usually identified as a single gyrus and less commonly as two gyri, at the posterior aspect of the superior temporal gyrus. It can be identified on sagittal, axial, and coronal planes at MRI (Fig. 8.9).16 Heschl’s gyrus roughly corresponds to the primary auditory cortex (A1) in Brodmann’s area 41 and 42, with both left and right hemispheres receiving information from each cochlea.16,17 Just as the motor cortex has the “homunculus” arrangement of motor tasks, the auditory cortex has a tonotopic organization of frequencies also.18 The superior surface of the temporal lobe behind Heschl’s gyrus is called planum temporale and is probably related to the cortical representation of language.18 Functional MRI (fMRI) studies, positron emission tomography (PET), and amobarbital angiographic studies have shown laterality for listening and speech. In most people, regardless of preferred handedness, the left temporal lobe appears to be better for processing language information, and the right temporal lobe more important for processing non-verbal natural sounds (Fig. 8.12).19–23
Imaging the Internal Auditory Canal, Cerebellopontine Angle, and Vestibulocochlear Nerve
There are two imaging tools available to the radiologist when evaluating a patient for SNHL. Computed tomography is ideal for evaluation of the inner ear and otic capsule when bone pathology is suspected such as with traumatic SNHL or with otodystrophies (otic capsule dysplasias such as osteogenesis imperfecta), or suspected acquired bone pathology such as Paget’s disease or cochlear otosclerosis. In these cases, CT evaluation of the temporal bone should be performed with thin sections of the order of 1 mm in thickness, preferably in two planes directly obtained, or with coronal reformations of axial slices. Images should always be processed with a high spatial frequency (bone) algorithm and viewed with wide windows (window width of ∼4000, window level ∼500 to 600).
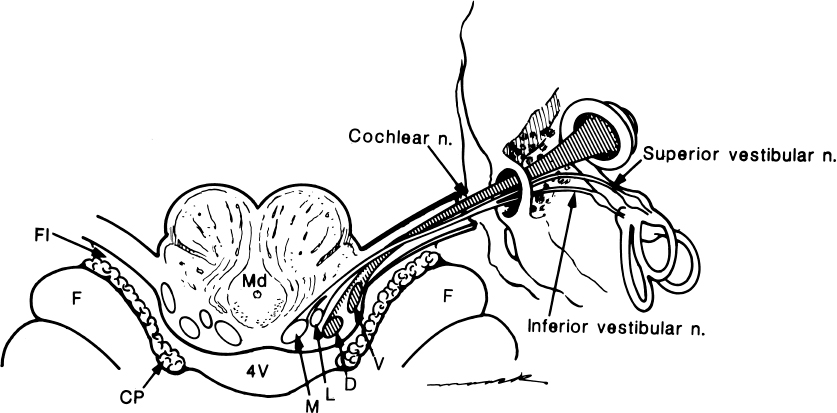
Fig. 8.10 Drawing of the vestibulocochlear nerve, root entry zone, and nuclei within inferior cerebellar peduncle (restiform body). Facial nerve has not been included in drawing. The cochlear nerve is drawn with hatch marks. Note the close relationship of the cochlear nuclei to the lateral recess of the fourth ventricle and foramina of Luschka. V, ventral cochlear nerve; D, dorsal cochlear nucleus; L, lateral vestibular nucleus; M, medial vestibular nucleus; Md, medulla; 4V, fourth ventricle; Fl, foramina of Luschka; F, flocculus; CP, choroid plexux. The cochlear nuclei are normally found on the superficial edge of the inferior cerebellar peduncle. Although the nuclei themselves are not visible, finding the inferior cerebellar peduncle contour on high-resolution MR imaging can localize their approximate location.
Most other causes of SNHL warrant MRI to best evaluate the nerves of the IAC and the CPA, as well as to review the central auditory pathway from the inferior cerebellar peduncle to the auditory cortex at the superior temporal gyrus. The MRI study for evaluation of SNHL can be performed as a “brain-IAC study” with the administration of gadolinium to evaluate for IAC or CPA masses and for complete coverage of the central auditory pathway. In some practices, high-resolution, thin-section T2WI “screening examination” is performed alone; however, this should only be performed in cases of uncomplicated unilateral SNHL when a vestibular schwannoma is strongly suspected by an otologist. More frequently, the high-resolution T2WI is employed in addition to a brain-IAC study.
Brain–Internal Auditory Canal Protocols
A protocol to encompass the central auditory pathway and IAC typically consists of whole brain sagittal and axial T1WIs, axial T2WIs, and fluid attenuated inversion recovery (FLAIR) images, and axial whole brain gadolinium-enhanced axial T1WIs. Axial and coronal postgadolinium thin-section (3 mm) images should be obtained through the IACs.
Gadolinium-enhanced MRI has a high sensitivity and specificity for the detection of vestibular schwannoma, which is the most common pathology found in cases of acquired, unilateral sensorineural hearing loss. The use of three-dimensional spoiled gradient recalled (3D SPGR) sequences allows 1.5 mm section thickness and thus better depiction of the tumor and its relation to the brainstem or the fundus of the IAC.
Screening Internal Auditory Canal Protocols
High-resolution, heavily T2WI of the IAC and CPA has been suggested as an alternative to whole brain gadolinium imaging as a significant reduction in scan time and cost.24 It should only be used alone as a screening study in selected cases of unilateral isolated SNHL, and is usually performed after complete clinical examination and audiometry. It should be recognized that this screening protocol has the potential to miss small tumors, many of which would be followed clinically at this size.
The choice of a T2-weighted sequence is a heavily debated, discussed, and published topic. Essentially there are two types with many variations: fast spin-echo (FSE) based sequences and gradient echo (GRE) based sequences. Initially, GRE-based sequences offered thinner slices for improved spatial resolution; however, these were hampered by local magnetic field susceptibility artifacts, which the FSE-based sequences are not. The recent advances in both types of sequences have been in an attempt to minimize scan time without loss of signal-to-noise (SNR) or resolution to make the study as acceptable to patients as possible. Below is a short summary of some of the myriad of sequences available. For the best spatial resolution and SNR, surface coils, such as the temporomandibular joint (TMJ) coil, can be used.
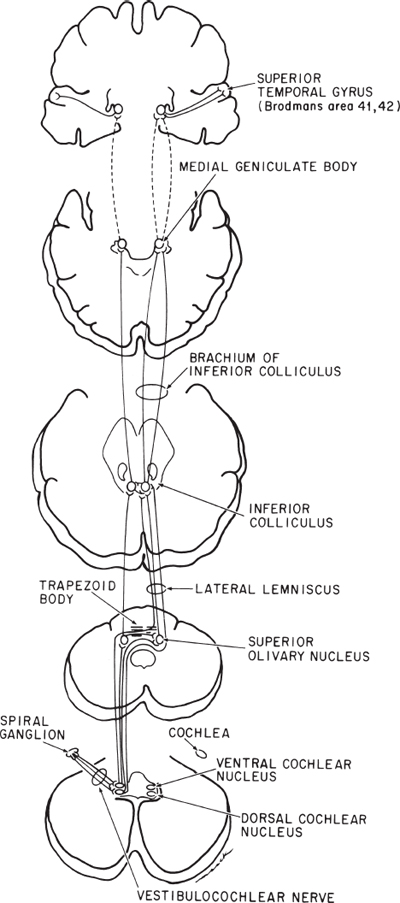
Fig. 8.11 Diagram of the entire course of the acoustic pathway from its end organ, the cochlea, to its central processing area, the superior temporal gyrus. Notice the majority of centrally projecting fibers cross in the midpontine tegmentum in the trapezoid body.
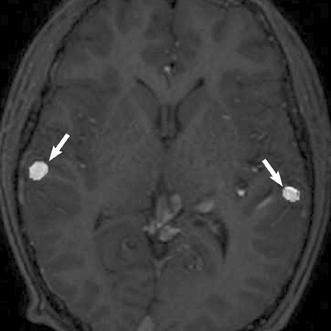
Fig. 8.12 Primary auditory cortex. Functional magnetic resonance imaging study in a teenager with a large left frontoparietal pericentral arteriovenous malformation, performed for evaluation of risks associated with possible treatment. Auditory evoked potentials localize bilaterally to the posterior superior temporal gyri (arrows), with the right being more anterior than the left side.
- 3D T2 FSE. Very good quality thin-section images; however, scan time is considered excessively long.
- 3D T2 FSE with fast recovery (FRFSE) (driven equilibrium pulse). The addition of 180 degrees (y plane) and –90 degrees (x plane) pulses at the end of the echo train flips residual spins from the transverse plane to the longitudinal axis. It thus enhances the signal of slowly recovering tissues such as CSF, allowing increased contrast resolution or a shorter time-to-repetition (TR) and therefore significant time reduction.25
- 3D fast asymmetric spin echo (FASE). This is a half Fourier rapid acquisition with relaxation enhancement (RARE) type sequence, based on 3D FSE, but with a long echo train length (ETL).26 A modification of this, the zero-fill interpolated (ZIP) fast recovery 3D FASE sequence, can significantly reduce the scan time of the FASE sequence.
- 3D constructive interference in the steady state (CISS). This sequence allows better spatial resolution than the initially available FSE sequences with minimization of CSF flow artifacts and a short scan time. The sequence is acquired by averaging two 3D Fourier transformed datasets, one with an alternating radiofrequency (RF) pulse and the other with a nonalternating RF pulse. The signal acquired is partially from GRE mechanisms, resulting in some problems with susceptibility artifacts in the membranous labyrinth mimicking pathology, and significant image degradation with excessive dental amalgam.27–29
- 3D segment interleaved motion compensated acquisition in the steady state (3D-SIMCAST). This sequence uses more than two-directional phase cycling and a shorter TE by fractional echo, resulting in reduced susceptibility artifacts but increased scan time. Nearly isotropic voxels are created after interpolation.30
- 3D true-fast imaging with steady-state precession (FISP) and a variant of this, full balanced steady-state coherent imaging (FBSSC). These have also been recommended. The FBSSC requires a high-performance gradient system but allows for very short TR and TE values, and therefore a shorter scan time.
Many of these 3D sequences can be postprocessed to produce maximum intensity projection or 3D volume-rendered or endoscopic-type images of the inner ear.25,26,31,32
3T Magnetic Resonance Imaging
The emergence of ultra-high field (3.0 Tesla) MRI has heralded the possibility of significant improvements in signal to noise and spatial resolution in IAC imaging. There are several technical and safety issues that must be addressed.
- A basic consideration in the modification of any neuro-radiology sequence for 3T imaging is that the T1 of CSF will be prolonged (as T1 α field strength x gyromagnetic ratio). A longer TR is thus required which directly increases scan time (as scan time TR).
- Susceptibility artifacts are more pronounced at 3T as compared with 1.5T, making the GRE-based sequences (e.g., 3D-CISS, 3D-SIMCAST, 3D true FISP) potentially more problematic.
- The specific absorption rate (SAR) is increased with a higher field strength, and this is particularly so with GRE-based sequences, which have a higher SAR at 3T than 3D FSE-based sequences. The higher field SAR concerns will also limit the use of a longer echo train length (ETL) with FSE-based sequences. It has been suggested that the fast recovery FSE will help to offset this problem.33
- Although TMJ surface coils are advantageous with high-resolution imaging to improve the signal-to-noise ratio, these are receive-only coils requiring the whole body coil for RF transmission. This is associated with an increase in SAR, which is within acceptable limits at 1.5T, but becomes unacceptable at 3T. Kocharian et al have suggested the use of a hybrid phased array coil (two circular surface receive-only coils combined with a transmitreceive birdcage head coil). Using this coil and the 3D FR FSE high-resolution sequence, higher signal-to-noise images can be obtained in less than 9 minutes.32
Pathology
Lesions of the Internal Auditory Canal and Cerebellopontine Angle
Among the lesions of the auditory pathway causing SNHL and detected at imaging, the IAC–CPA complex is the most common location, and vestibular schwannoma is the single most common pathologic entity responsible for SNHL.34,35 Meningioma and epidermoid are the next most frequently found CPA masses, with these three lesions responsible for more than 90% of all CPA lesions. The remaining < 10% are made up of more than 60 different CPA masses, reflecting a wide variety of pathologies.36 For discussion it is perhaps best to divide these lesions into the diagnostic groups of congenital and acquired neoplastic and nonneoplastic entities, with an emphasis on the “big three” masses of the CPA.
Congenital Internal Auditory Canal Malformations
The IAC essentially forms by default early in gestation. The presence of a vestibulocochlear nerve at the medial aspect of the otic capsule inhibits cartilage formation leaving a canal connecting the inner ear to the CPA. The facial nerve and its canal develop independently of the vestibulocochlear nerve but become enveloped in the developing otic vesicle. If the vestibulcochlear nerve is absent due to otic capsule malformation, such as a Michel anomaly with absence of the cochlea, the IAC cannot form and the canal becomes the caliber of the facial nerve alone.37,38 In early tomographic and CT studies, an IAC measuring less than 2 mm in diameter was defined as IAC aplasia.39,40 High-resolution MRI obtained with oblique sagittal thin slices perpendicular to the course of the IAC allows demonstration of this small canal, but also absence of the vestibulocochlear nerve (Fig. 8.13).4,41 A small or hypoplastic cochlear nerve may also be found, where the nerve is similar in size or smaller than either of the vestibular nerves. A normal-sized IAC with an absent or small cochlear nerve suggests that the nerve abnormality is acquired after formation of the internal auditory canal. This has been observed in patients with acquired SNHL such as labyrinthitis ossificans.4 Abnormality of vestibulocochlear nerve caliber may have important implications in patients with bilateral SNHL requiring cochlear implantation on one side, and it has been suggested that the more robustappearing nerve be selected. Absence of the cochlear nerve is a contraindication to implantation.40
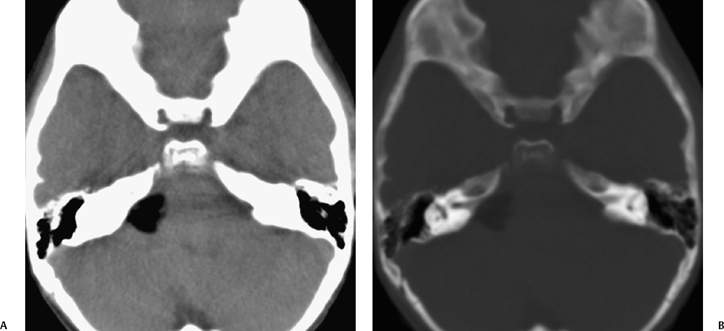
Acquired narrowing of the IAC secondary to bony over-growths, either exostoses or osteomas, has also been described.41 Although these bony lesions are particularly rare, they may clinically mimic vestibular schwannomas by compression of the vestibulocochlear nerve (Fig. 8.14). Hearing loss and vestibular symptoms, including dizziness and vertigo, may improve after surgical removal of the bony lesion.
An enlarged IAC (> 9 mm) may be seen as an incidental finding (Fig. 8.15), but has been described with the X-linked stapes gusher. This syndrome is typically seen in male patients with congenital mixed hearing loss and is characterized by the presence of a fixed stapes footplate and “gushing” of perilymph at attempted stapedectomy.42 An enlarged or “bulbous” IAC has been described at CT imaging, with incomplete separation of the basal turn of the cochlear from the IAC fundus (deficient lamina cribrosa) and widened labyrinthine and proximal tympanic facial nerve canals.43,44
Congenital Internal Auditory Canal and Cerebellopontine Angle Masses
Epidermoids
Epidermoids are benign extraaxial, intradural tumors that are most frequently located in the CPA. These masses arise as ectopic inclusions of epithelial cells at the time of neural tube closure around 3 to 5 weeks of gestation.45 Although they are congenital, they are also slow-growing masses that enlarge by accumulation of keratin and cholesterol as the lining epithelium desquamates. Malignant transformation is extremely rare.46
Epidermoids typically present in the third to seventh decades as irregular lobulated masses with prolonged symptoms due to involvement of nerves or displacement of the brainstem and cerebellum. These tumors tend to surround and encase neurovascular structures such as CN V, VII, and VIII, and the basilar artery. A tendency for adherence to these structures may also make complete surgical resection difficult.47
Focal calcifications are uncommonly found at CT. Epidermoid tumors are usually similar to water attenuation and do not typically enhance following contrast administration. At MRI, epidermoids appear similar to CSF in intensity on T1-weighted and T2-weighted sequences, which can make differentiation from arachnoid cysts difficult. However, on close inspection of these images and some manipulation of the windowing, one frequently may find tiny internal septations within these tumors. In addition, frequently on FLAIR imaging epidermoids are higher in signal intensity than CSF (Fig. 8.16).48 Another important sequence that may help in differentiating epidermoids from arachnoid cysts is diffusion weighted imaging (DWI) where epidermoids tend to be markedly hyperintense compared with arachnoid cysts.49,50 This also serves as a useful imaging feature for detection of postoperative residual tumor (Fig. 8.17).51
Arachnoid Cysts
An arachnoid cyst is a round, smooth mass contained within, but separated by a thin membrane from, the subarachnoid CSF. These lesions probably develop through failure of embryonic meninges to merge, resulting in a subarachnoid CSF-filled cyst.52 Pathologically, a thin membrane of flattened, nonneoplastic arachnoid cells is found and the cyst fluid is identical to CSF. Arachnoid cysts are most frequently found in the middle cranial fossa, with up to 5% located in the posterior fossa.53 Their clinical presentation may be related to compression of neurovascular structures (such as CN V, VII, or VIII) of the posterior fossa, though arachnoid cysts are frequently incidental findings at imaging.
At CT imaging, CPA arachnoid cysts are hypodense, nonenhancing rounded masses without calcifications. Scalloping of adjacent bone is more frequently reported in middle cranial fossa lesions. At MRI, arachnoid cysts are homogeneous and isointense to CSF on all sequences, including FLAIR and DWI, which differentiates them from epidermoid tumors (Fig. 8.18).49
Lipomas
Intracranial lipomas are hamartomatous lesions composed of mature adipose tissue, which are thought to result from in utero aberrant differentiation of the leptomeningeal component of the meninx primitiva.54 They are most commonly found as supratentorial midline lesions and are frequently associated with cerebral malformations, though are rarely themselves symptomatic. Lipomas are rarely found in the CPA, and in the posterior fossa they are usually isolated lesions not associated with other abnormalities; however, they can become symptomatic.55 CPA lipomas may present with symptoms related to cranial nerve irritation, such as trigeminal neuralgia (CN V), hemifacial spasm (CN VII), or vertigo and hearing loss (CN VIII).55,56 Symptoms such as hearing loss, vertigo, and tinnitus related to involvement of CN VIII are the most frequent presentation. At imaging, neurovascular structures appear to pass through the lipoma; at surgery, these structures are frequently found to be densely adherent to these elements. Hence, conservative management of lipomas is often preferred unless the symptoms are disabling and uncontrollable. Cranial nerve decompression or vestibular transection may then be considered for treatment.55,56
On CT imaging, lipomas are of fat attenuation (-50 to -100 Hounsfield units) and do not show contrast enhancement (Fig. 8.19). Lipomas on MRI follow the signal intensity of fat, with suppression of signal on chemical selective fat-saturation images (Fig. 8.20). CPA lipomas have been reported in association with intravestibular lipomas in patients with SNHL. It is proposed that this occurs very early in gestation from either direct incorporation of the meninx primitiva in the developing otocyst or from neural transport of meninx primitiva along the developing CN VIII.57
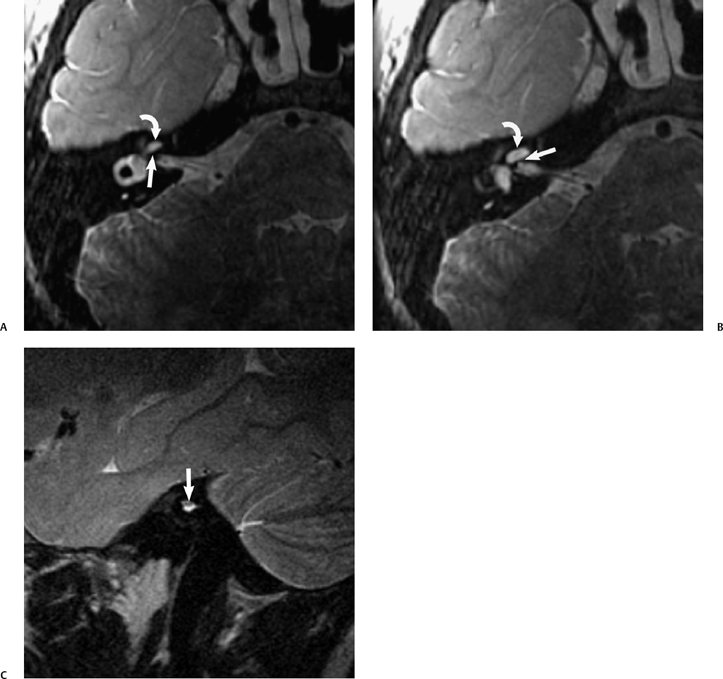
Fig. 8.13 Absent CN VIII. (A,B) Child with unilateral congenital sensorineural hearing loss has malformation of the right cochlea (curved arrow) and a bony bar at the expected cochlear aperture (arrow). The internal auditory canal (IAC) also appears to be narrow on these axial images. (C) This is confirmed on the oblique sagittal T2 MR where a small IAC is evident containing one nerve bundle superiorly (arrow), which represents the facial nerve.
Other hamartomatous lesions of the brain are of glioneural origin. These are rare lesions with few CPA hamartomas reported in the literature.58 Some authors describe only slight contrast enhancement, whereas others describe intense enhancement, mimicking a vestibular schwannoma.58,59 Like lipomas, hamartomas have a tendency for adherence to neurovascular structures making resection technically difficult or not possible (Fig. 8.21).
Acquired Neoplastic Internal Auditory Canal and Cerebellopontine Angle Pathology
Vestibular Schwannoma
Vestibular schwannoma, also known as acoustic neuroma, is the most common CPA tumor and the most common lesion found in patients with unilateral acquired SNHL.24,60 This benign tumor is sometimes referred to as a neurinoma or neurilemmoma. Despite the moniker “acoustic neuroma,” these tumors do not arise from the cochlear nerve. Vestibular schwannoma is considered the correct terminology as the tumor arises from Schwann cells covering the lateral aspect of either the superior or inferior vestibular.61 Most schwannomas arise lateral to the junction of Schwann cells and neuroglial cells (i.e., lateral to the Obersteiner-Redlich zone) in the IAC or at the porus acusticus.62 Uncommonly, schwannomas arise in the CPA and do not have an IAC component.63,64
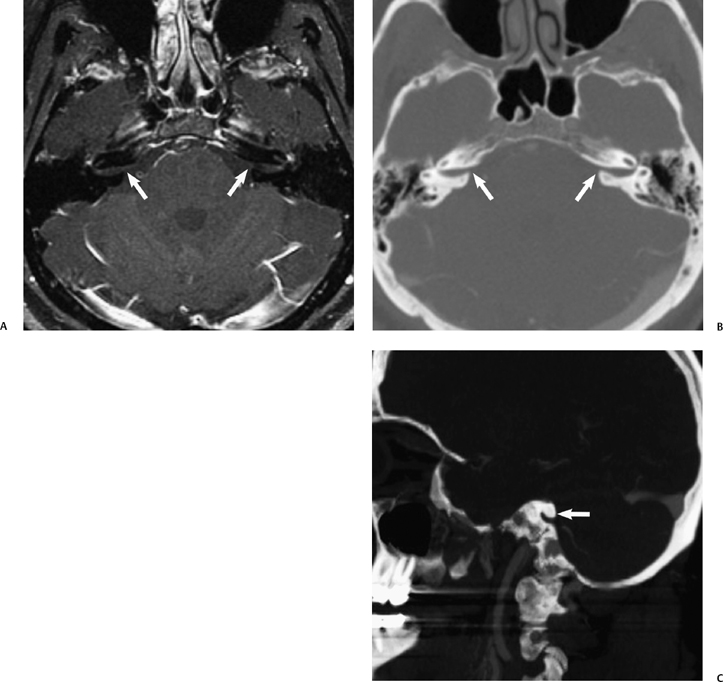
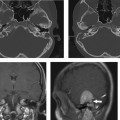
Fig. 8.14 Acquired stenosis of the internal auditory canal (IAC) porus. Adult with increasing bilateral but predominantly left sensorineural hearing loss and dizziness. (A) The postgadolinium fat saturated magnetic resonance image shows no evidence of an IAC or cerebellopontine angle mass, but an unusual contour of the porus on both sides (arrows). A computed tomography angiogram performed several months prior shows this similar bony overgrowth of the margins of the porus in (B) the axial and (C) reformatted left sagittal plane. The patient had bony exostoses elsewhere, and this was presumed to be a proliferative bone abnormality.
Adults with vestibular schwannomas typically present with slowly progressive hearing loss with or without tinnitus, which on audiometric testing is a high frequency SNHL.24 Schwannomas with a large CPA component may also impinge on the facial or trigeminal nerves resulting in additional cranial neuropathies, or compress the pons and produce fourth ventricular obstruction. The presence of bilateral vestibular schwannomas is diagnostic of neurofibromatosis type II (NF2). Identification of a single vestibular schwannoma in a child (< 20 years) should raise suspicion for the possibility of NF2 (Fig. 8.22).
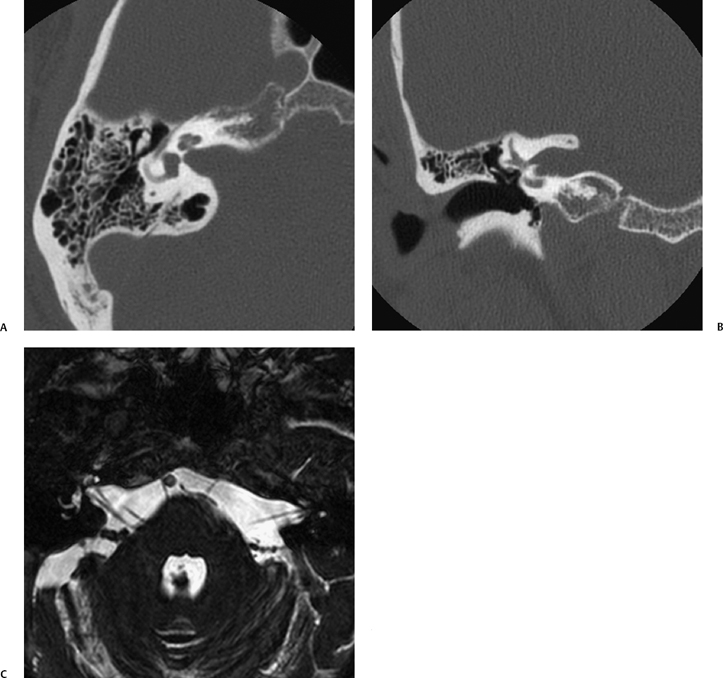
Fig. 8.15 Incidental large internal auditory canal (IAC). ”Patulous” internal auditory canals were noted on a computed tomography (CT) scan performed of the sinuses, which prompted a temporal bone CT (A,B) and MRI (C, constructive interference in steady state [CISS] sequence), where no abnormality other than the large canals was evident. The patient was asymptomatic and the anomaly symmetric.
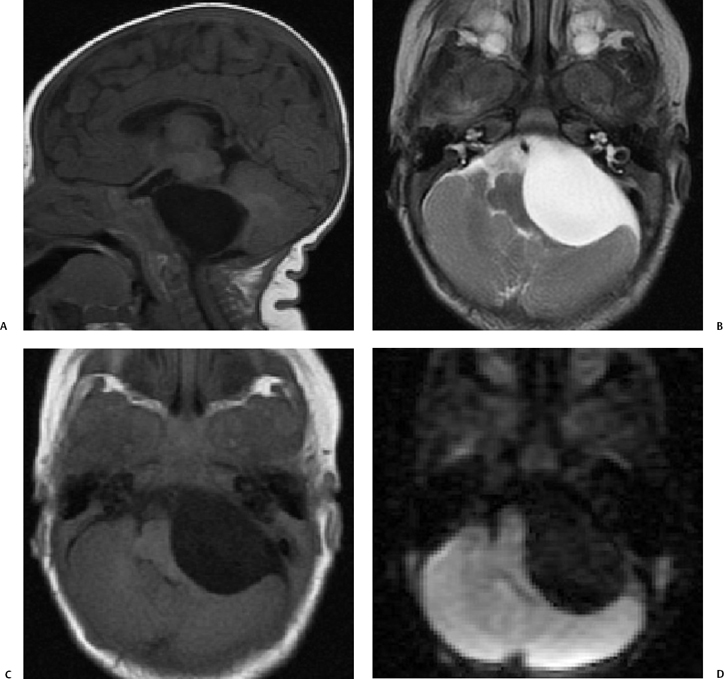
The classic large IAC–CPA schwannoma is described as an “ice cream and cone” shaped lesion with a larger, homogeneous, enhancing mass in the CPA, which has prolapsed from a smaller funneled IAC component, producing widening of the porus (Fig. 8.23). Large IAC–CPA vestibular schwannomas may have a cystic CPA component, which appears on CT as an area of low density, and on MRI as high T2 signal intensity (Fig. 8.24). These tumors have a much faster growth rate and a less favorable surgical outcome with a higher incidence of postoperative facial nerve dysfunction.65–67 This intratumoral cystic component should be distinguished from an extrinsic arachnoid cyst, which may be found in association with vestibular schwannomas in a small percentage of cases (Fig. 8.25 and Fig. 8.26). The CPA component of the neoplasm may sometimes appear hemorrhagic with a heterogeneous predominantly hyperdense appearance on CT, and more mixed signal intensity on T1WIs and T2WIs. Rarely, patients with vestibular schwannoma may present acutely with vertigo and vomiting from acute hemorrhagic expansion (Fig. 8.27 and Fig. 8.28).6,68
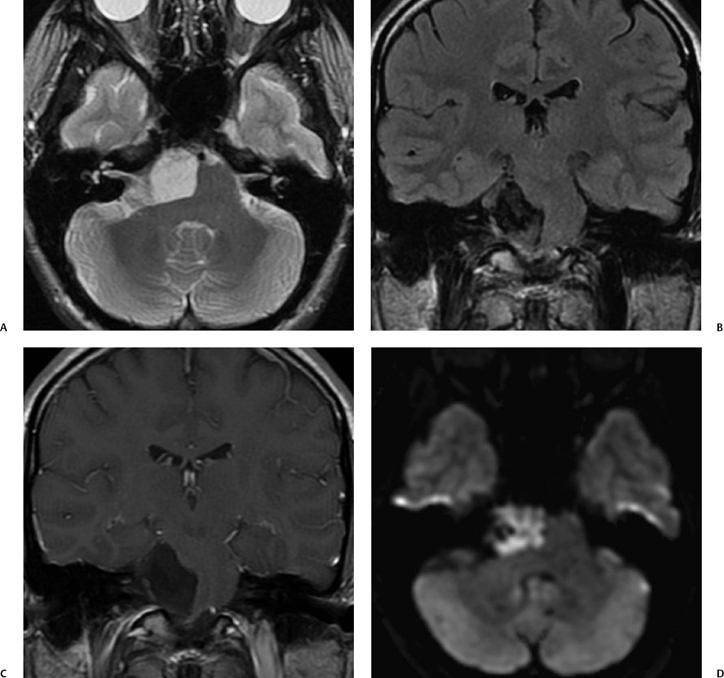
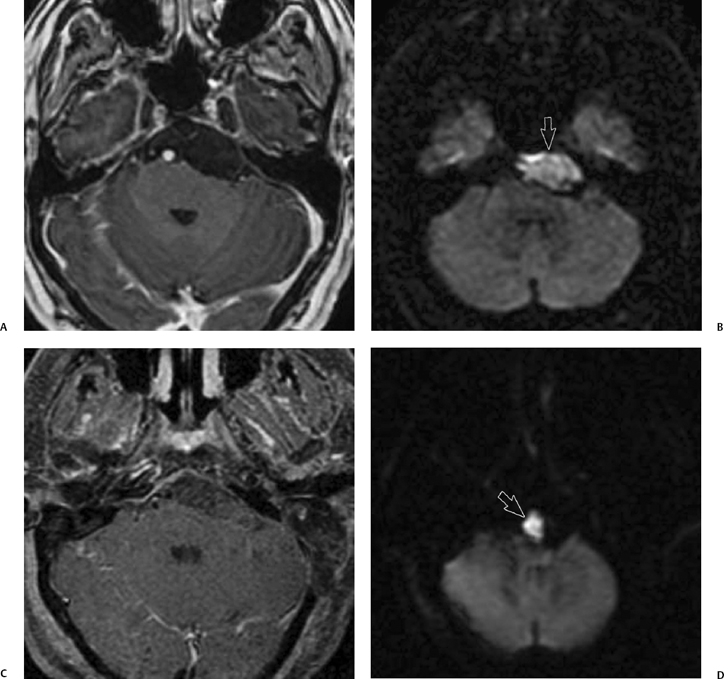
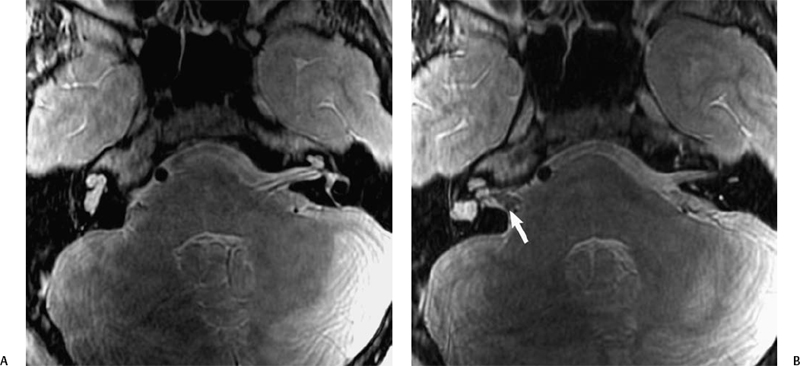
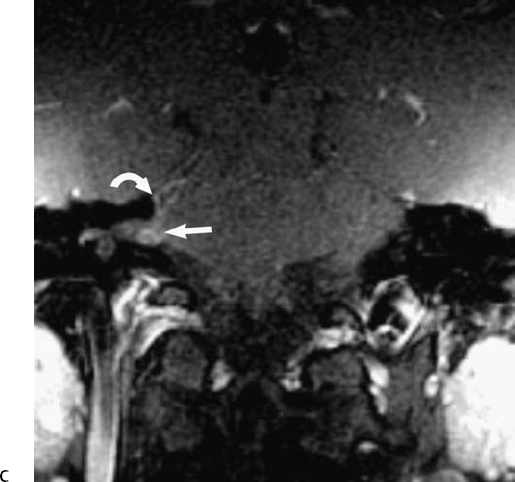
The intracanalicular portion of a schwannoma may extend laterally from the fundus to the cochlea or vestibule, or both, producing what has been described as a dumbbell schwannoma.69,70 This can be demonstrated with either high-resolution T2WI or with T1-weighted gadolinium enhanced images (Fig. 8.29). As hearing is never salvageable, these lesions are managed conservatively unless disabling vestibular symptoms or a large CPA mass is present.69 Rarely, a schwannoma may be localized only to the cochlea (Fig. 8.30). These are also treated conservatively.
In the era of high-resolution MRI of the IAC–CPA, it is most frequent to be searching for a small (< 10 mm) homogeneous, entirely intracanalicular schwannoma. On high-resolution T2WI, these vestibular schwannomas appear as oval-shaped, low-signal-intensity masses in close association with the nerves in the IAC. Strong contrast enhancement is observed with T1WI and gadolinium (Fig. 8.31). Early detection of schwannomas while they are still small is an important step to improve the surgical outcome, and specifically to preserve residual hearing and facial nerve function.63
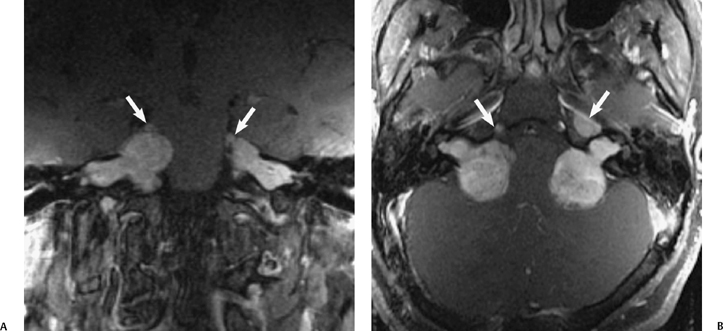
Fig. 8.22 Neurofibromatosis type II. (A) Coronal and (B) axial gadolinium enhanced T1-weighted magnetic resonance images show bilateral lobulated, enhancing cerebellopontine angle–internal auditory canal (IAC) vestibular schwannomas extending to the fundus of the IAC bilaterally, and compressing the pons. Note also the presence of bilateral enhancing masses involving the fifth cranial nerves and Meckel’s cave (arrows) consistent with schwannomas.
There are currently two options for treatment of vestibular schwannomas, both having merit and risks. Surgical treatment has been the gold standard of management with three main microsurgical techniques, described below. There has been increasing use of gamma knife radiosurgery, for the treatment of small- to moderate-sized tumors. With gamma knife radiosurgery, multiple radiation beams are focused on the center of the schwannoma using cobalt sources (12 to 16 Gy). This method has largely been reserved for schwannomas where the CPA component is < 3 cm in size and without large cystic components, and for patients who for other health reasons might be considered poor operative candidates.63,71,72 Hearing preservation and facial nerve function posttreatment appear to be comparable for the techniques, although the surgical and radiation therapy literature are conflicting as to the better outcome with each.73,74 The surgical literature cites the small risk of malignant degeneration of vestibular schwannomas and the lack of long-term data thus far with gamma knife treatment, in addition to the greater complexity of surgical resection if gamma knife fails to adequately treat a schwannoma.72,75
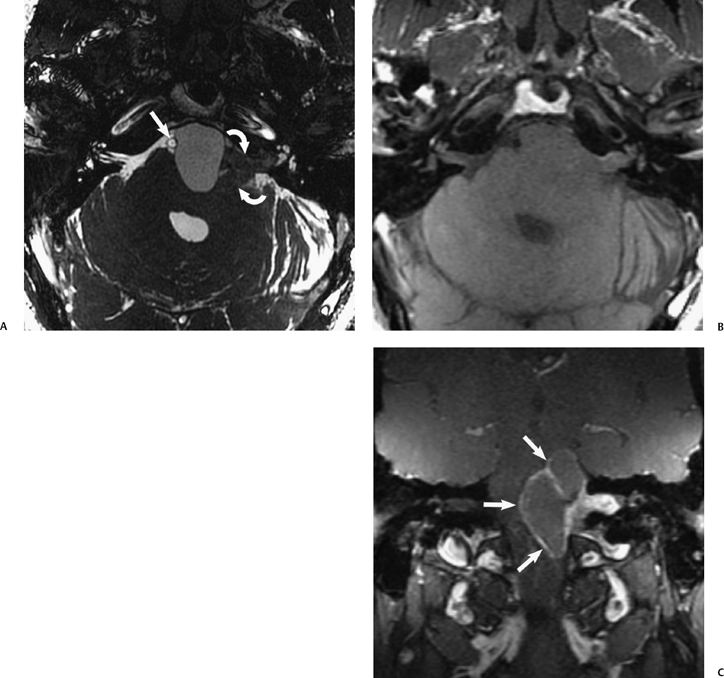
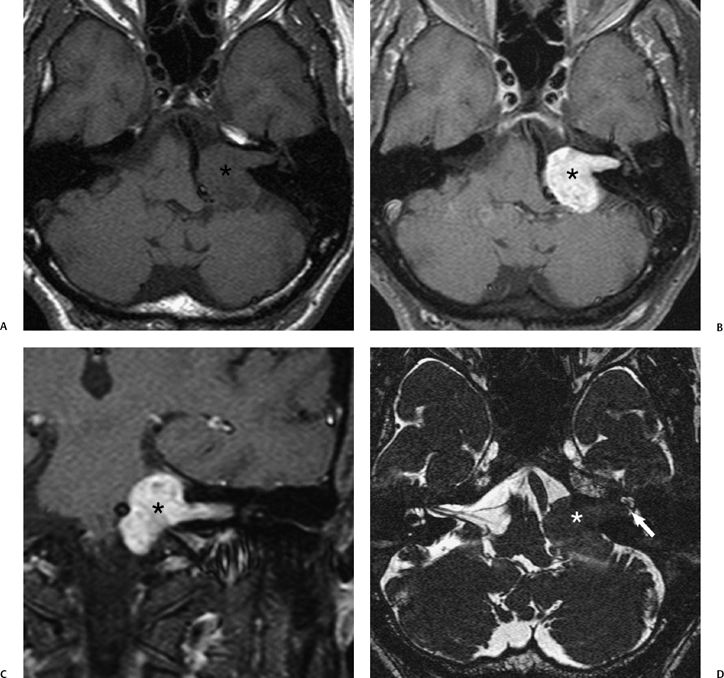
Fig. 8.24 Vestibular schwannoma with cystic change. (A) High-resolution axial T2-weighted magnetic resonance image (T2WI) shows a complex left cerebellopontine angle–internal auditory canal (CPA-IAC) mass with a solid component (curved arrows) and a proteinaceous cystic component in the prepontine cistern that displaces the basilar artery (arrow) to the right. (B) The cystic component is almost isointense to brain on unenhanced axial T1-weighted images. (C) On gadolinium-enhanced MR, there is rim enhancement of the cystic component and intense enhancement of the solid CPA-IAC component.
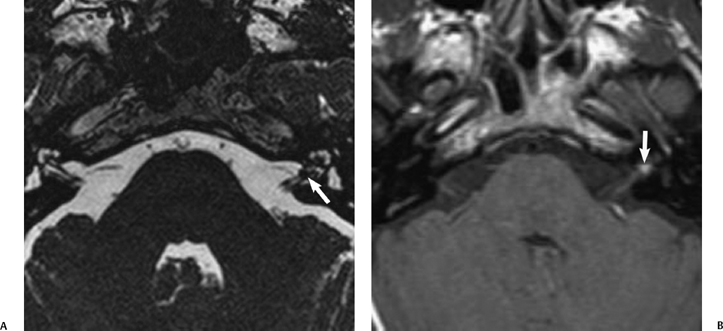
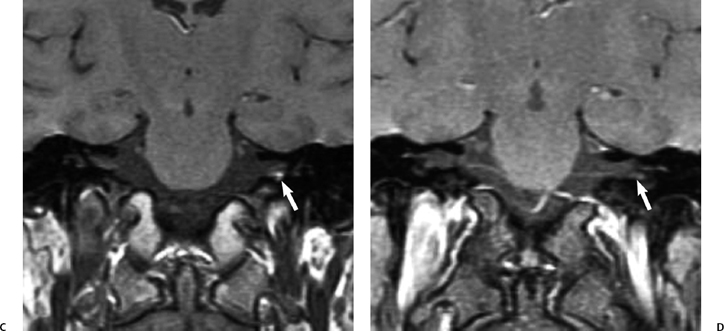
The surgical management for vestibular schwannoma is determined by several factors including tumor size and location, the patient’s age, health, and hearing. In patients with high surgical risk, advanced age, small tumors, or minimal symptoms, conservative management (“wait and scan”) with 6-month interval follow-up MRI evaluations may be acceptable as these tumors are usually slow-growing.64,71,74,75 Small intracanalicular lesions are often resected with the aim of conservation of current hearing and the intent of preservation of facial nerve function. A subtemporal middle cranial fossa approach allows excellent access to the entire length of the IAC, though it is complicated by the increased vulnerability of the more superficially situated facial nerve and limited access to the posterior fossa for a large CPA component. A retrosigmoid approach allows wide access to the posterior fossa, which may be important for a large IAC-CPA tumor, but only exposes the medial two thirds of the IAC. Extension of tumor to the lateral third of the IAC prohibits complete resection via this approach. Thus, the most lateral extent of the schwannoma is an important feature to describe at MRI evaluation.63,77 In addition, of the most significant radiological features correlating with poor hearing outcome is the presence of tumor at the IAC fundus.63,78–80 Evaluation of the extent of IAC and fundal involvement with 3D heavily T2WIs (characterized by loss of CSF bright signal) has been found to be more accurate than gadolinium-enhanced MRI, and is recommended for complete presurgical evaluation. An additional finding, which has been observed with certain heavily T2-weighted sequences, is a relative decrease in T2 signal intensity of the fundal CSF “cap” or of the membranous labyrinthine, as compared with the unaffected side. It has been postulated that this is due to impaired vascular supply from impaction of the tumor in the IAC. This finding has also been shown to correlate with a poorer hearing outcome (Fig. 8.32).80 It has not been observed with all 3D-FSE sequences (personal communication, Jan W. Casselman, April 2005).
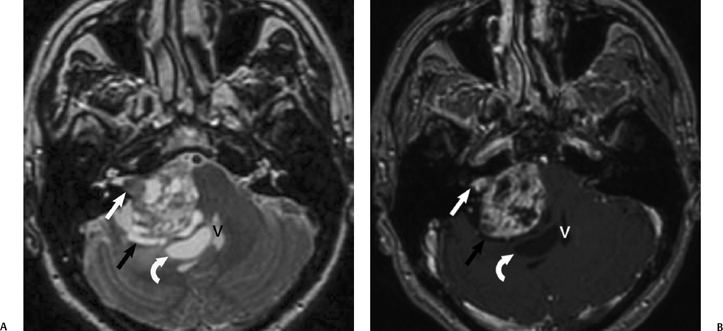
Fig. 8.25 Vestibular schwannoma with intratumoral cystic change. (A) Axial T2-weighted and (B) corresponding axial enhanced T1-weighted magnetic resonance image show a heterogeneous, predominantly hyperintense right cerebellopontine angle mass with a nodular, solidly enhancing intracanalicular component (white arrow). The mass has a surrounding cerebrospinal fluid cleft (black arrow), illustrating that it is extraaxial. The fourth ventricle (V) is displaced to the left. An adjacent associated arachnoid cyst is also noted (curved arrow), without rimenhancement.
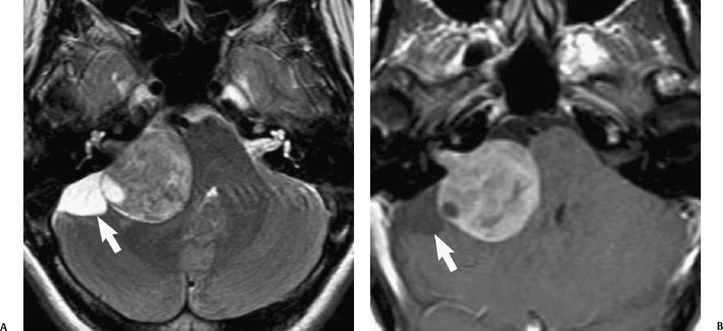
Fig. 8.26 Vestibular schwannoma and coexistent arachnoid cyst. (A) Axial T2-weighted and (B) corresponding enhanced axial T1-weighted magnetic resonance image show a large right cerebellopontine angle–internal auditory canal mass distorting the right brachium pontis. There is a cerebrospinal fluid intensity component (arrow) posterior to the schwannoma consistent with an arachnoid cyst. (Courtesy of Andy Whyte, MBChB, FRCR, FRANZCR.)
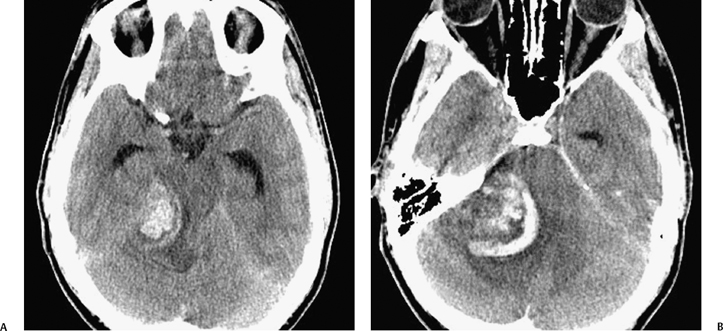
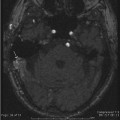
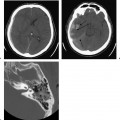
Fig. 8.27 Hemorrhagic vestibular schwannoma. (A,B) Noncontrast axial computed tomography images show a heterogeneous, hyperdense right cerebellopontine angle mass with significant mass effect upon the pons, right cerebellar hemisphere, and fourth ventricle, with adjacent low-density edema and hydrocephalus (dilated temporal horns). The patient had a prior diagnosis of vestibular schwannoma and presented with new headache and somnolence.
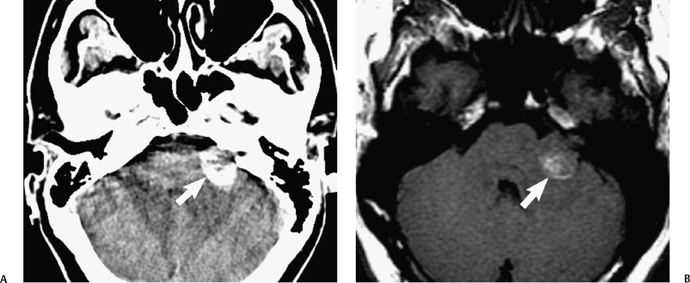
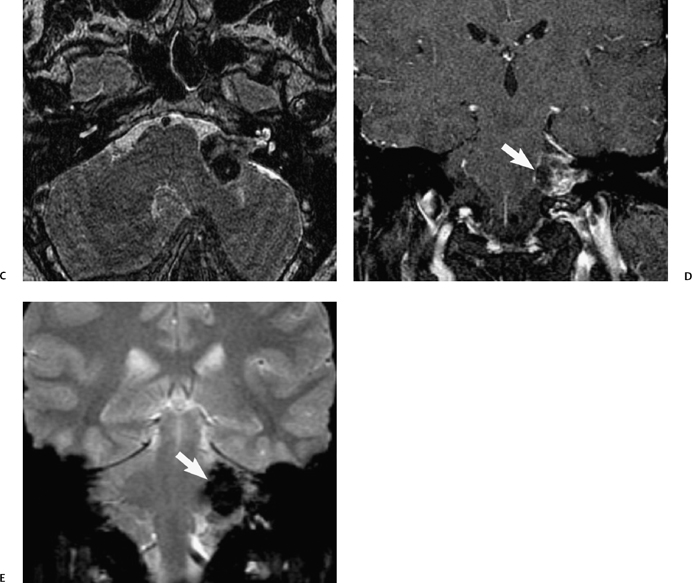
Fig. 8.28 Hemorrhagic vestibular schwannoma. (A)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree